- Service Details
- Case Study
Carbohydrates, often abbreviated as CHO, are ubiquitous in nature, serving as essential biomolecules with diverse roles ranging from energy provision to structural integrity within organisms. These organic compounds consist of carbon, hydrogen, and oxygen atoms, typically arranged in a ratio of 1:2:1. Monosaccharides, the simplest form of carbohydrates, are single sugar units such as glucose and fructose. Disaccharides, like sucrose and lactose, comprise two monosaccharide units, while polysaccharides, including starch and cellulose, are complex chains of monosaccharides.
Carbohydrate analysis plays a pivotal role in understanding fundamental biological processes. By quantifying and characterizing carbohydrate molecules, researchers gain insights into energy metabolism, cellular signaling, and disease mechanisms. Creative Proteomics recognizes the importance of carbohydrate analysis in elucidating these intricate biological pathways and offers a comprehensive suite of analytical services to support scientific inquiry and innovation.
Carbohydrate Analysis in Creative Proteomics
Monosaccharide Profiling: Quantitative determination of individual monosaccharides such as glucose, mannose, and galactose in biological samples.
Disaccharide Analysis: Identification and quantification of disaccharides like sucrose, lactose, and maltose using advanced chromatographic techniques.
Polysaccharide Characterization: Structural elucidation of complex polysaccharides, including starch, cellulose, and glycogen, through enzymatic assays and mass spectrometry.
Glycoprotein Analysis: Profiling of glycoprotein glycosylation patterns to investigate their roles in cellular recognition, signaling, and immune response.
Carbohydrate Metabolism Studies: Evaluation of carbohydrate metabolism pathways and regulatory mechanisms in various biological systems, offering valuable insights into metabolic diseases and therapeutic interventions.
Technical Platforms for Carbohydrate Analysis
Chromatography Techniques:
- High-Performance Liquid Chromatography (HPLC): HPLC is widely used for separating and quantifying carbohydrates. It utilizes a liquid mobile phase to elute carbohydrates through a stationary phase, with detection typically done by UV absorbance or refractive index.
- Gas Chromatography (GC): GC is used for analyzing volatile carbohydrates and their derivatives. Carbohydrates are converted into volatile derivatives (e.g., alditol acetates) and separated based on their vaporization and partitioning properties.
- Thin-Layer Chromatography (TLC): TLC involves separating carbohydrates on a thin layer of adsorbent material (e.g., silica gel) using a solvent system. It is a rapid and cost-effective technique for qualitative analysis.
Mass Spectrometry (MS):
- Matrix-Assisted Laser Desorption/Ionization Mass Spectrometry (MALDI-MS): MALDI-MS is used for analyzing intact carbohydrates and glycoconjugates. It involves desorbing and ionizing molecules from a solid matrix using a laser, allowing for accurate mass determination.
- Electrospray Ionization Mass Spectrometry (ESI-MS): ESI-MS is suitable for analyzing carbohydrates in solution. It involves ionizing molecules in solution using an electric field, followed by mass analysis. ESI is often coupled with liquid chromatography (LC-ESI-MS) for complex carbohydrate mixtures.
Nuclear Magnetic Resonance (NMR) Spectroscopy:
- 1H-NMR and 13C-NMR: NMR spectroscopy is used to elucidate the structural features of carbohydrates in solution. It provides information on the connectivity, configuration, and conformation of carbohydrate molecules, including anomeric configuration and glycosidic linkages.
Glycomic Techniques:
- Glycan Microarrays: Glycan microarrays enable high-throughput screening of carbohydrate-protein interactions. They consist of immobilized carbohydrates on a solid support, allowing for the simultaneous analysis of binding interactions with proteins or other biomolecules.
- Glycoprotein Analysis: Techniques such as lectin affinity chromatography, glycan release, and mass spectrometry are used to analyze the glycosylation patterns of proteins. This involves identifying and quantifying glycan structures attached to proteins.
Enzymatic Assays:
- Glycosidase Assays: Glycosidase enzymes are used to selectively cleave specific glycosidic linkages in carbohydrates. By monitoring the release of products or substrate consumption, the composition and structure of carbohydrates can be determined.
- Carbohydrate-Active Enzyme (CAZyme) Profiling: CAZyme profiling involves screening carbohydrate-active enzymes for their activity towards specific carbohydrate substrates. This technique is valuable for studying carbohydrate metabolism and enzyme specificity.
