- Service Details
- Case Study
Introduce of Glycoprotein Structure
Glycoprotein structure refers to the molecular architecture of proteins that contain attached carbohydrate molecules, known as glycans. These molecules are crucial for various biological processes, including cell-cell recognition, cell signaling, and immune response. The structure of glycoproteins can vary widely depending on the specific protein and its function, as well as the type and arrangement of attached glycans.
Generally, glycoproteins consist of one or more polypeptide chains (made up of amino acids) covalently linked to one or more carbohydrate chains (made up of monosaccharide units). The carbohydrate chains can be attached to the protein backbone through either N-glycosidic bonds, where the glycan is linked to the nitrogen atom of an asparagine residue, or O-glycosidic bonds, where the glycan is linked to the oxygen atom of a serine or threonine residue.
The structure of glycoproteins can be highly heterogeneous, with variations in the length, composition, and branching patterns of the attached carbohydrate chains. This heterogeneity can have significant functional implications, influencing protein stability, solubility, and interaction with other molecules.
Glycoprotein structure analysis involves the study of the molecular architecture of proteins that contain attached carbohydrate molecules (glycans). Creative Proteomics offers glycoprotein structure analysis services. Glycoprotein structure analysis involves the comprehensive characterization of the structure of glycoproteins, including the identification of glycan composition, glycosylation sites, glycan heterogeneity, glycan-protein linkage analysis and decoding the primary, secondary, and tertiary structures of glycoproteins.
What Can We Provide:
| Service | Description |
|---|---|
| Intact Glycoproteins Analysis | Comprehensive analysis of intact glycoproteins to identify primary structure and glycan composition. |
| Glycan Composition Analysis | Determination of types and relative abundances of glycans attached to glycoproteins. |
| Glycan Linkage Analysis | Characterization of glycan-protein linkages, including N-linked and O-linked glycosylation. |
| Sialic Acid Analysis | Quantitative analysis of sialic acid content in glycoproteins. |
| N- and O-Linked Glycosylation Mapping | - N- and O-Glycan Profiling Service - N- and O-Glycosylation Site Analysis Services - N- and O-Glycan Linkage Analysis - N- and O-Glycosylation Site Occupation |
| Glycoprotein Amino Acid Sequence Analysis | Determination of the amino acid sequence of glycoproteins. |
| Protein Glycosylation Analysis | Mapping of glycoprotein post-translational modifications, including glycosylation. |
| Glycoprotein Secondary Structure Analysis | - Glycoprotein LC-MS Disulfide Bridge Mapping - Glycoprotein Circular Dichroism (CD) - Glycoprotein Fourier Transform Infrared (FTIR) - Glycoprotein Intrinsic Tryptophan Fluorescence (ITF) - Glycoprotein Differential Scanning Calorimetry (DSC) |
Technology Platform for Structural Analysis of Glycoproteins
Mass Spectrometry (MS):
- MALDI-TOF MS (Matrix-Assisted Laser Desorption/Ionization Time-of-Flight MS): Used for glycan profiling, glycosylation site mapping, and glycan heterogeneity analysis.
- ESI-MS (Electrospray Ionization MS): Enables the detection and characterization of intact glycoproteins, glycopeptides, and glycan structures.
- LC-MS (Liquid Chromatography-MS): Coupling liquid chromatography with MS allows for the separation and identification of glycopeptides and glycan structures.
Liquid Chromatography (LC):
- HPLC (High-Performance Liquid Chromatography): Used for glycan separation, glycopeptide enrichment, and glycoprotein analysis.
- UHPLC (Ultra-High-Performance Liquid Chromatography): Provides higher resolution and faster separations compared to conventional HPLC.
Nuclear Magnetic Resonance (NMR) Spectroscopy:
- 1D and 2D NMR: Provides structural information on glycoproteins and glycans, including glycosidic linkage, conformational flexibility, and interactions with other molecules.
Glycan Arrays:
- Microarray-based Platforms: Enable high-throughput screening of glycan-protein interactions and glycan-binding specificity.
