- Service Details
- Case Study
Raffinose stands as a trisaccharide composed of galactose, glucose, and fructose units, intricately intertwined in its molecular structure. This oligosaccharide finds its natural abode in a plethora of organisms ranging from plants to microorganisms. Despite its prevalence, raffinose remains enigmatic, with its physiological roles spanning from a carbon reserve in seeds to a protective agent against environmental stressors.
The necessity to dissect the enigmatic functions of raffinose propels the need for meticulous analysis. Understanding its abundance, distribution, and dynamics in biological systems becomes imperative to decipher its functional significance. Creative Proteomics leads the field in raffinose analysis, offering specialized services tailored to meet diverse research needs.
Raffinose Analysis Services in Creative Proteomics
Raffinose Quantification: Our quantitative analysis service employs high-performance liquid chromatography (HPLC) coupled with mass spectrometry (MS) for precise quantification of raffinose in diverse sample types, achieving high sensitivity and accuracy.
Raffinose Structure Characterization: Utilizing nuclear magnetic resonance (NMR) spectroscopy and advanced mass spectrometry techniques, we provide detailed structural elucidation of raffinose molecules, including linkage positions and stereochemistry, essential for comprehensive understanding of its properties and functions.
Raffinose Metabolomics Profiling: Our metabolomics studies encompass targeted and untargeted approaches to investigate raffinose metabolism pathways in biological systems, utilizing cutting-edge analytical platforms such as liquid chromatography-mass spectrometry (LC-MS) and gas chromatography-mass spectrometry (GC-MS), enabling comprehensive metabolic profiling and pathway analysis.
Raffinose Detection and Quantitative Analysis: Leveraging highly sensitive and selective analytical methods such as enzyme-linked immunosorbent assay (ELISA) and liquid chromatography-tandem mass spectrometry (LC-MS/MS), we offer robust detection and accurate quantification of raffinose in complex matrices, facilitating precise measurement in various samples with excellent reproducibility and reliability.
Raffinose Stability Assessment: Through systematic stability studies employing accelerated degradation testing and storage stability evaluation under different conditions (e.g., temperature, pH), we assess the stability profile of raffinose-containing formulations, providing critical insights for formulation optimization and shelf-life determination in pharmaceutical and food industries.
Technical Platforms for Raffinose Analysis
High-Performance Liquid Chromatography (HPLC): HPLC is a widely used technique for raffinose analysis. It involves separating the components of a mixture based on their interactions with a stationary phase and a mobile phase. Raffinose can be separated and quantified using HPLC with a suitable column and mobile phase. Detection is often performed using a refractive index detector (RID) or a UV detector at specific wavelengths.
Gas Chromatography (GC): GC can also be employed for raffinose analysis, although it's less common compared to HPLC. Raffinose can be derivatized to make it volatile for GC analysis. After derivatization, the compounds are separated based on their vapor pressures as they pass through a GC column. Detection can be achieved using a flame ionization detector (FID) or a mass spectrometer (MS) for improved sensitivity and specificity.
Capillary Electrophoresis (CE): CE is a separation technique based on the differential migration of charged species in an electric field. Raffinose can be analyzed using CE by employing appropriate buffers and capillaries. Detection is typically done by UV absorption at specific wavelengths.
Enzymatic Assays: Enzymatic assays offer a specific and sensitive method for raffinose analysis. These assays typically involve the use of specific enzymes that hydrolyze raffinose into simpler sugars, such as glucose and fructose. The resulting products can then be quantified using various detection methods, such as colorimetric or fluorometric assays.
Mass Spectrometry (MS): MS can be coupled with chromatographic techniques (such as HPLC or GC) for the identification and quantification of raffinose. MS provides high sensitivity and specificity by measuring the mass-to-charge ratio of ions produced from the analyte molecules.
Nuclear Magnetic Resonance (NMR) Spectroscopy: NMR spectroscopy can be used for qualitative and quantitative analysis of raffinose in solution. By analyzing the chemical shifts and coupling constants of raffinose signals in the NMR spectrum, its concentration can be determined.
Sample Requirements for Raffinose Analysis
| Sample Type | Sample Amount |
|---|---|
| Plant Tissue | 0.5-1.0 g |
| Seeds | 50-100 seeds |
| Food Products | 5-10 g |
| Beverages | 50-100 mL |
| Blood Serum | 0.5-1.0 mL |
| Urine | 5-10 mL |
| Soil | 10-20 g |
Advantages of Raffinose Analysis
Expertise: With over two decades of experience, our team of seasoned scientists ensures the highest standards of analysis.
