- Service Details
- Case Study
N-Glycan profiling is a crucial aspect of understanding the glycosylation patterns in proteins, particularly in the context of biotherapeutics. Glycosylation, a common post-translational modification, plays a vital role in determining the stability, efficacy, and bioavailability of proteins. It involves the attachment of complex sugar molecules (glycans) to specific amino acid residues in proteins. This modification influences various biological processes, such as ligand-receptor interactions, immune response, and protein secretion.
The necessity for N-glycan profiling arises from its significant impact on disease, therapeutic development, and biopharmaceutical production. Alterations in glycosylation have been observed in conditions like cancer, Alzheimer's disease, and inflammatory disorders. In the biopharmaceutical industry, understanding and controlling glycosylation are critical for ensuring the efficacy, stability, and safety of therapeutic proteins.
Technology Platform for N-Glycan Profiling Analysis
Enzymatic and Chemical Release:
Our N-Glycan Profiling begins with the enzymatic release of glycans using cutting-edge enzymes. The use of the novel enzyme, Rapid™ PNGase F, allows for swift and unbiased deglycosylation of antibodies and fusion proteins in minutes. This not only accelerates the analysis process but also ensures the removal of all N-glycan sites without bias, allowing the measurement of all species with accuracy.
In addition to enzymatic release, we employ a chemical release method using hydrazine. This well-established and validated technique ensures the complete release of N-linked oligosaccharides, providing a robust alternative for glycan analysis.
Fluorescent Labeling for Sensitive Detection:
To enhance the sensitivity of our analysis, we employ fluorescent labeling of released N-glycans. This step is crucial for sensitive detection and quantitation during liquid chromatography coupled with fluorescence detection (LC-FLD). The choice of fluorescent tags, including 2-aminobenzamide (2AB), 2-aminobenzoic acid (2AA), procainamide (PCA), and 8-Aminopyrene-1,3,6-trisulfonic acid (APTS), ensures versatility in detection methods, including mass spectrometry and capillary electrophoresis.
High-Resolution Liquid Chromatography and Mass Spectrometry:
Our analytical approach involves high-resolution liquid chromatography (LC) coupled with tandem high-resolution mass spectrometry (HRMS). This powerful combination allows for superior resolution, enabling the easy recognition of slight changes in N-glycosylation profiles. The use of HRMS ensures accurate identification of N-glycan structures based on retention time and exact mass.
Exoglycosidase Array Analysis for Structural Detail:
For a detailed structural analysis of released N-glycans, we employ an exoglycosidase array analysis using the N-Glycan Sequencing Kit E0577s. This approach allows us to differentiate isobaric N-glycans and provides additional information about bisecting GlcNAc, antennary GlcNAc, or unwanted GalNAc.
Comprehensive Data Integration:
Our technology platform is designed to integrate data from multiple sources, combining fluorescence and mass spectrometry methods. This multi-method approach ensures a comprehensive analysis, providing you with a holistic view of the N-Glycan structures.
Advantages of Choosing Creative Proteomics's N-Glycan Profiling
- Efficient Deglycosylation: Swift and unbiased deglycosylation facilitated by the novel enzyme Rapid™ PNGase F.
- Precision Technology: Utilization of high-resolution LC and MS for achieving precise and detailed N-glycan profiling results.
- Holistic Analysis: Comprehensive multi-method approach combining fluorescence and mass spectrometry methods.
- Quality Assurance: Implementation of running standards and provision of detailed Certificates of Analysis for stringent quality control.
Applications of N-Glycan Profiling
Biopharmaceutical Development: Assessing N-glycan impact on stability, efficacy, and immunogenicity, ensuring consistent product quality during process development.
Disease Research and Diagnosis: Investigating disease-associated N-glycan alterations for diagnostic and prognostic purposes.
Cell Line Studies: Analyzing N-glycan release, recovery, and profiles in various cell lines to understand glycan-related mechanisms.
Biopharmaceutical Quality Control: Establishing connections between glycan structures and biopharmaceutical properties for quality assurance.
Monoclonal Antibody Optimization: Enhancing antibody function by analyzing glycan structures and optimizing for improved bioactivity.
Regulatory Compliance: Meeting regulatory requirements by including N-glycan analysis in biotherapeutic release criteria.
Continuous Manufacturing Improvement: Monitoring and adjusting glycosylation profiles during continuous manufacturing for consistent analysis.
Comparative Glycan Studies: Conducting qualitative and quantitative analyses of different N-glycan release and analysis approaches.
