- Service Details
- Case Study
What is Neutral and Amino Sugar?
Neutral sugars and amino sugars are two classes of monosaccharides, which are simple sugar molecules that serve as fundamental building blocks of carbohydrates.
Neutral sugars refer to monosaccharides that do not exhibit acidic or basic properties. They are characterized by a lack of charge and include common sugars such as glucose, fructose, and galactose. These sugars play vital roles in various biological processes, serving as a primary source of energy for cells and organisms. Neutral sugars are often derived from plant sources, and they contribute significantly to the composition of soil organic matter. Cellulose, a major component of plant cell walls, primarily consists of glucose monomers, while other polysaccharides like hemicellulose and starch also contribute to the pool of neutral sugars in soils.
Amino sugars, on the other hand, are monosaccharides that contain an amino group in addition to the sugar molecule. They are crucial structural components found in the cell walls of archaea, bacteria, and fungi. Examples of amino sugars include glucosamine, which is a common component of chitin in fungal cell walls, and muramic acid, which is specific to the murein layers of bacterial peptidoglycan. Amino sugars are significant contributors to soil organic matter, particularly when derived from microbial necromass. Glucosamine, in particular, is a prevalent amino sugar in soils, constituting a substantial portion of total amino sugars.
Why Need Neutral and Amino Sugar Analysis?
Understanding the composition and dynamics of neutral and amino sugars holds significant importance across diverse fields, including biomedicine, pharmaceuticals, and beyond. In the realm of biomedicine, these sugars are integral to cellular processes, serving as critical components in glycoproteins and glycolipids. The structural aspects of amino sugars, for instance, are essential for the formation of (pseudo)peptidoglycan and chitin in archaea, bacteria, and fungi. In this context, the analysis of neutral and amino sugars becomes indispensable for comprehending the intricate molecular landscapes within living organisms.
The pharmaceutical industry, characterized by its stringent quality control measures, also relies on neutral and amino sugar analysis. Amino sugars, acting as biomarkers, enable the assessment of microbial biomass, a crucial parameter in the production of pharmaceuticals. The identification and quantification of these sugars contribute to the verification of product authenticity and purity, ensuring that pharmaceuticals derived from plant sources meet regulatory standards.
Beyond the biomedical and pharmaceutical sectors, neutral and amino sugar analysis finds applications in various fields. In environmental studies, it aids in assessing microbial activity in soils and understanding the impact of land management practices. The agriculture industry benefits from insights into soil health, guiding sustainable practices. In food industries, the analysis ensures the quality of plant-derived products, safeguarding consumers.
Technology Platform for Neutral and Amino Sugar Analysis
Creative Proteomics provides the technology platform for neutral and amino sugar analysis.
- Derivatization Using 1-Phenyl-3-Methyl-5-Pyrazolone (PMP)
Derivatization is a crucial step in the analysis of neutral and amino sugars, enhancing their detectability. The use of 1-phenyl-3-methyl-5-pyrazolone (PMP) is a common derivatization method. PMP rapidly reacts with the aldehyde moiety of reducing sugars under basic conditions, forming bis-PMP derivatives. This reaction results in stable derivatives with no stereoisomers, facilitating their separation and analysis.
- Ultra-High Performance Separation Techniques:
To address the structural complexity of sugar-containing compounds, ultra-high performance separation techniques are employed. Reversed-phase HPLC with high-resolution mass spectrometry (RP-HPLC-HRMS) has proven effective in the analysis of compound mixtures with isobaric structures. This technology allows for the concurrent separation and mass spectrometric detection of a wide range of neutral and amino sugars.
- UPLC-Orbitrap MS System
This system, which combines ultra-performance liquid chromatography (UPLC) with Orbitrap high-resolution mass spectrometry, offers high sensitivity and accuracy. Chromatographic separation is achieved using a C18 column, and the mass spectrometer operates in full-mass scan mode, covering a wide mass range for comprehensive sugar detection.
- Nuclear Magnetic Resonance (NMR)
NMR provides detailed structural information about neutral and amino sugars, elucidating molecular structures and offering insights into configuration, conformation, and atom connectivity.
- Gas Chromatography-Mass Spectrometry (GC-MS)
GC-MS separates and identifies sugar compounds based on volatility. It is effective for analyzing volatile and semi-volatile compounds, providing high sensitivity and selectivity.
- Quantification Using Isotope Labeling and Correction:
Quantification of sugars is often achieved through isotope labeling, where stable isotopes are introduced into the samples. Isotopic dilution series are prepared, and the atom % 13C is calculated considering multiple isotopologues. Correction for natural isotope occurrence from derivatization reagents is crucial for accurate quantification.
Applications of Neutral and Amino Sugar Analysis
Soil Organic Matter Studies: Evaluate soil organic matter composition for insights into microbial contributions and soil fertility.
Microbial Biomass Characterization: Identify and quantify microbial necromass components for studying microbial community structure.
Plant Physiology and Metabolism: Analyze sugar profiles in plant tissues to understand cell wall composition and metabolic pathways.
Pharmaceutical Quality Control: Ensure drug formulation quality by verifying the composition of pharmaceutical compounds.
Food Industry and Nutrition Studies: Characterize carbohydrate content in food products for nutritional studies and quality control.
