- Service Details
- Case Study
The Essence of Sialic Acid Analysis
Sialic acid, a monosaccharide with a negative charge, plays a pivotal role in the structural integrity and functionality of glycoproteins. Positioned at the terminal ends of polysaccharides, sialic acid exerts significant influence on the stability and conformational dynamics of these biomolecules. Moreover, it actively participates in a myriad of biological interactions, thereby contributing to essential physiological processes.
The functional implications of sialic acid are multifaceted. For instance, N-linked polysialic acids have been shown to enhance the anti-inflammatory activity of immunoglobulin G (IgG), demonstrating the profound impact of sialylation patterns on protein function. Additionally, O-acetylated sialic acid residues can modulate ligand-receptor interactions, thereby influencing processes such as protein degradation. Notably, the presence of sialic acid residues on glycoproteins can prolong their circulating half-life by impeding their recognition and uptake by hepatic asialoglycoprotein receptors.
In the realm of biopharmaceuticals, sialic acid assumes critical importance, as its composition and abundance can significantly impact drug safety and efficacy. Two primary sialic acid species encountered in biopharmaceuticals are N-acetylneuraminic acid (Neu5Ac or NANA) and N-glycolylneuraminic acid (Neu5Gc or NAGA). While Neu5Ac is commonly found in human-derived products, Neu5Gc, which humans cannot synthesize, poses immunogenic risks and may trigger adverse immune responses. Monitoring sialic acid levels and types throughout the lifecycle of a biopharmaceutical product is imperative to ensure its safety and efficacy. Sialic acid analysis, therefore, transcends mere detection, encompassing the identification and quantification of specific sialic acid species. This comprehensive approach is indispensable across various stages of drug development, from preliminary investigations to batch-to-batch consistency assessments. A thorough understanding of the sialic acid composition in biopharmaceutical products enables researchers to mitigate risks associated with immunogenicity and optimize therapeutic outcomes.
Creative Proteomics provides comprehensive sialic acid analysis. Our sialic acid analysis service involves multiple steps including extraction, purification, labeling, separation, detection, and data interpretation. The analysis helps in determining the overall degree of sialylation, including the absolute quantitation of sialic acid residues per molecule, relative quantities of Neu5Ac and Neu5Gc, and relative percentage of O-acetylated sialic acids.
Our sialic acid analysis can also provide insights about the structural characterization of sialylated glycans that reflect the original ions, along with the detection of their molecular structure, aiding in confirming biopharmaceutical characters in compliance with the regulatory requirement (ICH Q6B).
Technology Platform for Sialic Acid Analysis
Chromatography: Chromatographic separation techniques, including high-performance liquid chromatography (HPLC) and capillary electrophoresis (CE), serve as the foundation for sialic acid analysis. These methods facilitate the separation of complex mixtures based on differences in their physicochemical properties, such as polarity and charge. In particular, HPLC with various modes, such as reversed-phase, ion-exchange, and hydrophilic interaction chromatography (HILIC), allows for the efficient separation and detection of sialic acid species.
Mass Spectrometry (MS): Coupling chromatography with mass spectrometry enhances the specificity and sensitivity of sialic acid analysis. MS-based techniques, such as electrospray ionization (ESI) and matrix-assisted laser desorption/ionization (MALDI), enable the accurate determination of molecular weights and structural elucidation of sialylated glycans. Additionally, tandem mass spectrometry (MS/MS) facilitates the characterization of sialic acid linkages and the identification of specific sialylation patterns.
High Performance Anion Exchange Chromatography with Pulsed Amperometric Detection (HPAEC-PAD): HPAEC-PAD is a specialized chromatographic technique widely employed for sialic acid analysis. This method enables the separation and quantification of sialic acid species based on their different chemical properties, particularly their acidic nature. The pulsed amperometric detection system offers high sensitivity and selectivity for detecting sialic acid residues, making it well-suited for pharmaceutical applications.
Linkage-Specific Derivatization Techniques: To distinguish between various sialic acid linkages, linkage-specific derivatization techniques are employed. These methods involve chemical modifications that selectively target specific sialic acid residues, thereby enabling differentiation between N-acetylneuraminic acid (Neu5Ac) and N-glycolylneuraminic acid (Neu5Gc), among others. By incorporating various condensing reagents and nucleophiles, researchers can achieve precise discrimination of sialyl linkages and enhance the specificity of sialic acid analysis.
