- Service Details
- Case Study
What is Glycan Branching?
Glycan branching refers to the structural arrangement in which complex carbohydrates, known as glycans, exhibit branched configurations. Glycans are composed of sugar molecules linked together through glycosidic bonds. The branching of glycans occurs when additional sugar units are attached to the main chain, forming side branches.
The process of glycan branching is crucial in the biosynthesis of glycoproteins and glycolipids. Glycoproteins are proteins that have covalently attached glycans, while glycolipids are lipids with attached glycans. Glycans play essential roles in various biological processes, including cell signaling, recognition, and adhesion.
The complexity of glycan branching contributes to the diversity of glycan structures found in nature. Different types of sugar residues and linkages can lead to a wide variety of branched configurations. The specific arrangement of glycans on glycoproteins or glycolipids is often critical for their proper functioning.
The study of glycan branching allows for an understanding of its impact on cellular processes and sheds light on the role of glycans in health and disease. The diversity of glycan structures, including branching patterns, affects their interactions with other biomolecules and has implications for disease mechanisms, immune responses, and other physiological functions.
Technology Platform for Glycan Branching Analysis
Creative Proteomics provides a technical platform for glycan branching analysis. We rely on advanced mass spectrometry techniques, especially multi-stage mass spectrometry (MSn), to fully understand the complex structure and branching patterns of glycans. Mass spectrometry (MS) involves two main stages: MS1 (first-stage mass spectrometry) and MS2 (second-stage mass spectrometry). Traditional MS, while valuable, may not offer sufficient information to elucidate intricate glycan branching patterns.
In MSn, glycans undergo consecutive fragmentation at multiple levels, allowing for enhanced information about their structures. This method is particularly effective in determining the exact branching patterns of glycans, providing a more detailed view compared to traditional MS.
The workflow for glycan branching analysis includes sample preparation, mass spectrometry analysis (e.g., MALDI-MS with a QIT-TOF configuration), data interpretation, and analysis outputs. Samples, such as permethylated oligosaccharide standards or N-glycans from glycoproteins, undergo specific preparation methods to facilitate analysis. The obtained MSn spectra are then subjected to data interpretation to identify isotopic peaks and understand the glycan branching patterns.
The advantages of using MSn in glycan branching analysis include isomeric discrimination, allowing researchers to distinguish between different forms of glycans, and the provision of detailed structural information. By generating multiple-level fragments, MSn facilitates a thorough elucidation of the complex branching patterns of glycans.
Application of Glycan Branching Analysis
Identification of Glycan Structures: The primary application is the identification of glycan structures, specifically focusing on elucidating branching patterns. Understanding the exact arrangement of sugar units in branched structures provides insights into the diversity of glycan structures, crucial for unraveling their functional significance.
Discrimination of Isomeric Glycans: Glycan Branching Analysis is applied to discriminate between isomeric glycans, which have identical molecular masses but different branching configurations. This is essential for distinguishing subtle variations in glycan structures that may have distinct biological implications.
Insights into Disease Mechanisms: The analysis is employed to investigate the involvement of glycans in the pathogenesis and progression of diseases. It provides valuable information on altered glycan structures associated with conditions such as cancer and rheumatoid arthritis.
Biomarker Discovery for Clinical Diagnosis: Identification of glycans that serve as biomarkers for specific diseases is a key application. Altered glycan structures, detected through branching analysis, can be utilized for clinical diagnosis and prognosis, contributing to personalized medicine approaches.
Functional Roles in Biological Processes: The analysis helps in investigating the functional roles of glycans in various biological processes. Understanding how glycan branching patterns influence protein conformation, cell proliferation and differentiation, cell-cell communication, immune response, and microbial adhesion is crucial.
Human Milk Oligosaccharides (HMOs) Analysis: Glycan Branching Analysis is applied to study the structural diversity of HMOs in human milk. The branching patterns of HMOs influence the bacterial composition in the infant's intestinal tract, providing protection against pathogens and infections.
Sample Requirements for Glycan Branching Analysis
| Sample Type | Sample Size Recommendations | Sample Type | Sample Size Recommendations |
|---|---|---|---|
| Amniotic Fluid | 100 µL | Saliva | 50 µL |
| Cell Culture Media | 1 mL | Serum | 100 µL |
| Cerebrospinal Fluid | 100 µL | Skin Biopsy | 5 mg |
| Feces | 50 mg | Stool | 50 mg |
| Hair | 10 mg | Synovial Fluid | 150 µL |
| Lymphatic Fluid | 150 µL | Tears | 50 µL |
| Mucus | 100 µL | Tissue Homogenate | 20 mg |
| Nasal Swab | Swab in 1 mL transport medium | Urine | 200 µL |
| Plasma | 150 µL | Whole Blood | 100 µL |
Case. Intelligent Analysis of N-Glycan Branching Patterns using GIPS-II and Mass Spectrometry
Background:
The study focuses on the development and application of a methodology called GIPS-II for the intelligent analysis of N-glycan branching patterns using mass spectrometry. N-glycans are complex carbohydrate structures attached to proteins, and their branching patterns play a crucial role in understanding glycoprotein structure and function.