Customization: Each project is meticulously tailored to meet the unique requirements of our clients, ensuring precise and relevant outcomes.
Quality Assurance: Rigorous quality control measures guarantee the reliability and reproducibility of our analyses.
Timely Delivery: We adhere to strict timelines, ensuring prompt delivery of results without compromising on accuracy or quality.
Applications of Raffinose Analysis
Food Industry: Assessing raffinose levels in food products aids in quality control and nutritional analysis.
Plant Physiology: Analysis of raffinose content contributes to understanding plant stress responses and seed development.
Medical Research: Raffinose analysis is important for studying its impact on human health, particularly in relation to gastrointestinal discomfort.
Biotechnology and Metabolic Engineering: Raffinose analysis is employed to optimize metabolic pathways for the production of valuable compounds.
Animal Feed Analysis: Assessing raffinose levels in animal feed helps in formulating balanced and digestible diets.
Dietary Studies: Raffinose analysis aids in dietary studies by determining the presence and levels of this oligosaccharide in various food sources, contributing to nutritional assessments and dietary recommendations.
Crop Improvement Programs: In agricultural research, raffinose analysis assists in crop improvement programs by identifying genetic traits associated with raffinose content, leading to the development of crop varieties with improved nutritional profiles and stress tolerance.
Healthcare and Digestive Disorders: Raffinose analysis is relevant in healthcare for individuals with digestive disorders such as irritable bowel syndrome (IBS) or lactose intolerance, as it helps in identifying foods with high raffinose content that may exacerbate symptoms.
Pharmaceutical Development: In pharmaceutical research, raffinose analysis may be employed in drug formulation studies, particularly for oral medications, to assess potential interactions with raffinose-containing food ingredients and optimize drug delivery systems.
Environmental Monitoring: Raffinose analysis has potential applications in environmental monitoring, particularly in assessing the impact of agricultural practices on soil and water quality through the detection of raffinose-containing agricultural residues.
Title: Raffinose Family Oligosaccharides (RFOs) in Casuarina glauca Under Salinity Stress Conditions
Background
Raffinose family oligosaccharides (RFOs) are vital water-soluble carbohydrates found abundantly in plants, serving various biological functions such as osmoprotection, carbon source and transport, and signaling against pathogen attacks. Understanding their biosynthesis and behavior under stress conditions is crucial for unraveling plant adaptation mechanisms.
Sample
Casuarina glauca, a nodulated plant species, was subjected to salinity stress by gradually increasing NaCl concentrations in the growth medium. Plant tissues, including nodules and branchlets, were harvested at different stress levels for analysis.
Technical Methods
Extraction: RFOs were extracted from C. glauca tissues using a methanol-based protocol followed by chloroform and water phase separation.
Mass Spectrometry: RFOs were analyzed using PGC-ESI-QIT-MSn method, involving chromatographic separation on a PGC Hypercarb column coupled with quadrupole ion trap mass spectrometry in the positive ion mode. CID MSn experiments were performed to characterize RFOs' fragmentation patterns.
LC-MS Method Transfer: The LC-MS method for RFO analysis was transferred from a larger column and mass spectrometer to a smaller column and instrument, ensuring compatibility and efficient analysis.
Quantification: Quantification of RFOs was achieved through calibration curves and peak area integration, allowing for accurate determination of RFO levels in plant tissues under different stress conditions.
Validation: The developed method was validated for linearity, selectivity, and reproducibility, ensuring its reliability for RFO analysis.
Results
The study successfully identified and quantified raffinose in C. glauca tissues under varying salinity stress levels. However, stachyose and verbascose were not detected. The method demonstrated good repeatability and sensitivity, providing valuable insights into the response of RFOs to salinity stress in C. glauca plants.
