- Service Details
- Case Study
- Description
What is Mannose?
Mannose is a monosaccharide, a type of simple sugar, with the molecular formula C6H12O6. It is classified as an aldohexose, meaning it contains six carbon atoms and an aldehyde functional group. Mannose is found naturally in various foods such as fruits, vegetables, and grains. It plays essential roles in biological processes such as glycosylation, where it serves as a precursor for the synthesis of glycoproteins and glycolipids. Additionally, mannose is involved in cell-cell recognition and immune response modulation.
At Creative Proteomics, our team of experts employ various innovative methodologies and advanced technologies to provide comprehensive mannose analysis, granting our clients and partners unprecedented breakthroughs in scientific research.
Mannose Analysis Services in Creative Proteomics
Quantitative Analysis of Mannose: Accurate determination of Mannose levels in various sample types using high-performance liquid chromatography (HPLC), mass spectrometry (MS), or enzymatic assays.
Structural Characterization of Mannose-containing Molecules: Elucidation of the structure and composition of Mannose-containing compounds, including glycoproteins, glycolipids, and polysaccharides, using advanced analytical techniques such as nuclear magnetic resonance (NMR) spectroscopy and liquid chromatography-mass spectrometry (LC-MS).
Metabolic Profiling of Mannose Metabolism: Investigation of Mannose metabolism pathways and related metabolites in biological samples through metabolomics analysis, providing insights into physiological and pathological processes.
Glycomic Analysis of Mannose Glycans: Profiling and characterization of Mannose-containing glycans and glycoconjugates in complex biological samples using techniques such as lectin microarrays, glycan microarrays, and capillary electrophoresis.
Biomarker Discovery and Validation: Identification and validation of Mannose-related biomarkers for disease diagnosis, prognosis, and therapeutic monitoring through targeted proteomics and metabolomics approaches.
Functional Studies of Mannose-binding Proteins: Assessment of the binding affinity and specificity of Mannose-binding proteins (lectins) using various binding assays, including surface plasmon resonance (SPR) and isothermal titration calorimetry (ITC).
Quality Control and Method Development: Development of customized analytical methods and quality control protocols for Mannose analysis in compliance with regulatory standards and industry guidelines.
Technical Platforms for Mannose Analysis
High-Performance Liquid Chromatography (HPLC)
HPLC offers precise quantification and separation capabilities. With its high sensitivity and excellent resolution, HPLC enables accurate measurement of mannose concentrations in diverse samples, making it indispensable for quality control and research purposes. Its versatility allows for the analysis of various sample types and matrices, providing valuable insights into mannose content and purity.
- Detection Limit: < 0.1 μg/mL
- Quantification Limit: < 0.5 μg/mL
- Precision: Relative standard deviation < 2%
- Accuracy: Recovery rate between 95% and 105%
- Calibration Curve: Linear over the concentration range of 1-100 μg/mL
- Solvent: HPLC-grade water and methanol
- Chromatographic Conditions: Column - C18, Mobile phase - 80:20 water:methanol, Flow rate - 1 mL/min, Detection wavelength - 210 nm
- Reporting Format: Concentration reported in μg/mL
Gas Chromatography-Mass Spectrometry (GC-MS)
GC-MS excels at identifying and quantifying mannose, even at trace levels within complex mixtures. Its ability to analyze volatile and semi-volatile compounds makes it suitable for a wide range of sample types, including biological samples and environmental matrices. GC-MS plays a vital role in characterizing mannose in diverse applications, from pharmaceutical formulations to environmental studies.
Nuclear Magnetic Resonance (NMR) Spectroscopy
NMR Spectroscopy offers unique insights into the structural characterization of mannose. By probing the magnetic properties of atomic nuclei, NMR provides detailed information about the molecular conformation, configuration, and interactions of mannose molecules. This non-destructive technique is particularly valuable for elucidating complex structures and studying mannose in solution.
UV-Vis and Infrared (IR) Spectroscopy
UV-Vis spectroscopy allows for quick qualitative assessments of mannose samples based on their absorption of ultraviolet and visible light. IR spectroscopy, on the other hand, provides valuable information about functional groups present in mannose molecules, aiding in structural elucidation. These user-friendly techniques complement other analytical methods, providing valuable insights into mannose composition and properties.
Sample Requirements for Mannose Analysis
| Sample Type | Recommended Sample Amount |
|---|---|
| Serum/Plasma | 100-200 µL |
| Urine | 1-2 mL |
| Tissue | 10-50 mg |
| Cells/Culture | 1-5 million cells |
| Food/Supplements | 1-5 grams |
| Pharmaceuticals | 10-50 mg |
| Environmental Samples | 1-10 mL (e.g., water, soil extracts) |
| Biological Fluids | 100-200 µL (e.g., saliva, cerebrospinal fluid) |
| Plant Extracts | 10-50 mg (e.g., leaf, root extracts) |
Deliverables for Mannose Analysis
- A detailed analysis report with comprehensive data interpretation.