Bioinformatics Tools:
- Database Search and Analysis Tools: Facilitate the interpretation of mass spectrometry data, glycan structure prediction, and glycoprotein database mining.
Sample Requirements for Glycoprotein Structure Analysis
| Sample Type | Sample Amount |
|---|---|
| Serum/Plasma | 100-500 µL |
| Cell Culture Supernatant | 1-5 mL |
| Tissue Homogenate | 50-200 mg |
| Cell Lysate | 1-10 x 10^6 cells |
| Urine | 1-10 mL |
| Saliva | 500 µL - 2 mL |
| CSF (Cerebrospinal Fluid) | 100-500 µL |
Applications of Glycoprotein Structure Analysis
Biological Function Elucidation: Glycoprotein structure analysis helps in understanding the role of glycans in various biological processes such as protein folding, stability, cell signaling, and cell-cell interactions. This aids in elucidating fundamental mechanisms in cell biology and physiology.
Glycan Diversity and Evolution: Studying glycoprotein structures across different species and evolutionary stages provides insights into the diversity and evolution of glycan structures. This knowledge aids in understanding the molecular basis of species-specific traits and evolutionary adaptations.
Quality Control in Bioprocessing: Analyzing glycoprotein structures is essential for quality control in bioprocessing industries such as food and beverage production, where glycoproteins play roles in product stability, texture, and sensory properties.
Environmental Monitoring: Glycoprotein analysis can be applied in environmental monitoring to assess the impact of environmental factors on glycan structures in organisms. Changes in glycosylation patterns may serve as biomarkers for environmental stressors and ecological health.
Forensic Science: Glycoprotein analysis can be utilized in forensic science for identifying and characterizing biological samples such as blood, saliva, and hair. Glycan profiling may provide additional information for sample identification and forensic analysis.
Developmental Biology: Investigating changes in glycoprotein structures during embryonic development and tissue differentiation contributes to our understanding of developmental biology and organogenesis. Glycan modifications play critical roles in cell differentiation and tissue morphogenesis.
Structural Glycobiology: Glycoprotein structure analysis provides valuable data for structural glycobiology, including glycan conformational studies, glycan-protein interactions, and glycan biosynthesis pathways. This information aids in elucidating the molecular mechanisms underlying glycan-mediated biological processes.
Bioinformatics and Computational Biology: Glycoprotein structure analysis generates large datasets that can be utilized in bioinformatics and computational biology for developing predictive models, analyzing glycan-protein interactions, and understanding glycan-related diseases.
Case. Investigation of Unconventional N-Glycosylation Patterns in Human Neutrophil Gelatinase-Associated Lipocalin (nCG)
Background:
Neutrophil gelatinase-associated lipocalin (nCG) plays a crucial role in immune response, and understanding its glycosylation patterns is essential for elucidating its functional significance. Previous studies have indicated subcellular-specific glycosylation in neutrophils, but comprehensive structural characterization is lacking.
Samples:
Purified human nCG originating from resting neutrophils isolated from healthy individuals was utilized for the study.
Technical Method:
Origin and Initial Handling of nCG:
- Purified nCG was obtained from Lee BioSolutions and assessed for purity using SDS-PAGE.
- Aliquots of nCG were stored at -20°C until use.
N-Glycan Release and Handling:
- Proteolytic inactivation of nCG was performed using PMSF.
- Reduction and alkylation of nCG were carried out.
- N-Glycans were released from the membrane and prepared for analysis.
Exoglycosidase Treatment of Released N-Glycans:
- Released and reduced N-glycans were digested with exoglycosidases.
- Exoglycosidases were removed by retention on strong cation exchange/C18.
PGC-LC-ESI-MS/MS-Based N-Glycome Profiling:
- N-Glycans were analyzed using capillary LC-MS/MS.
- Glycan structures were annotated and visualized.
In-Solution Glycopeptide Generation, Enrichment, and Deglycosylation:
- nCG was proteolytically digested using chymotrypsin.