Sample Requirements for Sialic Acid Analysis
| Sample Type | Sample Volume |
|---|---|
| Glycoproteins | 50-100 μg |
| Biopharmaceuticals | 100-500 μg |
| Serum/Plasma | 100-200 μL |
| Cell Culture Supernatant | 100-500 μL |
| Tissue Extracts | 10-50 mg |
| Cell Lysates | 10^6 - 10^7 cells |
| Cerebrospinal Fluid | 50-100 μL |
| Urine | 100-500 μL |
Applications of Sialic Acid Analysis
Biopharmaceutical Development:
- Quality Control: Sialic acid analysis is crucial for ensuring the consistency and quality of biopharmaceutical products, particularly glycoproteins such as monoclonal antibodies (mAbs) and recombinant proteins. By monitoring sialic acid levels and species, manufacturers can verify batch-to-batch consistency and adherence to regulatory standards.
- Process Optimization: Sialic acid analysis helps optimize bioprocess parameters to enhance the production yield and quality of glycosylated biologics. By understanding how cell culture conditions and purification methods affect sialic acid profiles, researchers can improve product stability, efficacy, and safety.
- Comparative Studies: Comparative sialic acid analysis enables researchers to evaluate glycosylation differences between biosimilar and innovator biopharmaceuticals. This comparative assessment is essential for demonstrating biosimilarity and supporting regulatory approval.
Biomedical Research:
- Glycoprotein Characterization: Sialic acid analysis provides valuable insights into the glycosylation patterns of proteins, allowing researchers to characterize glycoproteins and understand their structure-function relationships. This information is particularly relevant in studying protein folding, stability, receptor-ligand interactions, and immune modulation.
- Cellular Physiology: Investigating sialic acid dynamics in cell cultures, tissues, and model organisms elucidates their roles in cellular physiology, development, and disease pathology. Manipulating sialic acid metabolism and expression levels can uncover novel therapeutic targets and strategies for treating various disorders.
Food and Nutrition:
- Quality Assessment: Sialic acid analysis is utilized in food and nutritional science to assess the quality and authenticity of food products. Monitoring sialic acid levels in dairy products, meat, seafood, and infant formula helps ensure product safety, nutritional value, and compliance with regulatory standards.
Case. Quantification and Distribution of Sialic Acid Species in Pig Tissues
Background:
Understanding the distribution and levels of sialic acid (Sia) species in pig tissues is crucial for assessing potential health risks associated with dietary intake. Sia, including Neu5Ac, Neu5Gc, and Kdn, plays various roles in cellular processes and has been implicated in inflammatory diseases and cancer.
Samples:
Samples were obtained from healthy pigs of different ages (3 days, 38 days, and 180 days) from a commercial piggery in Xiamen City, China. Tissues including spleen, kidney, lung, heart, liver, and skeletal muscle were collected and stored at -80°C for analysis.
Technical Method:
Sample Preparation: Tissues were isolated and homogenized, with both uncooked and boiled samples prepared for analysis.
LC-MS/MS Analysis: Total levels of Neu5Ac, Neu5Gc, and Kdn were quantified in pig tissues using liquid chromatography-tandem mass spectrometry (LC-MS/MS) in negative electrospray ionization mode. Internal standards were used for accurate quantification.
Protein Determination: Protein concentrations in tissue homogenates were measured using the Pierce BCA protein assay kit to normalize Sia levels.
Statistical Analysis: Mann-Whitney U test and Kruskal-Wallis one-way ANOVA were employed to compare Sia concentrations between different organs and age groups, with significance set at P < 0.05.
Results:
Age-Related Changes: Total Sia levels were highest in young piglets and decreased with age in all organs except the liver at 38 days of age. Skeletal muscle exhibited the lowest Sia levels across all age groups.
Neu5Gc Distribution: Neu5Gc predominated in spleen across all ages, while Neu5Ac was the main Sia species in other organs. Skeletal muscle had the lowest Neu5Gc concentration.
Cooking Effects: Cooking increased Sia levels in all tissues except the heart. Neu5Gc proportions remained consistent except in skeletal muscle, where it increased significantly after cooking.