- Raw and processed numerical data files.
- Detailed methodology of the technologies and strategies used.
- Support for further and future analysis and interpretation.
Advantages of Creative Proteomics' Technical Platforms
- Versatility: Our technical platforms cover a wide range of analytical techniques suitable for different aspects of mannose analysis.
- Precision: High sensitivity and resolution of our instruments ensure accurate quantification and characterization of mannose and related compounds.
- Reliability: Rigorous quality control measures and calibration procedures guarantee the reliability and reproducibility of our analytical results.
- Expertise: Our team of scientists is proficient in operating and interpreting data from various analytical instruments, providing valuable insights into mannose analysis.
Title: Quantification of D-Mannose in Human Plasma Using LC-MS-MS: Method Development and Clinical Implications
Background
D-Mannose, a monosaccharide, has emerged as a potential biomarker for insulin resistance and sensitivity, particularly in patients with type 2 diabetes. However, accurate quantification of D-mannose in human plasma presents challenges due to its similarity to other sugars like D-glucose and D-galactose, necessitating the development of robust analytical methods.
Samples
Plasma samples were obtained from both healthy volunteers and patients diagnosed with type 2 diabetes. After overnight fasting, blood samples were collected and centrifuged for plasma separation. These samples were then stored at -20°C until analysis.
Technical Methods
The LC-MS-MS method employed in this study utilized stable isotope-labeled 13C6-D-mannose as an internal standard (IS) to compensate for variability in critical parameters such as MS ionization efficiency and sample stability during preparation. Chromatographic separation was achieved using a HILICpak VG-50 4E column, specifically chosen for its ability to separate D-mannose from interfering epimers like D-glucose and D-galactose. The column's polymer-based amino material facilitated hydrophilic interaction chromatography (HILIC), providing adequate separation efficiency.
Method validation followed EMA guidelines and included assessments of selectivity, sensitivity, accuracy, precision, and linearity. Selectivity was ensured by separate injections of water solutions containing D-mannose, D-glucose, and D-galactose, as well as combined solutions, to monitor retention times and exclude co-elution. Linearity was confirmed within a concentration range of 0.31–40.0 μg/mL, with calibration curves constructed using eight standard solutions. Instrumental sensitivity was evaluated with a lower limit of quantitation (LLOQ) of 1.25 μg/mL, which was at least six times lower than the lowest mannose concentration in the samples.
Matrix effects were assessed by comparing peak areas of 13C6-D-mannose in water solution to those in human plasma matrices, revealing a suppression of approximately 38%. Recovery rates varied, with higher values observed in water solutions compared to human plasma, indicating potential interference from plasma proteins affecting elution efficiency. Intra-day and inter-day accuracy and precision were within acceptable limits, demonstrating the method's reproducibility and reliability.
Results
The developed method exhibited good sensitivity, with a lower limit of quantitation (LLOQ) of 1.25 μg/mL. Matrix effects were observed but were manageable, with no significant alterations to the sample clean-up process. Recovery rates were higher in water solutions compared to human plasma, indicating potential interference from protein precipitates. Intra-day and inter-day accuracy and precision met acceptance criteria. Plasma concentrations of D-mannose were found to be higher in patients with type 2 diabetes compared to healthy volunteers, aligning with existing literature.
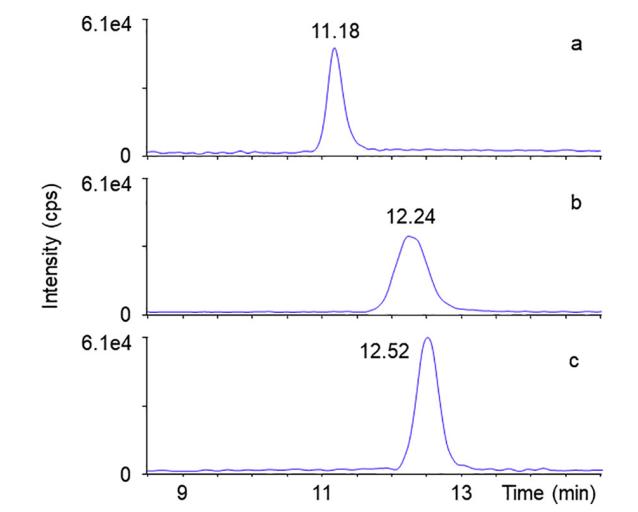 SRM chromatograms of m/z 178.9 → 88.9 Da from standard solutions containing mannose (a), galactose (b), and glucose (c) at a concentration of 1 μg/mL.
