Cell migration, a key property of live cells, is the process by which cells move from one location to another. Cells may migrate in a single cell form or by multicellular movement labeled collective migration or cell streaming. The single cell migration can be classified into amoeboid movement (characterized by cells moving as rounded, ellipsoid bodies without the involvement of focal adhesions and cell attachment but with the aid of propulsive blebs) and mesenchymal movement (involving strong focal attachment to the extracellular matrix, cytoskeletal contractility and elongated spindle-like cell bodies). Collective migration is characterized by the movement of a cellular cohort through the ECM with the preservation of functional cell-cell junctions. It is critical for normal development, immune response, and disease processes such as cancer metastasis and inflammation.
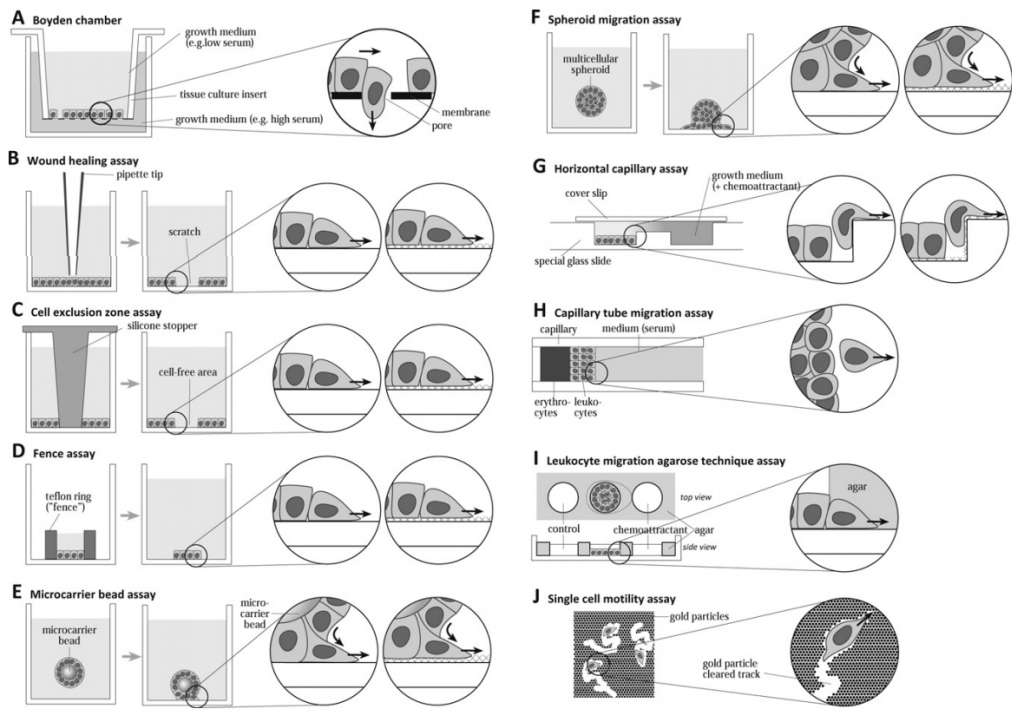
There are numerous ways to study cell migration, such as transwell migration assay(Boyden chamber assay), wound healing assay (scratch assay), cell exclusion zone assay (platypus migration assay), fence assay, microcarrier bead assay, leukocyte migration agarose technique assay (LMAT assay), spheroid migration assay, single cell motility assay, etc. And we will introduce two common methods in detail.
Boyden Chamber Assay
The Boyden chamber assay is suitable for the quantitative analysis of different migratory responses of cells, such as chemotaxis, haptotaxis, and chemokinesis. This relatively reliable and time-saving assay allows one to have versatility in conducting motility experiments and allows for cell-motility analysis on a basis without consideration of the effect from cell proliferation and cell-cell interactions.
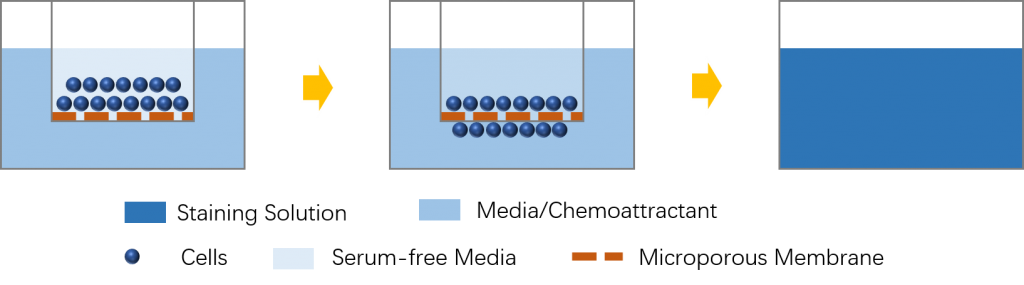
The Boyden chamber assay is based on a chamber of two medium-filled compartments separated by a microporous membrane. For the induction of the chemotactic or haptotactic response of cells, attractants are added to the lower compartment of the chamber. While for the induction of chemokinesis, equal concentrations of the agent are added on both sides of the membrane. And cells are placed in the upper compartment and can migrate through the pores of the membrane into the lower compartment. After the appropriate incubation time, the microporous membrane between the two compartments is fixed and stained, and the number of cells that have migrated to the lower side is determined.
