- Service Details
- Case Study
What is Glycosylphosphatidylinositol?
Glycosylphosphatidylinositol (GPI) is a complex molecule composed of a phospholipid anchor linked to a glycan moiety, which in turn is attached to a protein. This unique structure enables GPI to serve as a membrane anchor for proteins, facilitating their association with the cell membrane. The glycan portion of GPI can vary greatly in composition and complexity, contributing to the diversity of GPI-anchored proteins (GPI-APs) found in cells.
GPI-anchored proteins are involved in a myriad of cellular processes, including cell adhesion, signal transduction, and immune response modulation. They play crucial roles in neuronal development, host-pathogen interactions, and the regulation of enzymatic activities. Given their significance, elucidating the structure and function of GPI molecules is essential for understanding various physiological and pathological processes.
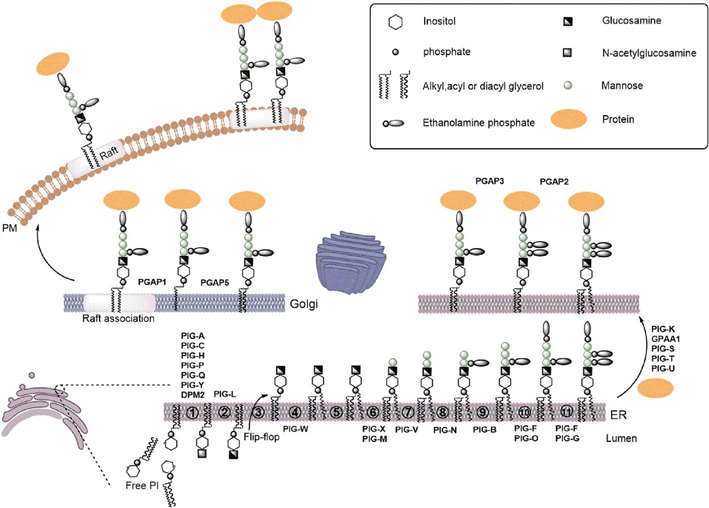 A scheme for the overall GPI-APs biosynthetic pathway, structural remodeling and transport (Wu et al., 2020).
A scheme for the overall GPI-APs biosynthetic pathway, structural remodeling and transport (Wu et al., 2020).
Creative Proteomics offers a comprehensive suite of services for glycosylphosphatidylinositol analysis. We deliver accurate and reliable results tailored to meet the diverse needs of researchers worldwide.
Glycosylphosphatidylinositol Analysis Services by Creative Proteomics
Our expertise in glycomics allows us to provide high-quality data and insights into GPI structures, compositions, and functions. Our GPI analysis services include:
- GPI Structure Determination: We employ advanced analytical techniques such as mass spectrometry (MS) and nuclear magnetic resonance (NMR) spectroscopy to elucidate the detailed structure of GPI molecules, including lipid and carbohydrate moieties.
- GPI Composition Analysis: Through chromatographic methods coupled with MS detection, we can accurately determine the composition of GPIs, including the identification and quantification of lipid and sugar components.
- GPI Biosynthesis Studies: Our services cover the investigation of GPI biosynthetic pathways and the identification of enzymes involved in GPI synthesis, providing valuable insights into GPI-related biological processes.
- GPI Modification Analysis: We specialize in the characterization of GPI modifications, such as fatty acid remodeling and carbohydrate substitutions, using cutting-edge analytical techniques.
- GPI-anchored Protein Analysis: We offer comprehensive analysis of GPI-anchored proteins, including identification, characterization, and quantification of GPI-APs in complex biological samples.
- GPI Function and Interaction Studies: Our services include functional analysis of GPI molecules, including their roles in cell signaling, membrane trafficking, and immune response, as well as studies on GPI-protein interactions.
- Custom GPI Analysis: We provide customized GPI analysis services tailored to meet specific research requirements, ensuring high-quality data and actionable insights.