- Glycopeptide enrichment was performed using ZIC-HILIC SPE.
- Deglycosylation of glycopeptides was carried out.
LC-MS/MS-Based N-Glycopeptide Analysis:
- Chymotryptic peptide mixtures were analyzed using LC-MS/MS.
- CID- and ETD-MS/MS were employed for glycopeptide analysis.
Intact nCG Profiling:
- Intact nCG glycoprotein was analyzed by ESI-MS in positive ion polarity mode.
- Spectra were deconvoluted to determine glycoform abundance.
Profiling nCG N-Glycans, N-Glycopeptides, and Intact Glycoprotein:
- N-Glycans and glycopeptides were manually characterized based on mass, fragmentation patterns, and retention times.
- Relative abundance of glycoforms was estimated from extracted ion chromatograms.
Results:
The comprehensive structural characterization of nCG glycosylation revealed unconventional patterns, including truncated chitobiose core, paucimannosidic, and monoantennary sialo N-glycans. These findings highlight the complexity and subcellular-specific nature of N-glycosylation in human neutrophils, providing insights into the functional role of nCG glycosylation in immune response.
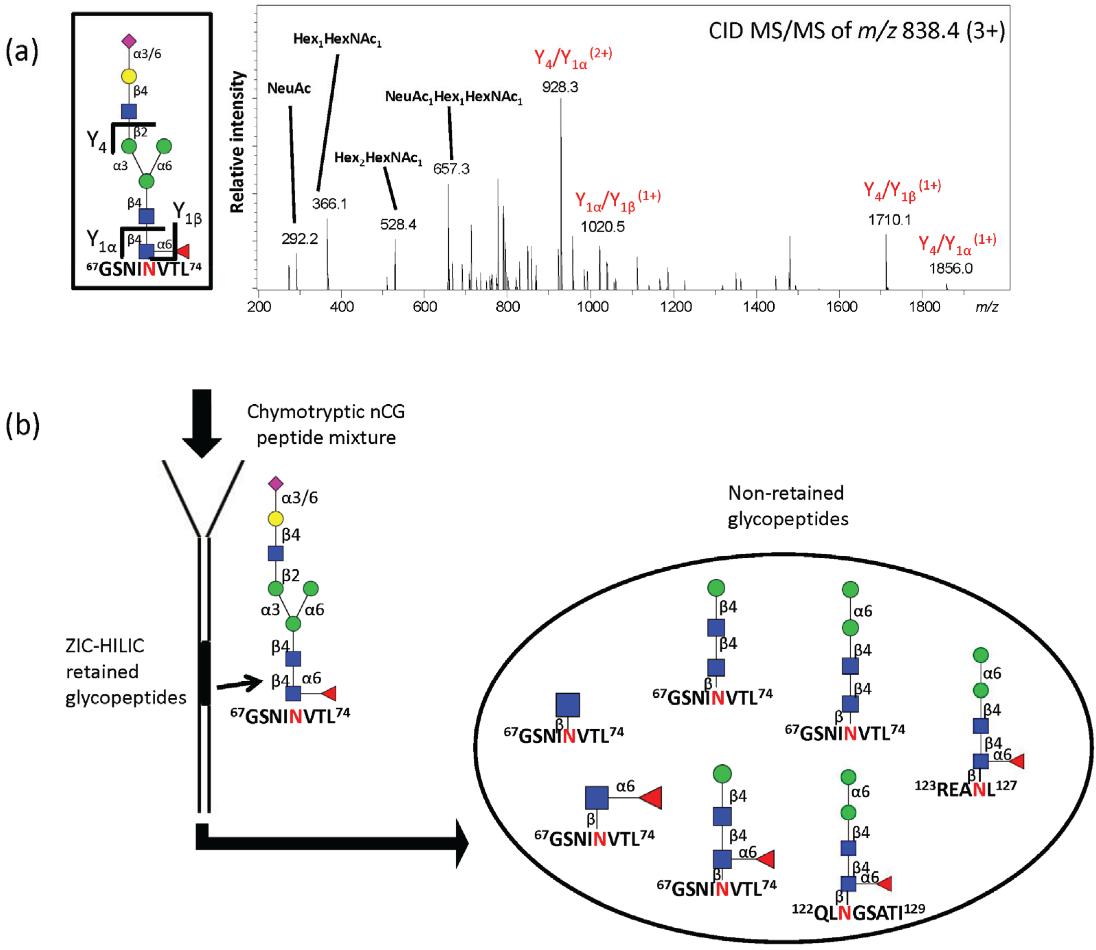 (a) RP-LC-CID-MS/MS confirming the structure of the single Asn71-glycopeptide of nCG retained on ZIC-HILIC SPE; (b) Schematic illustration showing the population of non-retained chymotryptic N-glycopeptides, i.e., Asn71, Asn126, and Asn124 containing glycopeptides of nCG, azurocidin, and NE, respectively, using ZIC-HILIC SPE for glycopeptide enrichment. All glycopeptides were identified by CID- and ETD-MS/MS.
