Creative Proteomics offers protein primary structure analysis services to help customers understand the amino acid sequence of proteins and meet the requirements of the ICH Q6B guidelines.
Overview
The primary structure of proteins, at its most essential level, pertains to the linear string of amino acids that constitute the detailed composition of a protein molecule. This fundamental structure is typically epitomized utilizing a singular or trinary alphabetic representation for each constituent amino acid in the sequence. Beginning with the amino terminus (N-terminus) and concluding with the carboxyl terminus (C-terminus), this chain can exhibit immense variation across disparate proteins, with each amino acid interconnected through peptide bonds.
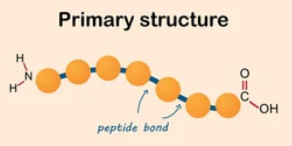 Fig 1. Protein primary structure.
Fig 1. Protein primary structure.
The primary structure represents the most rudimentary level of protein structure and carries significant implications in determining the overall protein conformation, biological functionality, and interactions. The unique organization and sequence of amino acids in a protein construct are intrinsically governed by the DNA sequence of their correlate gene. Profound comprehension of protein primary structure is pivotal within numerous domains of biological investigation; these include, but are not limited to, the engineering of proteins, the design and synthesis of novel therapeutic drugs, and elucidating the mechanisms driving various pathological conditions.
Protein Primary Structure Analysis Methods
1. Edman Degradation
This chemical technique lays the foundation for elucidating the sequence of amino acids in protein or polypeptide chains. The modus operandi involves sequenced extraction of amino acids from the termini of these chains and subsequent identification via liquid or gas chromatography.
2. Mass Spectrometry
This method is a valuable tool for analyzing protein composition and determining their primary structure. Through ionization and fragmentation of proteins, peptide fragments can be scrutinized based on their mass-to-charge ratio. By juxtaposing experimental mass spectra with established protein sequences in databases, the primary structure can be determined.
3. Protein Crystallography
Employing X-ray crystallography is extremely beneficial for deciphering the three-dimensional architecture of proteins. A detailed analysis of electron density maps, accrued from X-ray diffraction data, allows for the determination of individual atomic positions. This subsequently unmasks the overarching structure and sequential order of amino acids within the protein.
4. Tandem Mass Spectrometry (MS/MS)
Using this method, proteins are first enzymatically disassembled into individuated peptide units which are then subjected to mass spectrometry. Fragmentation of these peptides generates mass spectra that are used to infer the peptide sequence. By sequentially aggregating information from numerous peptides, the primary structure of the whole protein can be reconstituted.
Our Services
At Creative Proteomics, we offer a comprehensive range of services for protein primary structure analysis:
Our scientific service provides empirical methodologies for quantifying the molecular mass of proteins. This paramount analytic tool is routinely used to identify proteins, study protein expression and modification, investigate protein interactions, and facilitate drug development endeavors.
We proffer a service involving experimental techniques designed to identify the amino acid sequence of proteins. This essential source of data aids in detailing protein structure, function, and interaction, contributing significant insights to the realms of biology, biomedicine, and pharmaceutical development.
Our technical service deciphers the quantity, location, and nature of disulfide bonds within protein molecules. These covalent bonds, formed by dimerization of sulfhydryl groups, are critical for maintaining protein tertiary structure and modulating protein function.
Our free sulfhydryl analytical methodology detects the presence and location of free sulfhydryl groups within protein molecules. These findings can elucidate protein folding and structure, as well as their bioactivity and function in biological systems.
Covalent modification, a common form of chemical alteration involving molecules binding to specific protein amino acid residues, can dramatically affect protein structure, function, and stability.
We utilize advanced techniques to reveal variants within protein sequences, such as amino acid substitutions, insertions, or deletions. Any variation within a protein sequence can influence protein structure and stability, potentially altering biological functions and even precipitating the onset and progression of disease states.
Service Process

Identify Problem

Discussion
Testing

Reporting
Our Advantages
- Our commitment to cutting-edge technology ensures that we utilize the latest developments in mass spectrometry and other analytical platforms to deliver accurate and reliable results.
- Our team of highly skilled scientists possesses extensive knowledge in the field of protein analysis. Their expertise allows for the efficient and precise interpretation of complex datasets, providing meaningful insights into protein primary structure.
- Our customer-centric approach ensures that we maintain open lines of communication throughout the entire analysis process. We collaborate closely with our clients to fully understand their research goals and tailor our services accordingly.
Want to Learn More?
The inspection of protein primary structure necessitates imperative consideration in deducing the indispensable characteristics and operational behaviors of proteins. Here at Creative Proteomics, we extend unparalleled services in this specialized domain, employing avant-garde methodologies such as mass spectrometry to accurately decipher amino acid sequences of proteins. Our cadre of esteemed specialists, amalgamated with our pledge to perpetual innovation, situates us as a reliable collaborator in propelling the frontiers of scientific cognition. We cordially invite you to reach out to us should you possess any specific requisites concerning our protein primary structure analysis service.
Related Sections
Services
Applications
For research use only, not intended for any clinical use.

 Fig 1. Protein primary structure.
Fig 1. Protein primary structure.