Technical Platforms for Glycosylphosphatidylinositol Analysis
Mass Spectrometry (MS):
- High-resolution Mass Spectrometry (HRMS): Utilized for accurate mass determination of GPI molecules, enabling precise identification and structural characterization.
- Tandem Mass Spectrometry (MS/MS or LC-MS/MS): Enables fragmentation analysis for sequencing GPIs and elucidating their structural features.
- MALDI-TOF Mass Spectrometry: Applied for rapid profiling and fingerprinting of GPI molecules in complex samples.
Nuclear Magnetic Resonance (NMR) Spectroscopy:
- High-Field NMR Spectrometers (e.g., 600 MHz, 800 MHz): Employed for structural elucidation of GPIs, providing insights into their three-dimensional arrangement and conformational dynamics.
Liquid Chromatography (LC) Systems:
- High-Performance Liquid Chromatography (HPLC): Coupled with MS detection for chromatographic separation and quantification of GPI components.
- Ultra-High-Performance Liquid Chromatography (UHPLC): Enables fast and efficient separation of GPIs, enhancing analytical throughput and sensitivity.
Glycan Analysis Platforms:
- Glycan Profiling Systems: Used for the characterization of GPI-linked carbohydrate structures, including monosaccharide composition analysis and linkage determination.
- Glycan Microarray Technology: Applied for high-throughput screening of GPI interactions with carbohydrate-binding proteins and receptors.
Enzyme Assays and Biochemical Techniques:
- Enzyme Activity Assays: Employed for studying GPI biosynthetic pathways and identifying enzymatic activities involved in GPI modification processes.
- Western Blotting and Immunoprecipitation: Utilized for GPI-anchored protein analysis, including protein identification and quantification.
Customized Bioinformatics Tools:
- Data Analysis Software: Custom-built algorithms and bioinformatics pipelines for processing MS and NMR data, facilitating GPI structural annotation and interpretation.
Sample Requirements for Glycosylphosphatidylinositol Analysis
| Sample Type | Sample Quantity | Sample Preparation |
|---|---|---|
| Cell Culture Supernatants | 1-5 mL | Centrifugation to remove cells and debris, followed by filtration to remove particulates. Freeze immediately after collection. |
| Tissue Homogenates | 20-100 mg | Homogenization in appropriate buffer solution (e.g., PBS) using a tissue homogenizer. Centrifugation to remove debris. Aliquot and freeze. |
| Serum/Plasma | 100-500 µL | Centrifugation to remove cells and debris. Aliquot supernatant and freeze immediately. Avoid repeated freeze-thaw cycles. |
| Urine | 1-10 mL | Centrifugation to remove cells and debris. Aliquot supernatant and freeze immediately. Avoid repeated freeze-thaw cycles. |
| Cell Pellets | 1-10 million cells | Resuspend cells in appropriate buffer solution (e.g., PBS). Sonication or freeze-thaw cycles may be required for cell lysis. Centrifuge to pellet debris. Extract GPIs using suitable extraction methods. |
| Biological Fluids | 100-500 µL | Centrifugation to remove cells and debris. Aliquot supernatant and freeze immediately. Avoid repeated freeze-thaw cycles. |
| Tissue Sections | 10-50 mg | Homogenization in appropriate buffer solution (e.g., PBS) using a tissue homogenizer. Centrifugation to remove debris. Aliquot and freeze. |
| Cultured Cells | 1-10 million cells | Harvest cells using appropriate methods (e.g., trypsinization). Wash cells with PBS. Extract GPIs using suitable extraction methods. |
Applications of Glycosylphosphatidylinositol Analysis
Biomedical Research:
- GPI analysis enables researchers to elucidate the structure and function of GPI-anchored proteins (GPI-APs), shedding light on their roles in cellular processes such as signal transduction, immune response modulation, and cell adhesion.
- Studies on GPI modification patterns and interactions provide insights into their involvement in physiological and pathological conditions, facilitating the discovery of novel therapeutic targets and biomarkers.
