- Service Details
- Case Study
Nanotechnology has revolutionized the field of sugar-peptide detection, offering unparalleled precision and sensitivity in identifying these biomolecules. By leveraging nanoscale materials and techniques, researchers can enhance the detection limits and selectivity of assays for detecting sugars and peptides in biological samples.
One approach involves the use of functionalized nanoparticles, which can selectively bind to sugar or peptide molecules of interest. These nanoparticles are often modified with specific receptors or ligands that recognize the target molecules with high affinity. Additionally, nanomaterials such as carbon nanotubes or graphene have been employed to construct ultra-sensitive biosensors for sugar-peptide detection. These nanomaterial-based sensors can detect even trace amounts of target molecules due to their large surface area-to-volume ratio and unique electronic properties.
Furthermore, nanotechnology facilitates the development of miniaturized and portable detection devices, enabling point-of-care testing for sugars and peptides. These devices are particularly valuable in clinical settings, offering rapid and reliable analysis without the need for complex laboratory equipment.
Creative Proteomics offers a range of nanotechnology-based services tailored to meet diverse research needs in glycopeptide analysis:
Nanotechnology-Based Glycopeptide Analysis Services by Creative Proteomics
- Nanoparticle-Assisted Glycopeptide Enrichment: Utilizing functionalized nanoparticles as affinity probes to efficiently enrich glycopeptides from complex biological samples. Our methods enhance the recovery of low-abundance glycopeptides while minimizing non-specific binding, improving detection sensitivity and reproducibility.
- Nanomaterial-Enhanced Mass Spectrometry: Integrating nanomaterials with mass spectrometry (MS) platforms to enhance ionization efficiency and fragmentation patterns. This enables comprehensive characterization of glycopeptides, including the identification of glycan structures and site-specific glycosylation profiling with high accuracy.
- Nanoscale Biosensors for Glycopeptide Detection: Developing biosensing platforms that leverage nanomaterials for label-free and real-time detection of glycopeptides. These platforms offer sensitive and selective detection capabilities, facilitating rapid diagnostic applications and biomarker discovery.
Technical Platforms for Nanotechnology-Based Glycopeptide Analysis
The complexity of glycopeptide analysis demands high-quality, state-of-the-art technical platforms. At Creative Proteomics, our nanotechnology-based glycopeptide detection platforms focus on enhancing precision, speed, and efficiency in biomolecular analysis.
Our platforms include:
- Surface Plasmon Resonance Nano-chip (SPRNC): With its ability to detect molecular interactions in real-time without labeling, our SPRNC is at the vanguard of glycopeptide analysis.
- Nano Liquid Chromatrography-Mass Spectrometry (Nano LC-MS): Nano LC-MS allows in-depth characterization, separation, and identification of minute quantities of glycopeptides with great accuracy.
- Nano-HPLC (high-performance liquid chromatography): Offering high resolution, great sensitivity, and increased speed, our Nano-HPLC significantly optimizes the process of glycopeptide detection.
- Nano-Structure Initiator Mass Spectrometry (NIMS): With NIMS, we achieve direct and highly sensitive detection of glycopeptides, even from complex mixtures, without the need for extensive sample preparation or separation.
Sample Requirements for Glycopeptide Quantitative Analysis
| Sample Type | Sample Volume | Additional Notes |
|---|---|---|
| Serum/Plasma | 100-200 μL | Preferably collected using EDTA or heparin as anticoagulants |
| Tissue Homogenate | 10-50 mg | Fresh-frozen or freshly prepared samples are recommended |
| Cell Lysate | 1-5 x 106 cells | Harvested from cultured cells using appropriate methods |
| Urine | 500-1000 μL | Preferably collected in a sterile container |
| Saliva | 200-500 μL | Collected using non-invasive methods |
| Cerebrospinal Fluid | 100-200 μL | Preferably collected by a trained healthcare professional |
Applications of Nanotechnology-based Glycopeptide Detection and Analysis
Biomedical Research: Nanotechnology enhances understanding of glycopeptides' roles in cellular signaling, protein interactions, and disease mechanisms.