Health Implications: Higher Neu5Gc levels in organ tissues compared to skeletal muscle suggest potential health risks associated with organ consumption, emphasizing the importance of dietary choices.
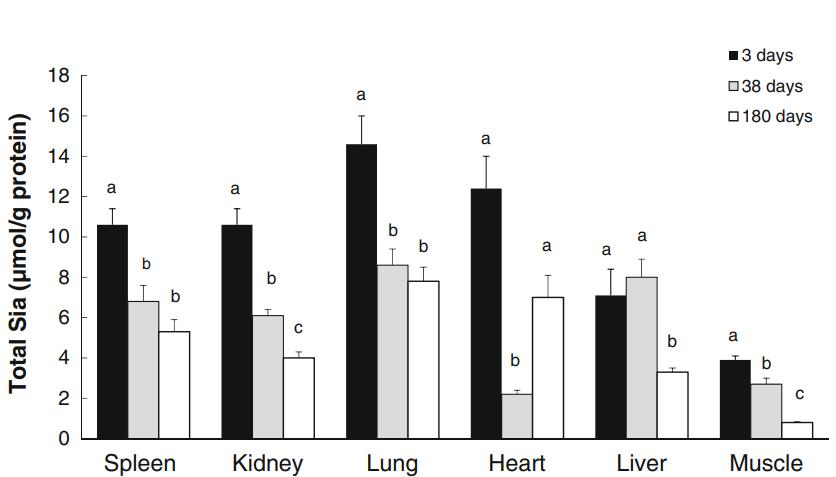 Comparison of the total concentration of Neu5Ac, Neu5Gc and Kdn in spleen, kidney, lung, heart, liver, and skeletal muscle in pigs at 3, 38 and 180 days of age (adult).
Comparison of the total concentration of Neu5Ac, Neu5Gc and Kdn in spleen, kidney, lung, heart, liver, and skeletal muscle in pigs at 3, 38 and 180 days of age (adult).
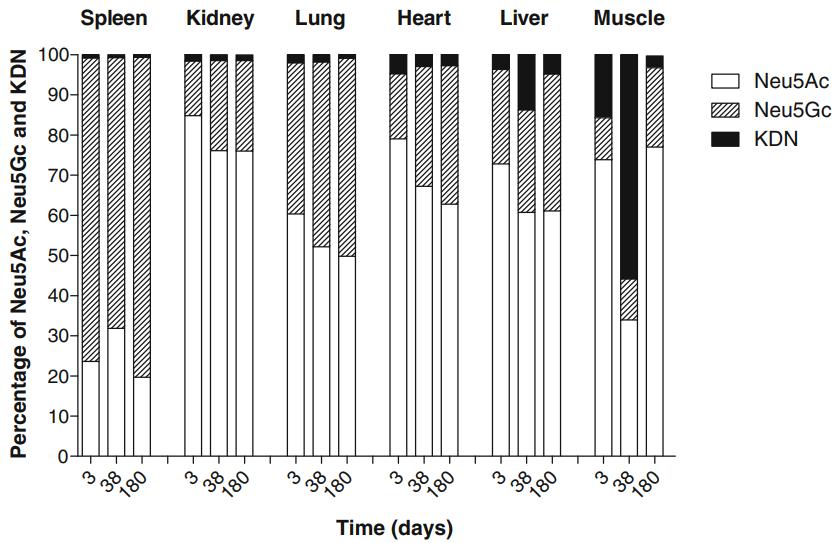 Summary analysis of the percent distribution of the total levels of Neu5Gc, Neu5Ac and Kdn in spleen, kidney, lung, heart, liver and skeletal muscle in pigs at 3, 38, and 180 days of age (adult).
Summary analysis of the percent distribution of the total levels of Neu5Gc, Neu5Ac and Kdn in spleen, kidney, lung, heart, liver and skeletal muscle in pigs at 3, 38, and 180 days of age (adult).
Reference
- Ji, Suna, et al. "Developmental changes in the level of free and conjugated sialic acids, Neu5Ac, Neu5Gc and KDN in different organs of pig: a LC-MS/MS quantitative analyses." Glycoconjugate journal 34 (2017): 21-30.




