- Service Details
- Case Study
What is Glycan Acetylation Modification?
Glycan acetylation modification refers to the process of adding acetyl groups to glycans, which are complex carbohydrates composed of sugar molecules linked together. Acetylation is a common post-translational modification that involves the addition of acetyl groups to proteins or, in this case, to glycans.
In the context of glycans, acetylation can occur on specific sugar residues within the glycan structure. This modification can have functional implications, influencing the properties and interactions of glycans. It is a dynamic process that plays a role in various cellular functions, including cell signaling, recognition, and adhesion.
The addition of acetyl groups to glycans is catalyzed by enzymes known as acetyltransferases. This modification can affect the overall charge, structure, and function of glycans, thereby influencing their biological activities. Glycan acetylation is an essential aspect of glycosylation, a complex and highly regulated process that plays a crucial role in various physiological and pathological processes within cells and organisms.
Technical Platform for Glycan Acetylation Analysis
Sample Preparation: Our expert team handles the extraction of glycans from your provided samples, typically proteins or lipids. Through enzymatic or chemical release methods and careful derivatization, we ensure optimal stability and solubility of the glycans.
Liquid Chromatography (LC): Utilizing state-of-the-art LC techniques, we separate released and derivatized glycans based on their hydrophobic or hydrophilic properties. Specialized high-resolution columns enhance the separation and resolution for detailed analysis.
Mass Spectrometry (MS): Our advanced MS platform, incorporating techniques such as FT-MS and TOF-MS, accurately determines glycan masses. Tandem MS (MS/MS) provides valuable fragmentation analysis, revealing structural details and acetylation patterns.
Data Analysis and Interpretation: Our skilled analysts process the data using cutting-edge glycomics software. Database searches match experimental data with known glycan structures, and quantification is performed to determine the abundance of glycan species and their acetylation modifications.
Structural Elucidation and Visualization: We interpret the data to assign glycan structures, creating visual representations of glycan structures and acetylation patterns using specialized tools.
Quality Control and Validation: Rigorous quality control measures are implemented throughout the process to ensure the reliability of results. Replicate analyses and standardization contribute to the validation of the outcomes.
Applications of Glycan Acetylation Modification Analysis
Biomedical Research: Uncover how glycan acetylation influences protein functionality and impacts disease mechanisms, providing insights for therapeutic development.
Therapeutic Development: Target specific glycan acetylation pathways for therapeutic interventions, assessing drug efficacy and potential side effects.
Immunology and Immune Response: Investigate the role of glycan acetylation in modulating immune responses, with implications for immunotherapies and vaccine development.
Cell Signaling and Communication: Explore the influence of glycan acetylation on cell-cell interactions and intracellular signaling pathways.
Biopharmaceutical Production: Ensure the quality and consistency of glycoprotein-based biopharmaceuticals through glycan acetylation analysis.
Neuroscience: Study the involvement of glycan acetylation in neurological processes and disorders, contributing to understanding neuronal function.
Developmental Biology: Investigate glycan acetylation in embryonic development, providing insights into its role in differentiation processes.
Agricultural Biotechnology: Explore the impact of glycan acetylation on plant cell walls and plant-microbe interactions in agricultural settings.
Environmental Sciences: Study glycan acetylation in microbial communities, contributing to insights into microbial interactions and environmental processes.
Food Science: Analyze glycan acetylation patterns in food products for insights into nutritional content, food quality, and health implications.
Materials Science: Explore the use of glycan acetylation in biopolymer engineering for applications in biomaterials and tissue engineering.
Biotechnology Process Optimization: Optimize glycan acetylation patterns in biotechnological processes for improved production of glycoprotein-based therapeutics.
Drug Metabolism Studies: Investigate the metabolism of glycan-modified drugs for insights into drug clearance, bioavailability, and pharmacokinetic interactions.
Regenerative Medicine: Explore the role of glycan acetylation in stem cell differentiation and regenerative processes, advancing regenerative medicine.