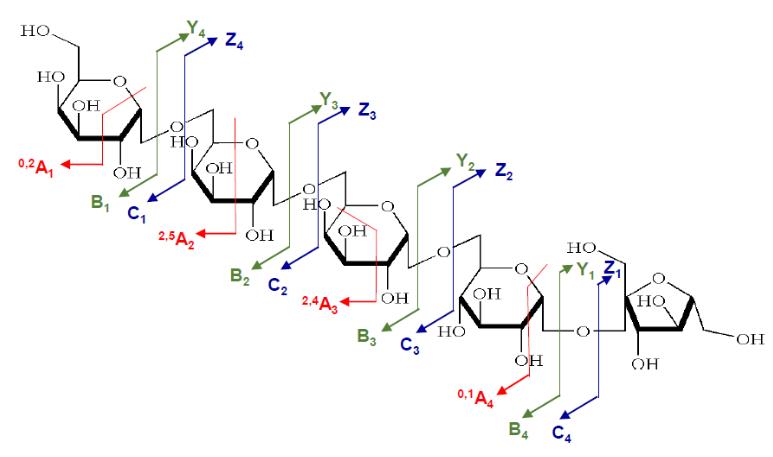 Positive ion CID spectra of raffinose obtained by PGC-ESI-QIT-MSn showing nomenclature of Domon and Costello
Positive ion CID spectra of raffinose obtained by PGC-ESI-QIT-MSn showing nomenclature of Domon and Costello
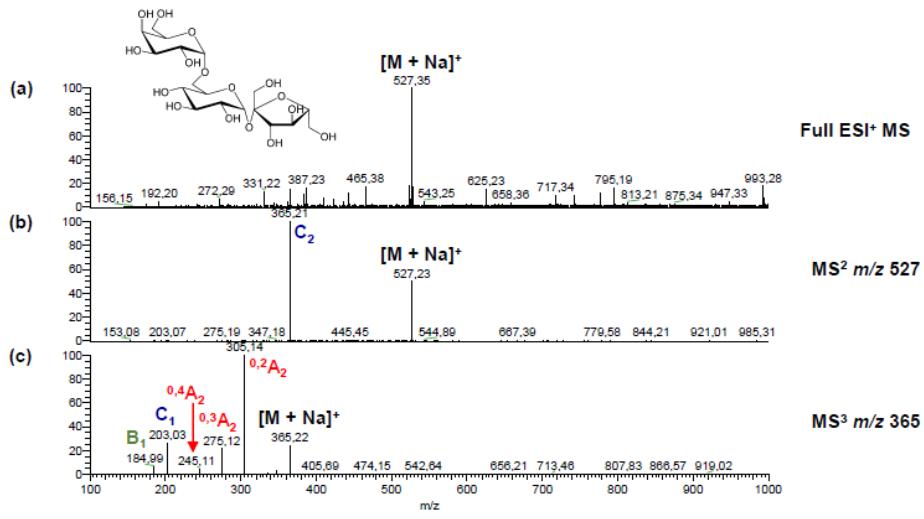 Positive ion CID spectra of stachyose obtained by PGC-ESI-QIT-MSn showing nomenclature of Domon and Costello
Positive ion CID spectra of stachyose obtained by PGC-ESI-QIT-MSn showing nomenclature of Domon and Costello
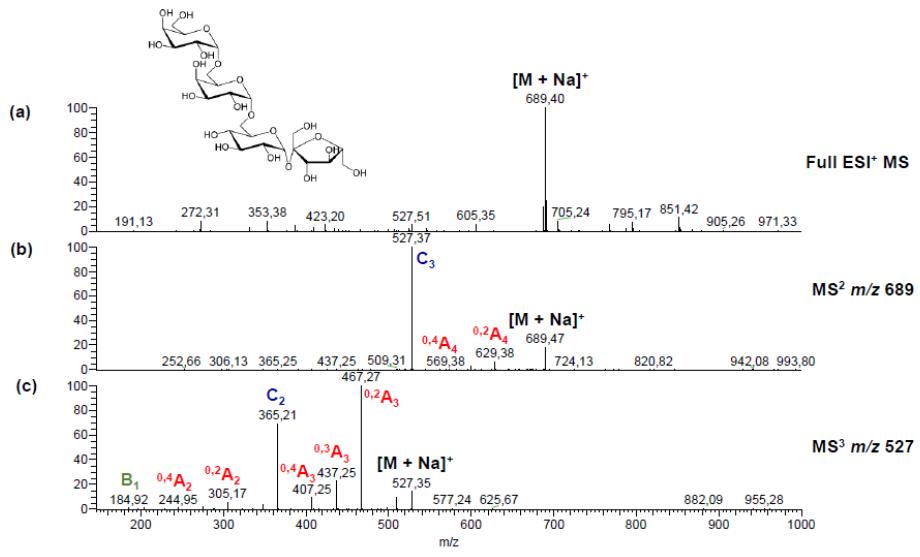 Positive ion CID spectra of verbascose obtained by PGC-ESI-QIT-MSn showing nomenclature of Domon and Costello
Positive ion CID spectra of verbascose obtained by PGC-ESI-QIT-MSn showing nomenclature of Domon and Costello
Reference
- Jorge, Tiago F., et al. "Quantification and structural characterization of raffinose family oligosaccharides in Casuarina glauca plant tissues by porous graphitic carbon electrospray quadrupole ion trap mass spectrometry." International Journal of Mass Spectrometry 413 (2017): 127-134.