Drug Discovery: Nanotechnology expedites screening of compounds targeting glycopeptides, accelerating the development of novel therapeutics.
Precision Drug Delivery: Nanotechnology enables targeted delivery of therapeutics to cells expressing specific glycopeptide markers, improving treatment efficacy.
Biomarker Discovery: Nanotechnology facilitates sensitive detection of glycopeptides, leading to the identification of novel biomarkers for disease diagnosis and monitoring.
Functional Proteomics: Nanotechnology-integrated methods enable comprehensive analysis of glycopeptides in complex biological samples, revealing insights into disease processes.
Regenerative Medicine: Nanotechnology aids in designing biomimetic scaffolds for tissue engineering, leveraging glycopeptides to enhance tissue regeneration and wound healing.
Environmental Monitoring: Nanotechnology-based sensors detect glycopeptide contaminants in environmental samples, aiding in pollution control and remediation efforts.
Title: Advancements in Nanotechnology for Glycoproteomics Analysis
Background
Protein glycosylation, a common post-translational modification, plays a crucial role in various biological processes and is implicated in diseases such as cancer. Detecting changes in glycoproteins can aid in disease diagnosis and therapy. However, traditional analytical methods face challenges in identifying glycopeptides from complex samples.
Technical Methods
1. Glycoprotein- and Glycopeptide-based Analysis:
- Glycoproteins are enriched using various separation methods, while glycopeptides undergo enzymatic or chemical degradation before enrichment.
- Enrichment methods include size exclusion, ion exchange, affinity chromatography, chemical immobilization, lectin-affinity chromatography, boronic acid-based approaches, hydrazide chemistry, and solid-phase extraction using hydrophilic interactions.
- Enriched glycopeptides are deglycosylated and quantified via mass spectrometry (MS) analysis, enabling identification of glycosylation sites and structures.
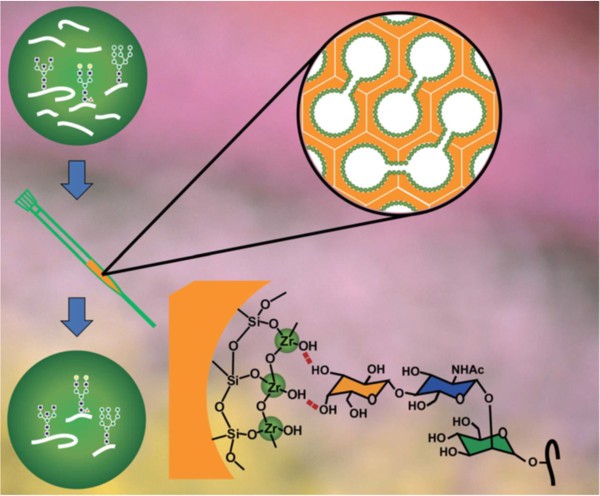 A schematic representation of glycopeptide isolation using mesoporous silica microspheres coated with zirconia
A schematic representation of glycopeptide isolation using mesoporous silica microspheres coated with zirconia
2. Nanotechnology Platforms:
- Nanoparticles: Surface-modified nanoparticles, such as those with boronic acid or polysaccharides, offer efficient glycopeptide enrichment.
- Mesoporous Materials: Functionalized mesoporous silica and mesoporous carbon provide high-affinity binding sites for glycopeptides, enhancing detection sensitivity.
- Two-Dimensional Crystalline Materials: Graphene and carbon nanotubes (CNTs) facilitate rapid and efficient glycan enrichment, enabling enhanced glycopeptide detection.