Sample Requirements for N-Glycan Profiling
| Sample Type | Sample Size Recommendation |
|---|---|
| Antibodies | 10 μg - 30 μg |
| Biopharmaceutical Products | 20 μg - 50 μg |
| Cell Lines (e.g., HT29) | 2 x 106 cells |
| Cerebrospinal Fluid (CSF) | 10 μL - 30 μL |
| Glycoproteins (e.g., IgG) | 15 μg - 50 μg |
| Plasma | 5 μL - 10 μL |
| Saliva | 50 μL - 100 μL |
| Serum | 5 μL - 10 μL |
| Tissue (e.g., Cancer Tumor) | 10 mg - 20 mg |
| Urine | 20 μL - 50 μL |
Case. Comprehensive N-Glycan Analysis Reveals Distinct Glycosylation Patterns in Lung Adenocarcinoma Patients
Background:
The study aimed to explore the applicability of a sample preparative procedure for N-glycan analysis in clinical settings, focusing on fresh and defrosted tissues of lung adenocarcinoma (LAC) patients. The objective was to correlate N-glycan profiles with different stages of LAC, identifying potential biomarkers for early detection and understanding glycosylation alterations in cancer.
Samples:
Tissue specimens were obtained from 24 LAC patients at various stages (IA to IV). The samples included tumors and adjacent noncancerous tissues. Additionally, bronchoscopy biopsies were collected from eight patients, enabling analysis of small-sized tissue fragments.
Technical Method:
Sample Preparative Procedure: The study employed a sample preparative procedure previously established for investigating N-glycans in lyophilized cancer cell lines. The focus was on analyzing both fresh and defrosted tissues to ensure clinical relevance. Sialylated glycans were confirmed through permethylation, and the primary profiling was conducted on desialylated N-glycans.
Comparative N-Glycan Analysis: Mass spectrometry (MS) was used for the identification and quantification of N-glycan structures in cancerous and adjacent control tissues. The analysis extended beyond known glycans, exploring new and complex structures in the mass range over 2600 Da. The study assessed core fucosylation and the expression of glycosyltransferases, such as FUT3 and GnT-V.
Data Analysis: Comparative analysis was conducted between glycan profiles of tumors and noncancerous tissues from the same patient. A reference control dataset, averaged from 16 patients, was utilized for a broader comparison. The study investigated changes in glycan profiles at different stages of LAC.
Clinical Applicability: The study tested the applicability of N-glycan data for the evaluation of biopsies with limited sizes and lacking control tissues. Despite tissue limitations in bronchoscopy biopsies, the accumulated data from glycan analyses of tumor and control tissues facilitated the determination of the nature of lung lesions in patients.
Identification of Potential Biomarkers: Newly identified glycan structures, not reported in previous studies, were explored as potential biomarkers for early disease stages and metastatic processes.
Results:
Glycan Alterations: High-mannose glycans increased in tumors, while Man5GlcNAc2 exhibited a decrease. Core fucosylated glycans declined, with new and complex structures dominating in tumors.
Enzyme Expression: Contradictory findings were observed in core fucosylation and FUT8 expression. Increased arm fucosylation indicated upregulated FUT3 activity, and the presence of non-galactosylated GlcNAc branches suggested enhanced GnT-V expression.
Differentiation Between Tissues: Clear differences were identified between cancerous and healthy tissues, becoming more prominent in advanced LAC stages.
Potential Biomarkers: Newly identified glycan structures in tumors may serve as promising biomarkers, offering insights into early malignancy and metastatic processes.
Clinical Applicability: The study demonstrated the effectiveness of the N-glycan analysis approach in clinical testing, allowing rapid discrimination between cancerous and healthy lung tissues, even in limited-size biopsy specimens.