Sample Requirements for Glycan Acetylation Modification Analysis
| Sample Type | Recommended Sample Size |
|---|---|
| Serum/Plasma | 100 μL - 500 μL |
| Tissue Biopsy | 5 mg - 20 mg |
| Cell Culture Supernatant | 1 mL - 5 mL |
| Urine | 0.5 mL - 2 mL |
| Cerebrospinal Fluid | 0.2 mL - 1 mL |
| Saliva | 0.5 mL - 2 mL |
| Synovial Fluid | 0.5 mL - 2 mL |
| Feces | 50 mg - 200 mg |
Case. O-Acetylation Impacts Neisseria gonorrhoeae Glycan Chain Length: Insights from Top-Down Mass Spectrometry Analysis
Background:
O-Acetylation of bacterial surface glycoconjugates, such as capsular polysaccharides (CPS) and O-specific polysaccharides of lipopolysaccharides (LPS), plays a crucial role in glycan structure and immunochemistry. This study focuses on elucidating the O-acetylation patterns in Neisseria gonorrhoeae (Ng) glycoconjugates, specifically in the context of pgl systems, which are involved in glycan biosynthesis.
Samples:
Bacterial strains of Ng, detailed in Table I, were cultivated on conventional GC medium. The investigation included strains with various pgl mutations (pglA, pglEon, pglI, pglH, pglH2, pglB2), allowing for the analysis of O-acetylation patterns in different glycoforms.
Technical Method:
- Acquisition of Bacterial Strains: Ng strains with specific pgl mutations were obtained, and antibiotic selection was employed during the transformation process.
- Purification of PilE Protein: The PilE protein, a key player in surface glycoconjugates, was purified from the bacterial strains.
- Top-Down ESI-MS Analysis: The intact PilE protein was subjected to top-down electrospray ionization mass spectrometry (ESI-MS) on the LTQ Orbitrap. This high-resolution technique allowed for detailed analysis of glycan modifications.
- Data Interpretation: Deconvoluted protein masses were determined and reported, considering both unmodified and modified proteins. The study particularly focused on post-translational modifications (PTMs) such as O-acetylation.
- Genetic Interactions Investigation: Genetic interactions between pglA, pglH2, and pglI were explored, revealing the impact of O-acetylation on oligosaccharide chain length.
Results:
Confirmation of O-Acetylation: Clear evidence was provided for O-acetylation of Gal, Glc, and GlcNAc residues in both diNAcBac- and GATDH-based disaccharides.
Unrecognized O-Acetylation Levels: Previously unrecognized O-acetylation of the distal Gal residue in trisaccharides was identified, with hierarchical patterns observed in glycoform modification.
Genetic Interactions: Strains co-expressing PglA, PglH2, and PglI exhibited mixtures of disaccharide forms with O-acetylated Gal and O-acetylated GlcNAc, impacting oligosaccharide chain length.
Trisaccharide Modification: A unique trisaccharide glycoform was detected in a pglI null background, revealing altered oligosaccharide chain length associated with PglI expression.
Potential Mechanisms: Two possible scenarios were proposed to explain the impact of O-acetylation on glycan biosynthesis, involving substrate synthesis or inhibition of glycosyltransferase activity.
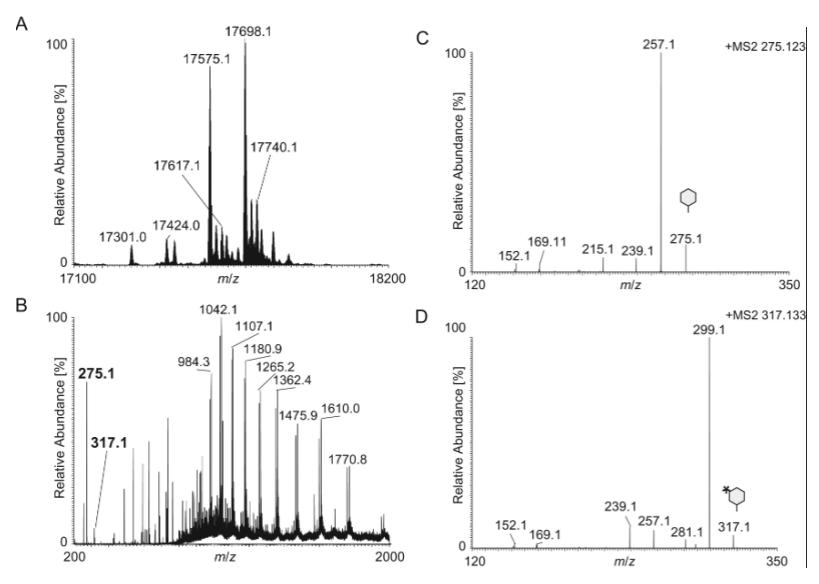 Acetylation of the GATDH.
