- Service Details
- Case Study
Disaccharides, comprising two monosaccharide units linked by a glycosidic bond, represent a fundamental class of carbohydrates ubiquitous in biological systems. From sucrose in plants to lactose in mammalian milk, these molecules serve as essential sources of energy and structural components. Analyzing disaccharides is imperative due to their pivotal roles in various physiological processes.
Understanding the composition and structure of disaccharides is essential for elucidating metabolic pathways and assessing nutritional intake. Disaccharide analysis enables precise quantification of sugars in food products, aiding in dietary planning and health management. Furthermore, aberrations in disaccharide metabolism are implicated in metabolic disorders such as diabetes mellitus and lactose intolerance. Accurate analysis of disaccharides in biological samples facilitates disease diagnosis and therapeutic interventions.
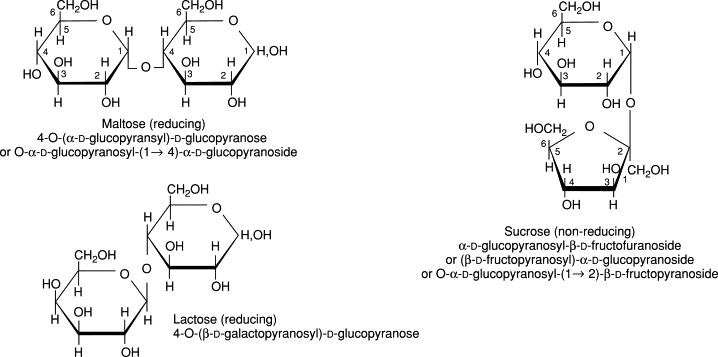 Formulae and nomenclature of three common disaccharides (R.F. Tester et al., 2003).
Formulae and nomenclature of three common disaccharides (R.F. Tester et al., 2003).
Moreover, disaccharide analysis drives advancements in biotechnology and biomedical research. Detailed characterization of glycan structures and carbohydrate-protein interactions contributes to drug development, immunotherapy, and understanding disease mechanisms. Thus, disaccharide analysis plays a critical role in unraveling the complexities of carbohydrate biology, fostering innovations in healthcare, food science, and industry.
Creative Proteomics offers a comprehensive suite of disaccharide analysis services tailored to meet the diverse needs of researchers and industry partners.
Disaccharide Analysis Services in Creative Proteomics
Glycan Profiling Techniques
Creative Proteomics utilizes state-of-the-art chromatographic and mass spectrometric techniques to conduct comprehensive glycan profiling, which characterizes the abundance, composition, and structural diversity of disaccharides in biological samples. Our key methodologies include:
- Hydrophilic Interaction Chromatography (HILIC): Optimized for polar compounds, HILIC effectively separates glycan molecules based on their hydrophilicity, crucial for analyzing complex mixtures of carbohydrates.
- Porous Graphitized Carbon Chromatography (PGC): This technique is particularly useful for the separation of structurally related isomers and highly polar glycans, providing detailed insights into glycan heterogeneity.
- Tandem Mass Spectrometry (MS/MS): MS/MS offers unmatched specificity and sensitivity in identifying and quantifying minute amounts of disaccharides, supporting both structural elucidation and quantitative analysis.
Disaccharide Profiling and Quantitative Analysis
In addition to glycan profiling, we conduct disaccharide profiling to explore the specific disaccharides present in samples, which aids in:
- Metabolic Pathway Exploration: Understanding how disaccharides metabolize in biological systems helps trace pathways that may be pivotal in disease processes or metabolic functions.
- Disease Mechanism Insights: Profiling disaccharides can reveal abnormalities or changes in carbohydrate metabolism associated with various diseases, providing targets for therapeutic intervention.
Glycosidic Bond Analysis
By employing enzymatic cleavage and chemical derivatization strategies, we analyze glycosidic bond linkages within disaccharide molecules. Using specific glycosidases and glycan-modifying reagents, this analysis helps elucidate:
- Connectivity and Stereochemistry: Detailed understanding of glycosidic linkages informs on the biosynthesis and structural intricacies of carbohydrate molecules, enhancing our understanding of their biological functions.