There are several steps involved in the Boyden chamber assay, including cell preparation, loading and assembling the chamber, staining the membrane, counting the cells, and cleaning the chamber. During a Boyden chamber assay, some important factors need to be considered, such as the number of cells to load on the chamber, the pore size of the membranes, the type and concentration of the attractant, and the incubation time.
In vitro wound-healing assay (scratch assay)
This method is a non-demanding and cheap assay to study the migration of cells on 2Dsurfaces and mimics cell migration during wound healing in vivo. In this assay, when the cells grow to a single layer state, a blank area is created in the monolayer cells, called "scratches". The cells at the edge of the scratch will gradually enter the blank area to allow the "scratches" to heal. Therefore, the basic steps involved in the methods include creating a “scratch” in a cell monolayer, capturing the images at the beginning and at regular intervals during cell migration to close the scratch, and comparing the images to quantify the migration rate of the cells.
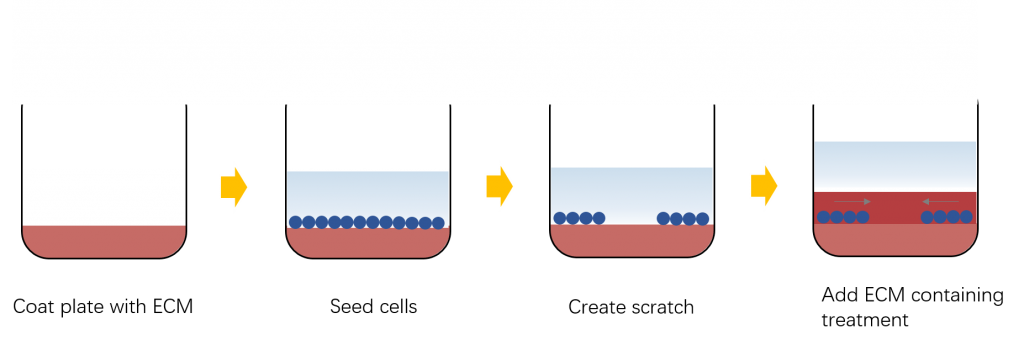
This method has some advantages, such as mimicking to some extent migration of cells in vivo, suitable to study the regulation of cell migration by cell interaction with extracellular matrix(ECM) and cell-cell interactions. It also has been combined with other techniques, such as microinjection or gene transfection, to assess the effects of expression of exogenous genes on the migration of individual cells. However, in vitro scratch assay has some disadvantages and limitations. It needs a relatively longer time to perform than some other cell migration assays and a relatively large amount of cells and chemicals will be required.
In addition, the scratch is often unevenly thick, so the migration speed of cells just prior to wound closure is often increased. The plastic surface or the ECM substrate is scraped off in an uncontrolled manner and artifacts might be induced by mechanical cell damage. To address these problems, new techniques have been developed to ensure invariable wound sizes with defined edges, such as electric cell-substrate impedance sensing (ECIS) that generates wound areas of defined size without significant variation.
CreativeProteomics offers tailored cell migration services and powerful analysis for your research.
- Matrix Degradation Analysis Service
- Boyden Chamber Assay
- Cell Scratch Assay
- Cell Exclusion Zone Assay
- Cell Fence Assay
- Microfluidic Chamber Assay
- Microcarrier Bead Assay
- Three-Dimensional (3D) Spheroid Assay
- Single Cell Migration Chip Assay
- Leukocyte Migration Agarose Technique Assay (LMAT Assay)
- Single Cell Motility Assay
- Cell Tracking Assay
- Scanning Electrochemical Microscopy (SECM)
- Fluorescent-labeled Cell Migration Assay
- Vascular Permeability Assay
- Vascular Permeability Assay In Vivo
- Vascular Permeability Assay with Microvessels
- Tube Formation Assay
References:
- Kramer N, Walzl A, Unger C, et al. In vitro cell migration and invasion assays. Mutation Research/Reviews in Mutation Research, 2013, 752(1):10-24.
- Liang C C, ParkA Y, Guan J L. In vitro scratch assay: a convenient and inexpensive method for analysis of cell migration in vitro[J]. Nature protocols, 2007, 2(2): 329.