- Service Details
- Case Study
Glycoproteomics, a specialized branch of proteomics, focuses on the study of glycosylated proteins, or more specifically, glycopeptides. Being a pivotal PTM, glycosylation directly correlates with various dynamic biological processes, including cell-cell signaling, molecular recognition, immune responses, and disease progression. Consequently, glycopeptides bear significant potential as practical biomarkers for diagnosing and treating various debilitating diseases, such as cancer, cardiovascular disease, and neurodegenerative disorders.
Glycopeptides analysis provides invaluable insights into the glycosylation sites, glycan structure, and attachment sites, which hold immense influence on the protein function and stability. This information is, thereby, progressively becoming critical for drug discovery, disease profiling, and therapeutic applications.
Creative Proteomics' Glycopeptides Analysis Solutions
Glycopeptide Enrichment and Preparation: We use enrichment strategies such as hydrophilic interaction liquid chromatography (HILIC) and lectin chromatography to improve the sensitivity of glycopeptide detection in complex protein mixtures.
High-Throughput Glycopeptide Identification/ Glycopeptide Sequence Analysis: Glycopeptide characterization is optimized based on mass spectrometry. This includes identification of glycosylation sites, mapping of glycopeptide sequences, and elucidation of glycan structures.
Quantitative Glycopeptide Analysis: Given the dynamic nature of glycosylation and its role in disease progression, it is critical to ensure accurate quantitative data on differentially expressed glycopeptides. We rely on reliable quantification techniques such as label-free quantification, isotopic tagging (iTRAQ) for relative and absolute quantification, and tandem mass tagging (TMT) in combination with mass spectrometry to help achieve accurate quantification.
Nanotechnology-based Glycopeptide Analysis: Leveraging nanotechnology platforms, we develop innovative approaches for sensitive and selective detection of glycopeptides, enhancing the efficiency and sensitivity of glycoprotein analysis.
Additionally, Creative Proteomics offers other specialized services:
Monosaccharide Analysis & Sialic Acid Analysis: Quantitative analysis of monosaccharide composition and sialic acid content in glycopeptides.
Complete Glycan Typing Analysis: Comprehensive characterization of glycan structures attached to glycopeptides.
Unlabeled Glycan Analysis: Analysis of unlabeled glycopeptides to identify glycan composition and peptide sequence.
Site-Specific Glycan Analysis: Determination of glycosylation sites on proteins, essential for understanding structure-function relationships.
Technical Platforms for Glycopeptide Analysis
At Creative Proteomics, we utilize state-of-the-art technical platforms to ensure the highest quality and accuracy in glycopeptide analysis.
Thermo Scientific Orbitrap Fusion Lumos Tribrid Mass Spectrometer
The Orbitrap Fusion Lumos Tribrid mass spectrometer offers unparalleled flexibility in fragmentation techniques and MSn functionality, enabling in-depth analysis of glycopeptides. Its advanced capabilities, including multiple fragmentation modes (CID, HCD, ETD), allow for precise localization of glycosylation sites and characterization of glycan structures. Combined with the Biopharma Finder 3.0 software, this platform streamlines data analysis and interpretation, facilitating comprehensive glycopeptide profiling.
Vanquish Flex UHPLC Systems
The Vanquish Flex series of ultra-high-performance liquid chromatography (UHPLC) systems, equipped with binary or quaternary solvent mixing capabilities, ensures optimal separation and resolution of glycopeptides. With features such as precise gradient formation and compatibility with various detection modes (DAD, VWD, MS), these systems deliver robust and reproducible chromatographic results, essential for accurate glycopeptide quantification and identification.
Thermo Scientific Acclaim 120 C18 Columns
The Acclaim 120 C18 chromatography columns, specifically designed for reverse-phase peptide separation, offer exceptional resolution and peak shape for glycopeptide analysis. Their low-metal content silica-based matrix minimizes peak tailing effects, ensuring superior chromatographic performance and reliable glycopeptide detection.
Sample Requirements for Glycopeptides Analysis
| Sample Type | Sample Quantity |
|---|---|
| Serum or Plasma | 100-200 μL |
| Cell Culture Supernatant | 1-2 mL |
| Tissue Homogenate | 10-50 mg |
| Cell Lysate | 1-5 million cells |
| Urine | 1-2 mL |
| Cerebrospinal Fluid | 100-200 μL |
| Saliva | 100-200 μL |
| Synovial Fluid | 100-200 μL |
Applications of Glycopeptides Analysis
Biomedical Research: Glycopeptides play crucial roles in various biological processes, including cell signaling, immune response, and protein folding. Analyzing glycopeptides helps in understanding these processes and their implications in health and disease. For example, studying glycopeptides can aid in cancer biomarker discovery, monitoring disease progression, and understanding the immune response to pathogens.
Pharmaceuticals: Glycopeptides are important targets for drug discovery and development. Analyzing glycopeptides can help in characterizing the glycosylation patterns of therapeutic proteins, ensuring their safety, efficacy, and consistency. This is particularly important in the production of biopharmaceuticals such as monoclonal antibodies, where glycosylation can affect their pharmacokinetics and immunogenicity.