Advanced Structural Elucidation
Our structural elucidation uses high-end spectroscopic techniques combined with computational tools:
- Nuclear Magnetic Resonance (NMR) Spectroscopy: NMR provides detailed information about the molecular structure and dynamics of disaccharides at the atomic level.
- Fourier-Transform Infrared (FTIR) Spectroscopy: FTIR spectroscopy helps in identifying functional groups and characterizing molecular bonding and structure.
- Computational Modeling and Bioinformatics: These tools are integral for interpreting complex data, modeling molecular structures, and predicting functional sites on carbohydrates.
Custom Services for Targeted Needs
Technical Platforms for Disaccharide Analysis
High-Performance Liquid Chromatography (HPLC)
- Reverse-Phase HPLC: Ideal for non-polar to moderately polar disaccharides, using hydrophobic stationary phases and a gradient elution of water and organic solvents.
- Ion-Exchange Chromatography: Used for charged disaccharides, separating molecules based on their charge at specific pH levels.
- Size-Exclusion Chromatography: Suitable for determining the molecular size and shape of disaccharides, useful in understanding polymer distributions and molecular weight.
Mass Spectrometry (MS)
- Liquid Chromatography-Mass Spectrometry (LC-MS): Combines the separation capabilities of liquid chromatography with the mass analysis power of MS, ideal for complex sample analysis.
- Matrix-Assisted Laser Desorption/Ionization Mass Spectrometry (MALDI-MS): Excellent for high-throughput analysis of carbohydrates, allowing rapid characterization of disaccharide compositions and structures.
Nuclear Magnetic Resonance (NMR) Spectroscopy
Fourier-Transform Infrared (FTIR) Spectroscopy
List of Detectable Disaccharide
| Sucrose | Lactose | Maltose | Trehalose | Cellobiose |
| Isomaltose | Sophorose | Melibiose | Gentiobiose | Laminaribiose |
| Turanose | D-Mannobiose | D-Turanose | D-Allose | D-Gulose |
| D-Idose | D-Galactose | D-Glucose | D-Mannose | D-Xylose |
| D-Lyxo-hexose | D-Ribose | D-Arabinose | L-Rhamnose | D-Altrose |
| D-Gulonic acid | D-Galacturonic acid | D-Glucuronic acid | D-Mannuronic acid | D-Talose |
| D-Tagatose | L-Fucose | L-Rhamnulose | L-Arabinose | L-Altrose |
| L-Idose | L-Gulose | L-Galactose | L-Glucose | L-Mannose |
| L-Xylose | L-Ribose | L-Allose | L-Lyxose |
Sample Requirements for Disaccharide Analysis
| Sample Type | Minimum Volume | Optimal Volume | Analysis Technique | Comments |
|---|---|---|---|---|
| Biological Fluids | 50 µL | 200 µL | LC-MS, NMR, HPLC | Includes plasma, serum, urine. |
| Tissue Samples | 10 mg | 50 mg | HPLC, MS | Homogenize thoroughly to ensure consistency. |
| Cell Cultures | 1 x 10^6 cells | 5 x 10^6 cells | LC-MS, NMR | Cells should be pelleted and washed. |
| Plant Extracts | 20 mg | 100 mg | HPLC, MS | Dry and grind to a fine powder for uniform extraction. |
| Food Products | 20 mg | 100 mg | HPLC, FTIR, NMR | Homogenization is crucial for heterogeneous samples. |
| Synthetic Mixtures | 10 µL | 50 µL | MALDI-MS, LC-MS | Ensure samples are well-dissolved in a suitable solvent. |
Applications of Disaccharide Analysis
Biomedical Research
- Disease Diagnosis: Disaccharide levels serve as biomarkers for conditions like diabetes mellitus and certain cancers.
- Drug Development: Understanding disaccharide structures aids in developing therapeutic agents targeting glycoproteins and glycolipids.
Food Industry
- Quality Control: Disaccharide analysis ensures food product labels are accurate and complies with regulations.