Environmental Monitoring and Pollution Studies: Monitor sugars in water samples to assess microbial activity and pollution levels.
Biomedical Research: Contribute to cellular structure studies, including cell walls and glycans, in biomedical research.
Agricultural and Crop Sciences: Aid in soil management and crop nutrition studies for sustainable farming practices.
Biotechnology and Biofuel Production: Guide biofuel production and biotechnological applications by understanding sugar composition in biomass.
Environmental Impact Assessments: Use in environmental impact assessments to evaluate human activities' effects on ecosystems.
Sample Requirements for Neutral and Amino Sugar Analysis
| Sample Type | Sample Size Recommendation | Additional Considerations |
|---|---|---|
| Biological Fluids (e.g., Blood, Urine) | 0.5-1 milliliter | Handle biological fluids with care to prevent contamination. Store at appropriate temperatures before analysis. |
| Soil (Environmental Studies) | 5-10 grams | Collect samples from various depths for comprehensive analysis. |
| Plant Tissues (e.g., Leaves, Stems) | 20-50 milligrams (dry weight) | Homogenize tissues for uniform representation. Consider specific plant parts based on research objectives. |
| Food Products (e.g., Fruits, Vegetables) | 5-20 grams | Homogenize food samples to obtain representative portions. Consider the diversity of food components. |
| Microbial Biomass (e.g., Peptidoglycan) | 1-5 milligrams (dry weight) | Lyophilize microbial biomass for accurate dry weight measurement. |
| Pharmaceutical Compounds | 1-10 milligrams | Ensure the representative sampling of pharmaceutical products for accurate analysis. |
| Environmental Water Samples | 50-100 milliliters | Collect samples using clean containers and avoid contamination. Filter water samples to remove particulate matter if needed. |
Case. Enhanced Anti-Proliferative Activity of Sugar Beet Pectin Extract on Colon Cancer Cells: Role of Neutral Sugar Side-Chains
Background:
The study aims to explore the structural features of modified pectin extracts, particularly from sugar beet, and their relationship with anti-cancer activity. Previous research suggested a potential link between neutral sugar-rich regions in pectin and enhanced bioactivity against cancer cells.
Samples:
Various pectin extracts were obtained from sugar beet and citrus peel, each exhibiting distinct structural characteristics. Sugar beet pectin extracts (SBC, SBH, SBA, SBO, SBOPG) were analyzed for their neutral sugar and GalA composition, degree of esterification (DE), degree of acetylation (DAc), and molecular weight (MW). These samples were compared with a non-bioactive citrus peel pectin extract (CP).
Technical Method:
Pectin extracts were characterized for their structural features, including the composition of neutral sugars (Gal, Ara, Rha), GalA, and MW.
Effects on colon cancer cell proliferation (HT29 and DLD1) were assessed after treatment with different pectin extracts at varying concentrations.
Enzymatic treatments were applied to modify sugar beet pectin (SBA), targeting galactan and arabinan side-chains, and the resulting samples were analyzed using NMR spectroscopy.
The role of neutral sugar side-chains in the enhanced bioactivity of sugar beet pectin was investigated by evaluating the effects of enzyme-digested pectins on cell proliferation, apoptosis, and cell cycle regulation.
Results:
Sugar beet pectin extracts, particularly SBA, demonstrated significant anti-proliferative effects on HT29 colon cancer cells.
Enzymatic removal of galactan side-chains increased the anti-proliferative effect of SBA, while removal of arabinan side-chains had a counteractive effect.
The enhanced bioactivity of SBA was associated with increased apoptosis in HT29 cells, without significant cell cycle arrest.
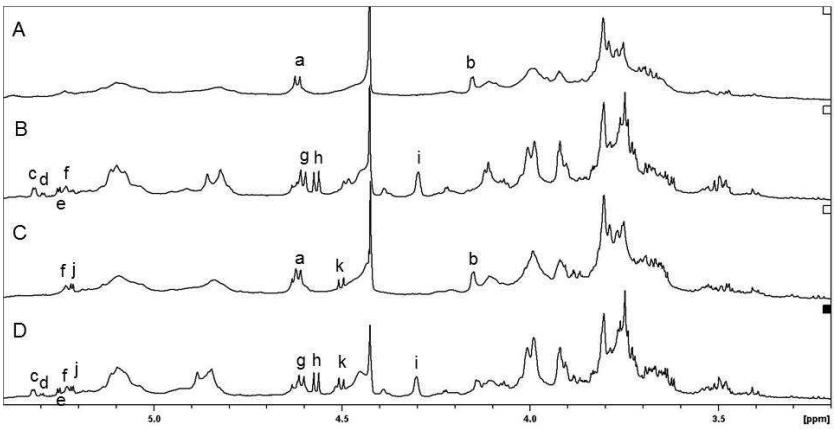 600MHz 1H NMR spectra of enzyme-digested SBAs in D2O at 3340
600MHz 1H NMR spectra of enzyme-digested SBAs in D2O at 3340
Reference
- Ono, Yuko, Miyuki Takeuchi, and Akira Isogai. "Changes in neutral sugar composition, molar mass and molar mass distribution, and solid-state structures of birch and Douglas fir by repeated sodium chlorite delignification." Cellulose 29.4 (2022): 2119-2129.