Acetylation of the GATDH.
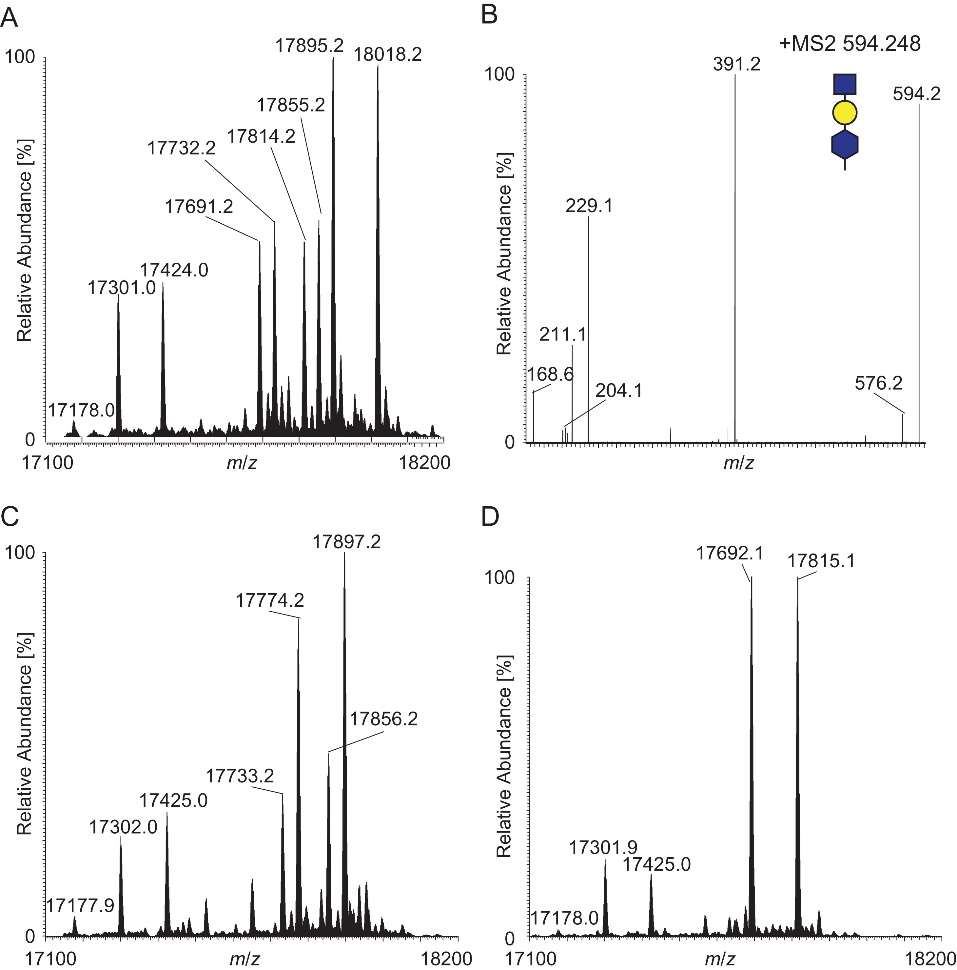 Acetylation determines glycan length.
Acetylation determines glycan length.
Reference
- Anonsen, Jan Haug, et al. "Structural and genetic analyses of glycan O-acetylation in a bacterial protein glycosylation system: evidence for differential effects on glycan chain length." Glycobiology 27.9 (2017): 888-899.