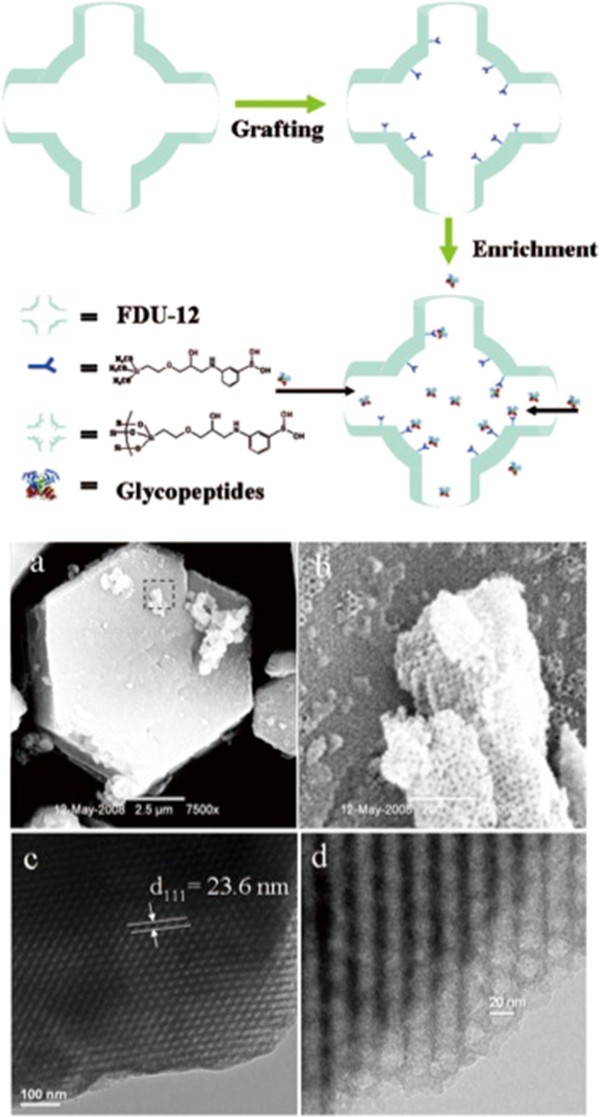 A schematic of glycopeptide enrichment from dilute solutions using mesoporous silica material modified with boronic acid.
A schematic of glycopeptide enrichment from dilute solutions using mesoporous silica material modified with boronic acid.
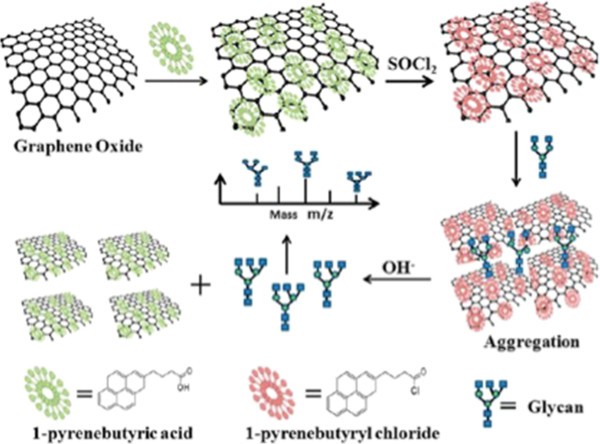 Using functionalized graphene for glycan separation
Using functionalized graphene for glycan separation
Results
Transition metal oxides coupled with mesoporous silica enable effective glycopeptide isolation via hydrophilic interaction chromatography.
Oxidized mesoporous carbon demonstrates remarkable hydrophilicity, enabling efficient glycopeptide separation prior to mass spectrometry profiling.
Functionalized graphene and CNTs serve as effective platforms for glycan enrichment and glycopeptide detection, enhancing sensitivity and selectivity.
Reference
- Zhao, Hu, Yaojun Li, and Ye Hu. "Nanotechnologies in glycoproteomics." Clinical proteomics 11 (2014): 1-10.