Disease Mechanisms and Biomarker Discovery:
- Understanding the role of GPI molecules in disease pathogenesis is critical for developing targeted therapies and diagnostic tools. GPI analysis helps researchers investigate the association between GPI modifications and various diseases, including cancer, neurodegenerative disorders, and infectious diseases.
- Identification of GPI-APs as potential biomarkers offers opportunities for early disease detection, prognosis assessment, and monitoring of therapeutic responses.
Drug Development and Target Identification:
- GPI analysis assists in identifying and characterizing GPI-APs involved in disease pathways, providing valuable targets for drug development and therapeutic intervention.
- By elucidating the structural and functional properties of GPI molecules, researchers can design and optimize therapeutic agents targeting GPI-anchored proteins, offering new avenues for personalized medicine and precision therapies.
Neuroscience and Neurological Disorders:
- GPI-anchored proteins play crucial roles in neuronal development, synaptic transmission, and synaptic plasticity. GPI analysis contributes to understanding the molecular mechanisms underlying neurological disorders such as Alzheimer's disease, Parkinson's disease, and schizophrenia.
- Investigation of GPI modifications in neural cells and tissues offers insights into disease mechanisms and potential therapeutic strategies for neurodegenerative diseases and psychiatric disorders.
Infectious Diseases and Host-Pathogen Interactions:
- GPI-anchored proteins are involved in host-pathogen interactions, serving as receptors for pathogens or modulators of immune responses. GPI analysis helps elucidate the molecular interactions between GPI molecules and pathogens, contributing to the development of vaccines and antimicrobial therapies.
- Understanding GPI modifications in infectious agents and host cells facilitates the identification of novel drug targets and the development of strategies to combat infectious diseases.
Title: Novel method for the identification and analysis of GPI-Anchored Proteins by MS/MS analysis and database search
Background
Glycosylphosphatidylinositol (GPI) anchors are post-translational modifications that localize proteins to the outer leaflet of the plasma membrane. The common GPI core structure is conserved among eukaryotic species. GPI-Anchored Proteins (GPI-APs) associate preferentially with lipid rafts and are involved in a wide range of biological functions. Understanding the mechanics of GPI-anchor remodeling and the function of GPI-APs requires accurate identification of GPI anchors and many ω-sites of GPI-APs.
Sample
The study focused on potential GPI-Anchored Proteins from ovarian clear cell adenocarcinoma OVISE cells, cultured in RPMI 1690 medium.
Technical Methods
The method developed here incorporates several stages. First, DRMs (detergent-resistant membranes) which contain GPI-APs were isolated from cancer cell lysates subjected to sucrose gradient centrifugation. Next, GPI-APs were purified using two-phase separation with Triton X-114. The lipid moieties of the GPI-APs were removed by Phosphatidylinositol-specific phospholipase C (PI-PLC) treatment and the lipid-free proteins were then recovered in the aqueous phase. The proteins were concentrated through TCA/acetone precipitation, separated by SDS-PAGE, and visualized by SYPRO Ruby staining. To analyze the C-terminal GPI-anchored peptide sequence, the phosphodiester bond in the GPI anchor was cleaved by treatment with Hydrogen Fluoride (HF). The HF-treated GPI-anchored peptides, which contain only EtN moiety of the GPI anchor, were then identified and analyzed by MS/MS biomolecular analysis and database search for validation. Proteins were identified as GPI-APs if a minimum of two peptides matched with a Mascot score more significant than 50.