- Processing Optimization: Monitoring disaccharide changes during processing improves product quality and shelf life.
Environmental Sciences
- Microbial Activity: Disaccharide levels indicate microbial activity in ecosystems, aiding in environmental monitoring.
Agricultural Research
- Plant Breeding: Disaccharide analysis helps breeders select plant traits for improved yield and stress resistance.
- Soil Health: Analyzing disaccharides in soil assesses microbial activity and nutrient cycling, impacting plant growth and agricultural practices.
Title: Glycosaminoglycans (GAGs) in Pluripotent Cells Using Teratocarcinoma Cells as a Model
Background
Glycosaminoglycans (GAGs) are essential components of the extracellular matrix and play crucial roles in various biological processes, including cell proliferation, differentiation, and signaling. Understanding the structure and function of GAGs in pluripotent cells such as embryonic stem cells (ESCs) and teratocarcinoma cells is vital for elucidating their roles in development and disease.
Sample
The study utilizes teratocarcinoma cells (NCCIT), a model for pluripotent cells, to investigate the glycosaminoglycanome (GAGome) and its dynamics during differentiation. NCCIT cells are cultured and harvested for GAG extraction and analysis.
Technical Methods
Cell Culture and Harvesting: NCCIT cells are cultured and maintained in a specific medium, with regular medium changes and passaging to ensure optimal cell growth.
Recovery and Purification of GAGs: GAGs are extracted from lyophilized NCCIT cells using a multistep procedure involving defatting, proteolysis, and extraction with CHAPS/Urea. GAGs are then purified using ion chromatography and desalting spin columns.
Analysis of Intact GAGs: Molecular weights of purified GAGs (chondroitin sulfate and heparan sulfate) are analyzed by polyacrylamide gel electrophoresis (PAGE) with Alcian blue staining.
GAG Depolymerization: Purified GAGs are enzymatically depolymerized into disaccharides using chondroitinase and heparinase enzymes.
Disaccharide Analysis by LC-MS: Disaccharides from GAG depolymerization are analyzed by liquid chromatography-mass spectrometry (LC-MS) using optimized elution conditions for CS/DS and heparin/HS disaccharides.
Results
The study provides comprehensive insights into the structure and composition of GAGs in pluripotent cells, revealing changes in the GAGome during differentiation. Molecular weight analysis, disaccharide composition, and abundance of GAGs are elucidated, enhancing our understanding of their functional roles in stem cell biology.
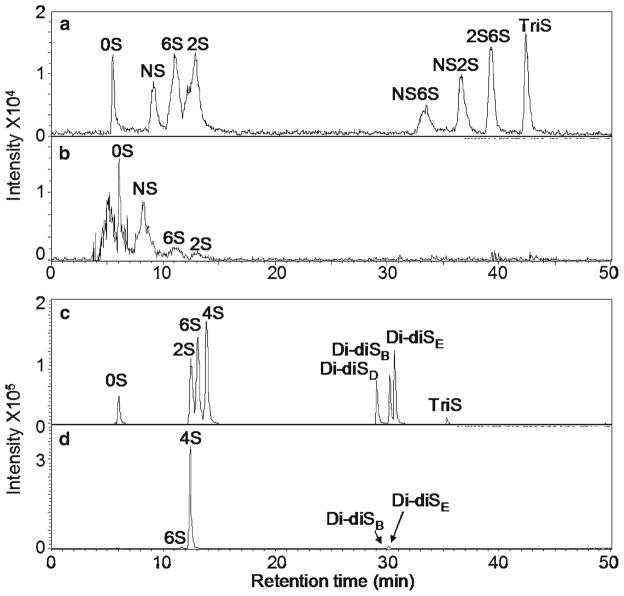 LC-MS disaccharide analysis of NCCIT cells.
LC-MS disaccharide analysis of NCCIT cells.
Reference
- Li, Boyangzi, et al. "Analysis of glycosaminoglycans in stem cell glycomics." Embryonic Stem Cell Therapy for Osteo-Degenerative Diseases: Methods and Protocols (2011): 285-300.