Clinical Diagnostics: Glycopeptides can serve as biomarkers for various diseases, including cancer, autoimmune disorders, and infectious diseases. Analyzing glycopeptides in biological samples such as blood, urine, and tissues can provide valuable diagnostic information, aiding in early detection, prognosis, and personalized treatment strategies.
Glycomics: Glycopeptides are integral components of the glycome, which encompasses all glycans (carbohydrates) in an organism. Glycomics focuses on the systematic study of glycan structures and their functions. Analyzing glycopeptides is essential for unraveling the complexity of the glycome and understanding its roles in physiology and pathology.
Food and Agriculture: Glycopeptides are present in various food sources, including milk, eggs, and plants. Analyzing glycopeptides in food products can provide insights into their nutritional quality, authenticity, and safety. Additionally, glycopeptide analysis can be used to detect food adulteration and ensure compliance with regulatory standards.
Environmental Monitoring: Glycopeptides produced by microorganisms play important roles in environmental processes such as biodegradation and nutrient cycling. Analyzing glycopeptides in environmental samples can help in assessing microbial activity, monitoring pollution levels, and understanding ecosystem dynamics.
Forensics: Glycopeptides found in bodily fluids such as saliva, sweat, and tears can serve as potential forensic markers. Analyzing glycopeptides in forensic samples can aid in identifying individuals, determining the timing of events, and providing evidence in criminal investigations.
Title: High-throughput Method for Comprehensive Characterization of N-linked Glycoproteins
Background
Glycosylation, particularly N-linked glycosylation, is a common protein modification associated with various diseases. Traditional glycoproteomic analysis methods are limited in throughput and reproducibility, necessitating the development of high-throughput approaches to efficiently characterize protein glycosylation.
Sample
The study utilizes human urine samples and a glycoprotein standard (fetuin) to validate the high-throughput method for glycoproteomic analysis.
Technical Methods
The high-throughput method for comprehensive characterization of N-linked glycoproteins consists of a sequential workflow employing tip-based procedures. Initially, proteins are digested using trypsin and bound to C4-Tips for proteolytic digestion. Subsequently, intact glycopeptides (IGPs) are enriched using C18/MAX-Tips, while glycans and deglycosylated peptides are separated using C18-Tips. The enriched IGPs undergo LC–MS/MS analysis, while glycans are analyzed using MALDI-MS. Optimization steps include determining the optimal peptide binding time and validation using fetuin as a glycoprotein standard. The method's efficiency and specificity are assessed through evaluation of enrichment effectiveness and reproducibility using human urine samples. Overall, this method provides a rapid, reproducible, and comprehensive approach for studying N-linked glycoproteins in complex biological samples.
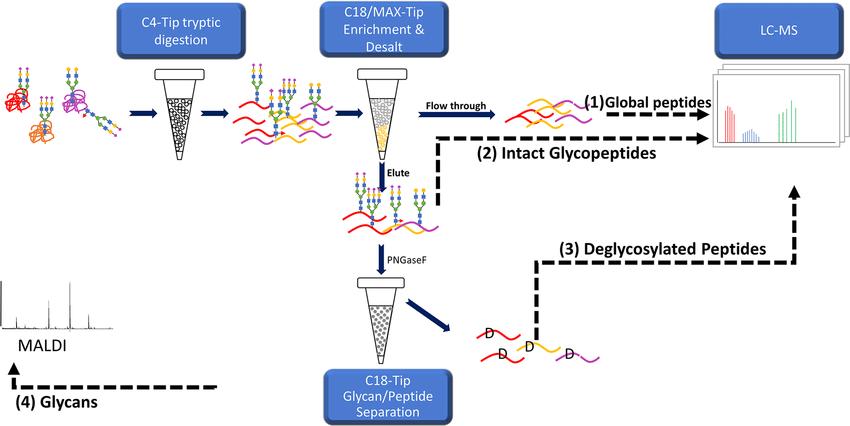 Workflow for glycans, glycosite, and intact glycopeptide analysis.
Workflow for glycans, glycosite, and intact glycopeptide analysis.
Results
The optimized method demonstrates rapid and efficient enrichment of global peptides, IGPs, deglycosylated peptides, and glycans from fetuin and human urine samples. The technique offers comprehensive coverage of glycopeptides carrying diverse glycans, including sialylated, oligomannose, and fucosylated glycans. Evaluation of IGP enrichment efficiency and reproducibility shows high specificity and reduced presence of nonglycosylated peptides in the final eluate.