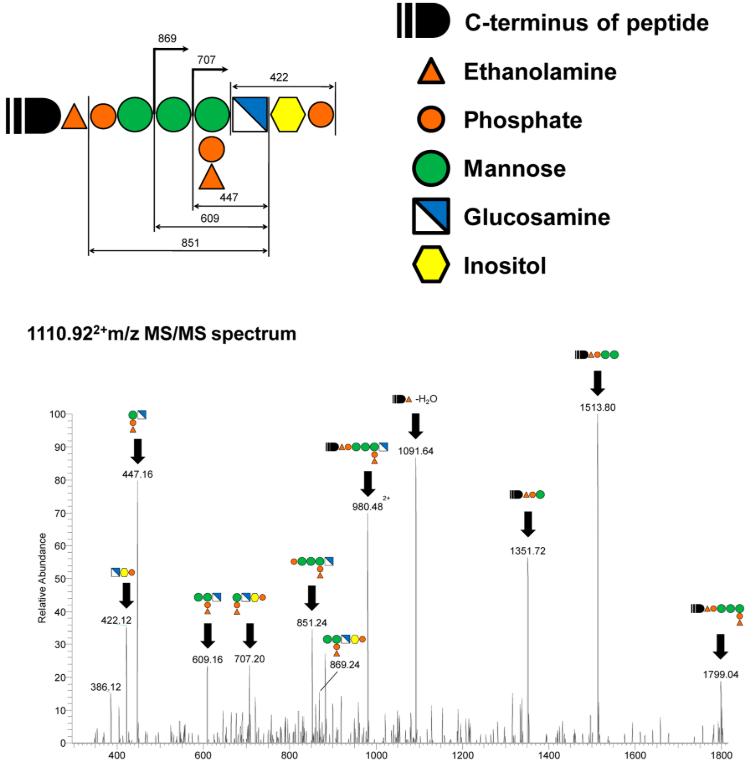 Structural analysis of the GPI-anchored peptide
Structural analysis of the GPI-anchored peptide
Results
Through these methods, 25 GPI-anchored peptide sequences corresponding to 10 GPI-APs were identified, as well as 12 ω-sites that provided new insights into the specificity of GPI-APs. Notably, the study found that SMPDL3B is a novel GPI-AP and that protein BST2 has more than one ω-site. The results demonstrated that the method developed in this study is effective for the efficient analysis of GPI-APs.
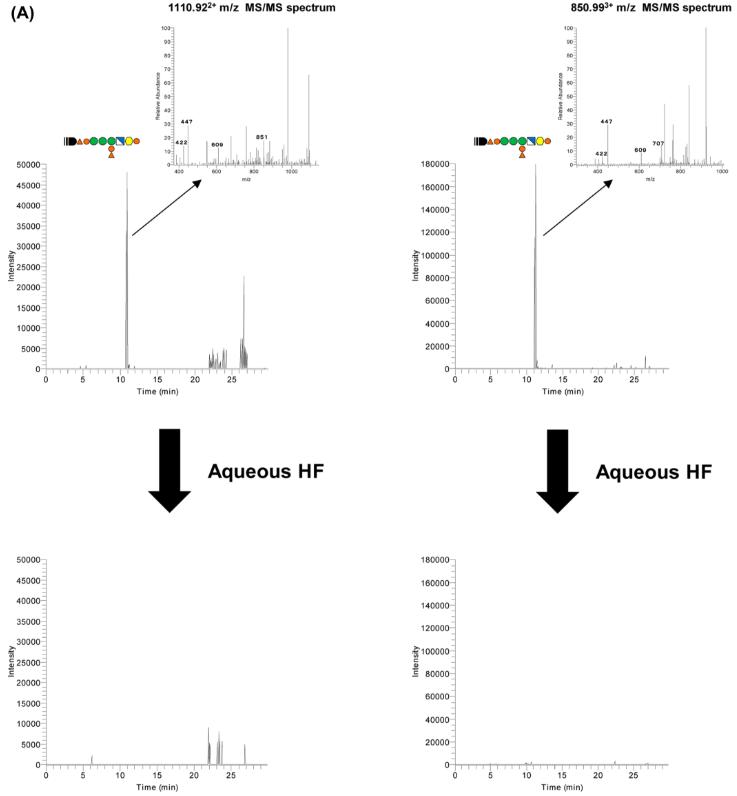 Analysis by MS/MS of GPI-anchored peptides
Analysis by MS/MS of GPI-anchored peptides
Reference
- Masuishi, Yusuke, et al. "Mass spectrometric identification of glycosylphosphatidylinositol-anchored peptides." Journal of proteome research 12.10 (2013): 4617-4626.