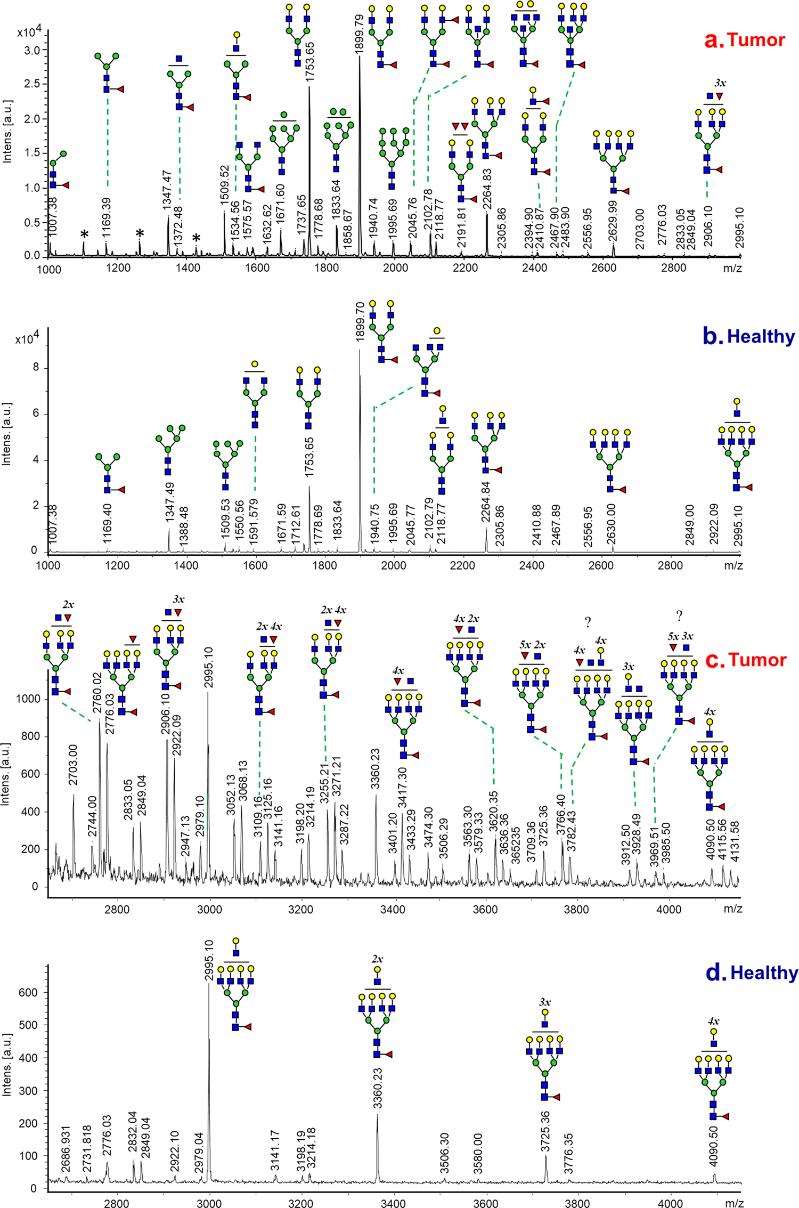 MALDI-MS spectra of N-glycans recorded from cancerous and control lung tissues of patient with LAC.
MALDI-MS spectra of N-glycans recorded from cancerous and control lung tissues of patient with LAC.
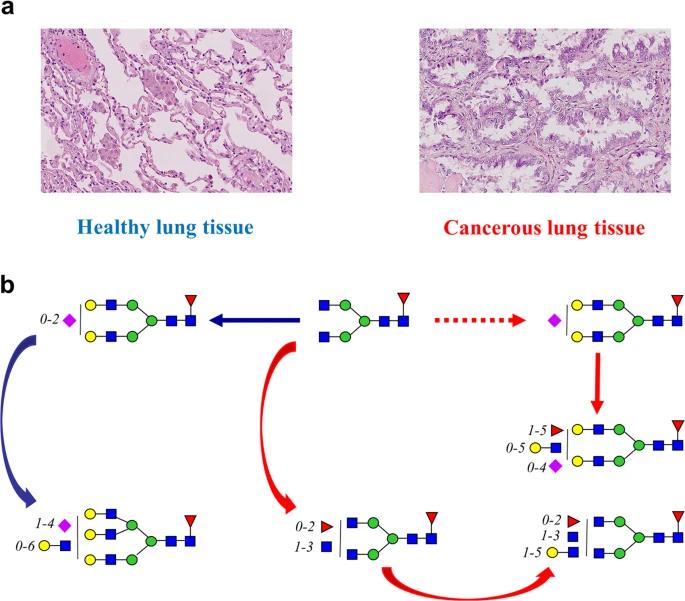 Comparison of healthy and cancerous lung tissues.
Comparison of healthy and cancerous lung tissues.
Reference
- Lattová, Erika, et al. "N-Glycan profiling of lung adenocarcinoma in patients at different stages of disease." Modern Pathology 33.6 (2020): 1146-1156.