List of Detectable Carbohydrate Analysis
| Category | Specific Substances |
|---|---|
| Monosaccharides | Glucose, Fructose, Galactose, Mannose, Xylose, Ribose, Arabinose |
| Disaccharides | Sucrose, Lactose, Maltose, Trehalose, Cellobiose, Isomaltose |
| Polysaccharides | Starch, Cellulose, Heteropolysaccharides, Glycosaminoglycans, Glycogen, Chitin, Xylan, Pectin, Inulin |
| Oligosaccharide | Raffinose, Stachyose |
| Glycoproteins | Immunoglobulins, Transferrin, Fetuin, Mucins, Glycophorins, Thyroglobulin |
| Others | Cyclodextrins, Oligosaccharides, Sugar alcohols (e.g., sorbitol, mannitol), Glycolipids (e.g., cerebrosides, gangliosides), Glycosides (e.g., flavonoid glycosides) |
Sample Requirements for Carbohydrate Analysis
| Sample Type | Sample Quantity | Additional Notes |
|---|---|---|
| Solid (e.g., tissues) | 2 grams | Ensure representative sampling of the biological material. |
| Cell (e.g., cultured cells, cell pellets) | ≥ 5 million cells | Maintain cell viability and integrity during sample preparation. |
| Liquid (e.g., serum, plasma, culture media) | 500 μl | Collect and store samples under appropriate conditions to prevent degradation of carbohydrates. |
Note:
For solid samples, ensure homogenization and representative sampling to account for heterogeneity.
Cell samples should be collected using standardized protocols to minimize variability.
Store liquid samples at appropriate temperatures (e.g., -80°C for long-term storage) to preserve carbohydrate stability.
Applications of Carbohydrate Analysis
Food Industry: Carbohydrate analysis is vital in determining the composition and quality of food products. It helps in assessing the levels of sugars, starches, fiber, and other carbohydrates in foods, which is essential for nutritional labeling, quality control, and product development.
Nutritional Research: Carbohydrate analysis helps in understanding the nutritional content of different foods and their impact on human health. It aids in studying the effects of carbohydrates on metabolism, glycemic index, and dietary recommendations for various populations.
Glycobiology: Carbohydrate analysis is critical for studying the structure and function of glycans (carbohydrates attached to proteins and lipids). It helps in elucidating glycan structures, glycosylation patterns, and their roles in cell signaling, immune response, and disease mechanisms.
Biofuel Production: Carbohydrate analysis is essential in the production of biofuels from renewable biomass sources such as plant materials and algae. It helps in determining the carbohydrate composition of biomass, optimizing enzymatic hydrolysis processes, and maximizing the yield of fermentable sugars for biofuel production.
Pharmaceuticals: Carbohydrate analysis is important in pharmaceutical research and development for characterizing and quality control of drugs and biologics. It helps in analyzing carbohydrate-based drugs, such as polysaccharides, glycoproteins, and glycolipids, ensuring their purity, stability, and efficacy.
Disease Diagnosis: Carbohydrate analysis plays a role in diagnosing certain diseases and disorders characterized by abnormal carbohydrate metabolism or glycosylation patterns. For example, it is used in diagnosing diabetes mellitus by measuring blood glucose levels and in detecting genetic disorders like congenital disorders of glycosylation.
Biotechnology: Carbohydrate analysis is utilized in various biotechnological applications, including the production of enzymes, bio-based materials, and therapeutic proteins. It helps in engineering carbohydrate-modifying enzymes, optimizing fermentation processes, and analyzing carbohydrate-based biomaterials.
Environmental Monitoring: Carbohydrate analysis can be applied in environmental monitoring and remediation efforts. It aids in assessing the degradation of carbohydrates in soil and water ecosystems, studying microbial carbohydrate metabolism, and evaluating the impact of pollutants on carbohydrate-containing organic matter.
Title: Characterization of Novel Serine Proteinases and Lectins from Lachesis muta rhombeata Venom
Background
Lachesis muta rhombeata venom is a complex mixture of toxins with diverse enzymatic activities, including serine proteinases and lectins. Previous studies have identified major toxin transcripts in L. muta venom glands, but comprehensive characterization of venom components and their functional properties is essential for understanding venom composition and potential therapeutic applications.
