- Service Details
- Case Study
What is Glycoprotein Quantification?
Glycoprotein quantification is a sophisticated process that plays a crucial part in proteomics research. Glycoproteins, proteins modified by the addition of carbohydrate or sugar moieties, are pivotal for various biological processes such as cell-cell communication, immune response, and pathogenesis of diseases. Glycoprotein quantification involves the measurement of actual glycoprotein concentration in a biological sample, providing insights into potential changes in key cellular processes.
Quantitative glycoproteomics techniques aim to evaluate glycoproteins in a highly accurate and comprehensive manner. This high-throughput approach improves research outcomes in biomarker discovery, therapeutic target validation, and clinical diagnostics.
Creative Proteomics offers a wide range of projects for glycoprotein quantification analysis, tailored to meet the diverse needs of researchers. Our services include:
- Relative quantification of glycoproteins: Using isotope labeling or label-free techniques combined with advanced mass spectrometry, we provide precise measurement of changes in glycoprotein abundance under different experimental conditions.
- Absolute quantification of glycoproteins: Leveraging our expertise in glycoprotein analysis, we offer absolute quantification methods based on the unique characteristics of glycoproteins, enabling accurate comparisons across samples and experimental conditions.
Technology Platform for Glycoprotein Quantification
Creative Proteomics offers leading-edge technical platforms for glycoprotein quantification analysis, ensuring precise, reliable, and comprehensive analytics for researchers worldwide.
Relative Quantification of Glycoproteins
Our relative quantification projects utilize a variety of techniques, including isotope labeling and label-free approaches, combined with state-of-the-art mass spectrometry platforms. These projects are ideal for researchers seeking to quantify changes in glycoprotein abundance across different experimental conditions or biological samples. By accurately measuring relative changes in glycoprotein levels, researchers can gain valuable insights into biological pathways, disease mechanisms, and drug responses.
Absolute Quantification of Glycoproteins
Creative Proteomics offers absolute quantification projects tailored to provide precise measurements of glycoprotein concentrations in biological samples. Leveraging our expertise in glycoprotein analysis and quantitative proteomics, we develop customized assays and calibration curves to quantify target glycoproteins with high accuracy and sensitivity.
Glycoprotein Profiling and Characterization
In addition to quantification, Creative Proteomics offers projects focused on comprehensive profiling and characterization of glycoproteins. These projects utilize advanced mass spectrometry techniques, lectin microarray analysis, and glycan profiling to identify glycosylation sites, characterize glycan structures, and elucidate glycoprotein heterogeneity. By providing detailed information on glycoprotein composition and structure, these projects facilitate a deeper understanding of glycosylation dynamics and its functional implications in health and disease.
Customized Analytical Solutions
We offer customized analytical solutions to meet the specific needs of our clients. Whether you require targeted quantitative analysis of specific glycoproteins, comparative analysis of multiple samples, or comprehensive glycoprotein characterization, our team of experienced scientists will work closely with you to design and execute a custom project that meets your research goals.
Sample Requirements for Glycoprotein Quantification
| Sample Type | Sample Volume | Description |
|---|---|---|
| Serum/Plasma | 100-500 µL | Blood samples collected in EDTA or heparin tubes. Centrifuge to separate serum or plasma before storage at -80°C. |
| Cell Culture | 1x106 cells | Adherent or suspension cells grown to desired confluence. Harvest cells using appropriate methods such as trypsinization or scraping. |
| Tissue Homogenate | 10-50 mg | Fresh or frozen tissue samples homogenized in lysis buffer containing protease inhibitors. |
| Biopsy Specimen | 2-5 mm3 | Tissue samples obtained via biopsy procedures using sterile instruments. Immediately immerse in appropriate storage medium or snap-freeze in liquid nitrogen. |
| Urine | 10-100 mL | Collect fresh urine samples in sterile containers. Centrifuge to remove cellular debris before storage at -80°C. |
| Saliva | 1-2 mL | Collect saliva samples using non-invasive methods such as saliva collection kits or oral swabs. Store at -80°C to preserve glycoprotein integrity. |
| Cerebrospinal Fluid | 0.5-1 mL | Collect CSF samples via lumbar puncture or other sterile procedures. Handle with care to avoid contamination. Store at -80°C. |
| Synovial Fluid | 100-500 µL | Aspirate synovial fluid from affected joints using sterile syringes. Centrifuge to remove cells and debris before storage at -80°C. |
| Tumor Tissue | 10-50 mg | Obtain tumor tissue samples from biopsies or surgical resections. Immediately snap-freeze in liquid nitrogen or store in RNAlater solution. |
| Fecal Samples | 1-5 g | Collect fresh fecal samples using sterile containers. Store at -80°C to preserve glycoprotein integrity. |
Applications of Glycoprotein Quantification
Biomarker Discovery: Identify and validate glycoprotein biomarkers for disease detection in cancer, cardiovascular diseases, and infectious diseases.
Disease Mechanism Elucidation: Investigate aberrant glycosylation patterns associated with disease pathogenesis to unravel molecular mechanisms underlying conditions such as neurodegenerative disorders, autoimmune diseases, and metabolic syndromes.
Pharmacokinetics and Drug Development: Evaluate pharmacokinetic profiles of therapeutic glycoproteins, including monoclonal antibodies, vaccines, and protein-based drugs, to optimize dosing regimens and improve therapeutic outcomes.