(a) RP-LC-CID-MS/MS confirming the structure of the single Asn71-glycopeptide of nCG retained on ZIC-HILIC SPE; (b) Schematic illustration showing the population of non-retained chymotryptic N-glycopeptides, i.e., Asn71, Asn126, and Asn124 containing glycopeptides of nCG, azurocidin, and NE, respectively, using ZIC-HILIC SPE for glycopeptide enrichment. All glycopeptides were identified by CID- and ETD-MS/MS.
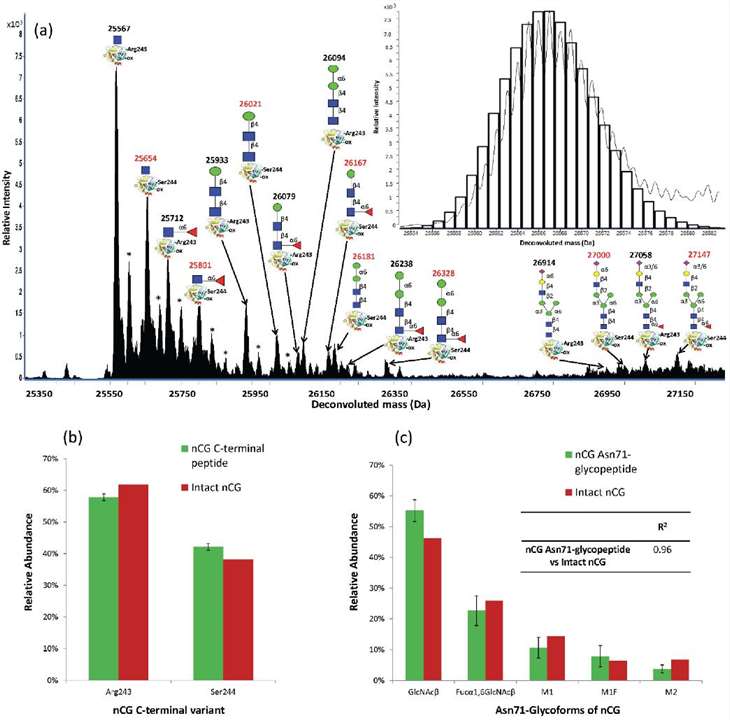 QTOF LC-ESI-MS glycoprofiling of intact nCG at relative low fragmentor potential (200 V).
QTOF LC-ESI-MS glycoprofiling of intact nCG at relative low fragmentor potential (200 V).
Reference
- Loke, Ian, Nicolle H. Packer, and Morten Thaysen-Andersen. "Complementary LC-MS/MS-based N-glycan, N-glycopeptide, and intact N-glycoprotein profiling reveals unconventional Asn71-glycosylation of human neutrophil cathepsin G." Biomolecules 5.3 (2015): 1832-1854.