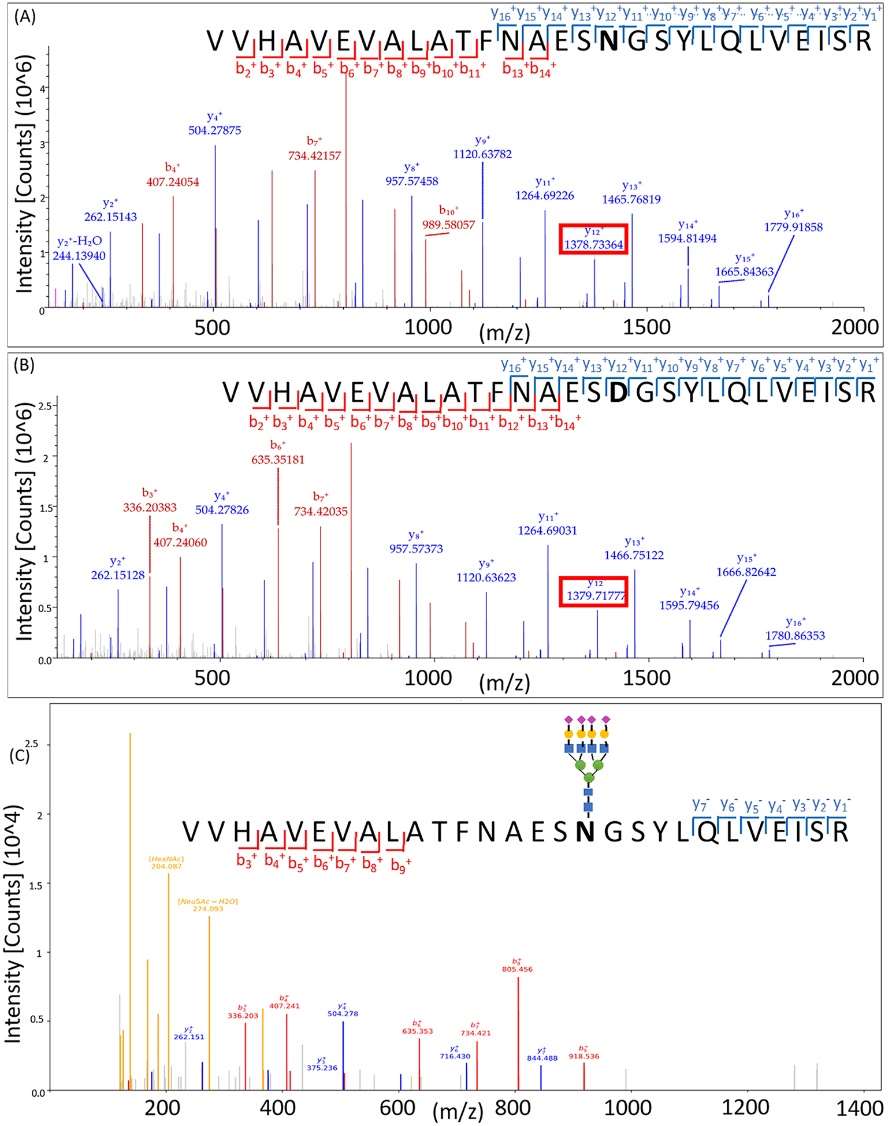 Nonglycosylated peptide and deglycosylated peptide spectra from the same glycosylation site of fetuin indicating site specific information and enrichment specificity.
Nonglycosylated peptide and deglycosylated peptide spectra from the same glycosylation site of fetuin indicating site specific information and enrichment specificity.
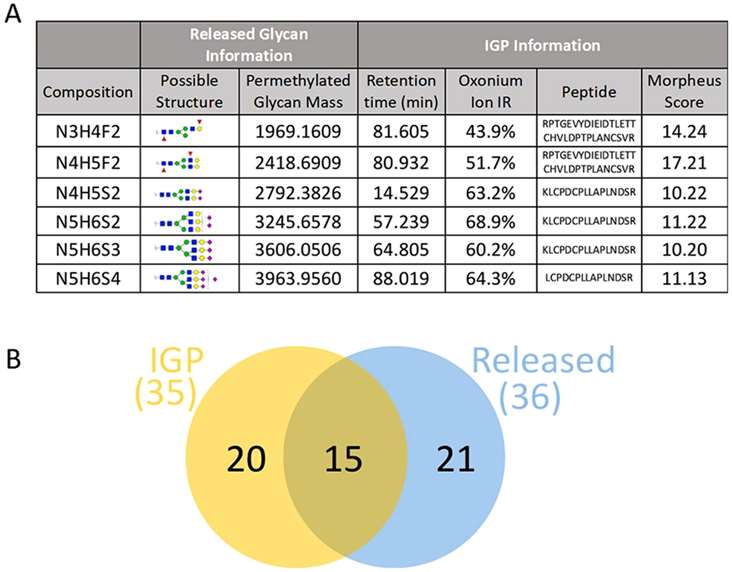 Fetuin glycan analysis using released glycans and IGPs.
Fetuin glycan analysis using released glycans and IGPs.
Reference
- Chen, Shao-Yung, et al. "Glycans, glycosite, and intact glycopeptide analysis of N-linked glycoproteins using liquid handling systems." Analytical chemistry 92.2 (2019): 1680-1686.