Personalized Medicine: Implement personalized medicine approaches by leveraging glycoprotein quantification to tailor diagnostic and therapeutic interventions based on individual patient characteristics and treatment responses.
Immunotherapy and Cancer Research: Characterize glycoprotein profiles in tumor microenvironments to guide the development of immunotherapies and targeted therapies for cancer treatment.
Infectious Disease Research: Study host-pathogen interactions and immune responses by quantifying glycoprotein expression and glycosylation changes in infectious diseases such as HIV/AIDS, influenza, and COVID-19.
Neurobiology and Neurodegenerative Disorders: Investigate the role of glycoproteins in neuronal development, synaptic function, and neurodegenerative diseases such as Alzheimer's and Parkinson's disease.
Glycoengineering and Biotechnology: Engineer glycoproteins with desired glycosylation patterns for biopharmaceutical production, glycan array development, and functional studies in biotechnology and drug discovery.
Food and Environmental Sciences: Analyze glycoprotein composition in food products and environmental samples to assess quality, safety, and potential allergenicity.
Case. Comprehensive Profiling of Cell Surface Glycoproteins Using CSC Technology
Background:
Characterizing the expression patterns of cell surface proteins is crucial for understanding cellular function and disease mechanisms. Traditional methods relying on antibodies face limitations due to the lack of specific reagents and the complexity of multiplexed assays. The Cell Surface Capture (CSC) technology offers a promising alternative by selectively targeting cell surface glycoproteins, which are commonly expressed across various cell types.
Samples:
The study utilized different cell types, including Jurkat T cells, Ramos B cells, Drosophila Kc167 cells, and embryonic stem (ES) cells. These diverse cell types allowed for the investigation of cell surface glycoproteins across different organisms and cell states.
Technical Method:
Harvesting and Oxidation: Cells were collected and treated with sodium-meta-periodate to oxidize cell surface glycoproteins.
Cell Surface Labeling: Biocytin hydrazide was used to selectively label glycoproteins on the cell surface.
Cell Lysis and Membrane Preparation: Cells were lysed, and the membrane fraction was isolated using detergent-free, hypotonic lysis buffer followed by ultracentrifugation.
Digestion of Membrane Preparation: Proteins in the membrane fraction were digested with LysC and trypsin.
Glycopeptide Capture: Glycopeptides were captured using Streptavidin Plus beads, followed by enzymatic release with PNGaseF.
LC-MS and Database Analysis: Released glycopeptides were analyzed by nanospray LC-MS/MS, and database searches were performed for protein identification. Statistical analysis and validation were conducted using bioinformatic tools.
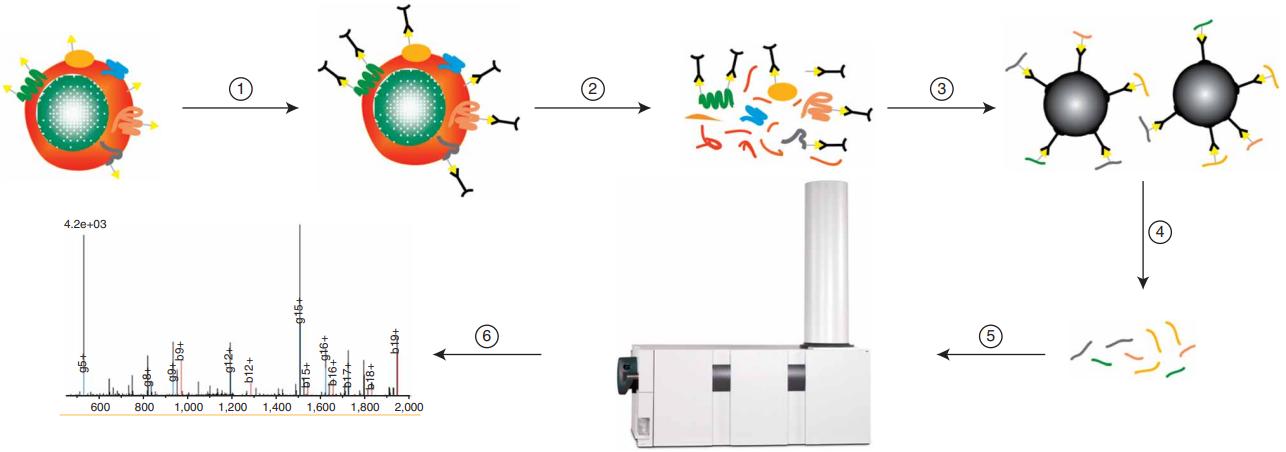 CSC uses a multistep tandem affinity labeling strategy to confer the desired specificity for the glycoproteins on the cell surface
CSC uses a multistep tandem affinity labeling strategy to confer the desired specificity for the glycoproteins on the cell surface
Results:
The CSC technology enabled comprehensive profiling of cell surface glycoproteins with high specificity and sensitivity.
Various cell types were analyzed, revealing a diverse landscape of cell surface proteins.
Confocal imaging and flow cytometry confirmed the efficiency and specificity of cell surface glycoprotein labeling.
The method provided insights into cell surface protein expression patterns, offering potential applications in disease research and therapeutic target discovery.
CSC technology holds promise for future studies aiming to characterize cell surface protein barcodes and their role in cellular processes and disease mechanisms.
Reference
- Wollscheid, Bernd, et al. "Mass-spectrometric identification and relative quantification of N-linked cell surface glycoproteins." Nature biotechnology 27.4 (2009): 378-386.