Sample
The study focused on the major fraction (S5) of L. muta venom obtained through molecular exclusion chromatography. This fraction exhibited proteolytic activity upon fibrinogen, indicating the presence of serine proteinases. Subsequent fractionation (S5C1-S5C6) allowed for the isolation and characterization of specific venom components.
Technical Methods
Transcriptomic and Proteomic Analysis: Transcriptomic studies revealed the major toxin transcripts in L. muta venom glands, while proteomic studies provided insights into the composition of Lachesis venoms.
Fractionation and Analysis: The S5 fraction of L. muta venom was subjected to reverse-phase fast protein liquid chromatography (RP-FPLC) using a C4 column to obtain subfractions (S5C1-S5C6). Each subfraction was analyzed to identify and characterize venom components.
Structural Characterization: The identified serine proteinases and lectins were structurally characterized using techniques such as SDS-PAGE, Edman degradation for N-terminal sequencing, and mass spectrometry.
Enzymatic Activity Assays: Enzymatic activities of serine proteinases were assessed using various substrates, including fibrinogen and specific peptide substrates. Inhibitor sensitivity assays were conducted to elucidate the mechanism of action.
Optimal Conditions Determination: The optimal pH and temperature for enzyme activity were determined to assess the suitability of the isolated serine proteinases for biological applications.
Results
Identification of Novel Components: The study identified novel serine proteinases (LmrSP-2, LmrSP-3, LmrSP-4, and LmrSP-5) and the first lectin (LmrLEC-1) from L. muta rhombeata venom.
Structural Characterization: Structural analysis revealed sequence homology and potential glycosylation sites in the identified serine proteinases.
Enzymatic Activities: LmrSP-4 exhibited proteolytic activity on fibrinogen and plasma kallikrein substrate, indicating its potential role in modulating blood coagulation and bradykinin-related processes.
Inhibitor Sensitivity: LmrSP-4 was sensitive to serine proteinase inhibitors but remained active in the presence of a metalloproteinase inhibitor.
Optimal Conditions: The optimal pH and temperature for LmrSP-4 activity were determined, suggesting its suitability for physiological processes.
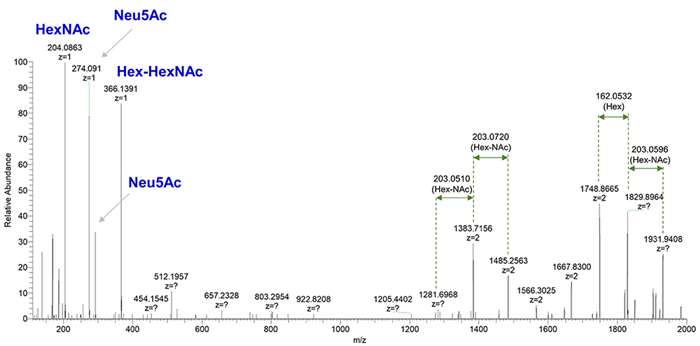 Carbohydrate content analysis by MS/MS.
Carbohydrate content analysis by MS/MS.
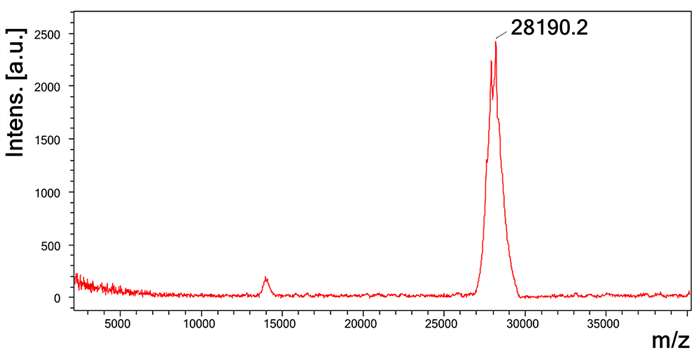 Molecular mass determination of LmrSP-4 through MALDI-TOF mass spectrometry.
Molecular mass determination of LmrSP-4 through MALDI-TOF mass spectrometry.
Reference
- Wiezel, Gisele A., et al. "Subproteome of Lachesis muta rhombeata venom and preliminary studies on LmrSP-4, a novel snake venom serine proteinase." Journal of Venomous Animals and Toxins including Tropical Diseases 25 (2019): e147018.