SRM chromatograms of m/z 178.9 → 88.9 Da from standard solutions containing mannose (a), galactose (b), and glucose (c) at a concentration of 1 μg/mL.
Reference
- Campi, Beatrice, et al. "Quantification of d-mannose in plasma: development and validation of a reliable and accurate HPLC-MS-MS method." Clinica Chimica Acta 493 (2019): 31-35.
What is the function of the mannose?
Cellular Energy Production: Mannose is a hexose sugar that can be metabolized to produce energy through cellular respiration. It enters the glycolytic pathway, where it is converted into intermediates that ultimately yield ATP, the primary energy currency of cells.
Glycosylation: Mannose is a component of glycoproteins and glycolipids, where it participates in glycosylation processes. Glycosylation involves the attachment of sugar molecules, including mannose, to proteins or lipids, which plays crucial roles in protein folding, stability, localization, and cell-cell recognition.
Cell-Cell Recognition: Mannose residues on the surface of cells or proteins can act as recognition signals for cell-cell interactions and adhesion. This function is particularly important in processes such as immune response, where mannose-binding proteins (lectins) recognize and bind to mannose residues on pathogens, facilitating their clearance by the immune system.
Mannose Salvage Pathway: In certain tissues, such as the brain, mannose can be utilized via the mannose salvage pathway to produce mannose-6-phosphate. Mannose-6-phosphate is an essential precursor for the synthesis of glycoproteins and glycolipids involved in maintaining cellular structure and function.
Antioxidant Properties: Mannose has been reported to possess antioxidant properties, helping to neutralize harmful free radicals and protect cells from oxidative damage. This antioxidant activity may contribute to its potential health benefits.
What is the difference between D-glucose and mannose?
D-glucose and mannose are both monosaccharides, meaning they are simple sugars consisting of a single sugar molecule. However, they differ in their structural configuration and some functional properties. Here are the main differences between D-glucose and mannose:
1. Structural Isomers: D-glucose and mannose are structural isomers of each other, meaning they have the same chemical formula (C6H12O6) but differ in the arrangement of atoms. Specifically, they differ in the spatial arrangement of hydroxyl (OH) groups around their carbon atoms.
2. Stereochemistry: Both D-glucose and mannose are chiral molecules, meaning they have asymmetric carbon atoms. However, they have different configurations at the carbon atom adjacent to the aldehyde or ketone group (the anomeric carbon). In D-glucose, the hydroxyl group (-OH) attached to the anomeric carbon is in the axial position in the D-configuration, while in mannose, it is in the equatorial position.
3. Biological Occurrence: D-glucose is the most abundant monosaccharide in nature and serves as a primary source of energy in cellular metabolism. It is found in various foods such as fruits, vegetables, and grains. Mannose, on the other hand, is less abundant in nature but is still present in certain foods and is an essential component of glycoproteins, glycolipids, and other complex carbohydrates.
4. Metabolic Pathways: While both D-glucose and mannose can be metabolized through glycolysis to produce energy, they enter the pathway at different points. D-glucose is metabolized directly through glycolysis, while mannose can be converted to mannose-6-phosphate via the mannose salvage pathway before entering glycolysis. Mannose-6-phosphate can then be used for glycosylation reactions or further metabolized for energy production.
5. Biological Functions: D-glucose serves as a primary source of energy for cells and is involved in various metabolic processes, including cellular respiration, glycogen synthesis, and glycosylation. Mannose is primarily involved in glycosylation processes, where it serves as a precursor for the synthesis of glycoproteins and glycolipids. Additionally, mannose has been reported to have potential therapeutic effects in certain conditions, such as urinary tract infections, due to its ability to inhibit bacterial adhesion to cell surfaces.
What is the difference between mannose and glucose in organic chemistry?
| Characteristic | Mannose | Glucose |
|---|---|---|
| Chemical Formula | C6H12O6 | C6H12O6 |
| Structural Isomer | Yes | Yes |
| Stereochemistry | Equatorial hydroxyl at anomeric carbon | Axial or equatorial hydroxyl at anomeric carbon (α or β configuration) |
| Ring Structure | Typically forms pyranose ring | Can form both pyranose and furanose rings |
| Abundance | Less abundant | Most abundant |
| Biological Occurrence | Present in certain foods; essential component of glycoproteins and glycolipids | Most abundant monosaccharide in nature; primary source of energy in cellular metabolism |
| Metabolic Pathway | Converted to mannose-6-phosphate via mannose salvage pathway before entering glycolysis | Metabolized directly through glycolysis |