Samples:
The samples analyzed in the study include various N-glycans listed in the CarbBank database. Specifically, high mannose N-glycans (e.g., Man-7D1 and Man-7D3), complex N-glycans (e.g., A2G2S2, A3, A4), and hybrid N-glycans were used as representatives for demonstrating the identification process.
Technical Method:
The GIPS-II methodology involves the following steps:
- Precursor Ion Selection: Identification of candidate glycans by comparing their molecular mass with the sample's primary mass spectrum.
- MSn Analysis: Iterative rounds of MSn analysis, where distinctive fragment ions are used to differentiate between candidate glycans.
- GIPS-II Concept: Introduction of the GIPS-II concept to address challenges in cases where distinctive fragments are lacking, including a significance test using the Bayes factor model.
- Implementation of GIPS-II: Application of GIPS-II involves a significance test to confirm hypotheses and a biased-DP-model for experimental verification in situations where candidate probabilities remain equal.
Results:
Analysis of Individual N-Glycans: GIPS-II successfully identified individual N-glycans, including challenging cases where conventional methods failed. The methodology outperformed the MIPS strategy in identifying representative N-glycans.
Application to Glycoproteins: GIPS-II was applied to glycoproteins RNase B, enabling N-glycan profiling and detailed identification. The results were corroborated with other methods and literature data.
N-Glycan Profiling of Human Plasma and Serum: GIPS-II was applied to the N-glycan profiling of glycoprotein alpha-1-acid glycoprotein (AGP) and human serum. The methodology successfully identified specific branching patterns and fucose residue locations.
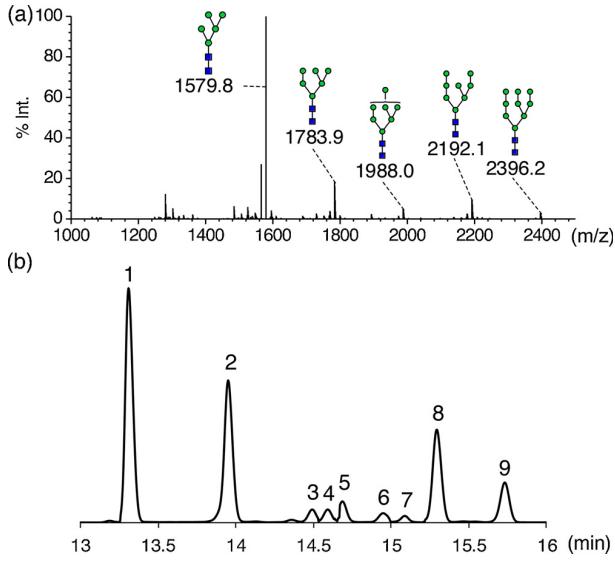 Profiling and subsequent assignment of N-glycans released from RNase B.
Profiling and subsequent assignment of N-glycans released from RNase B.
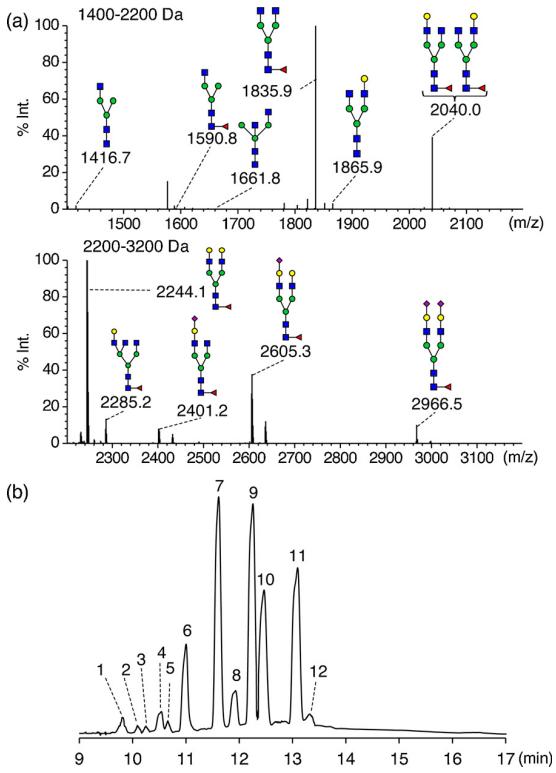 Profiling and subsequent assignment of N-glycans released from a monoclonal antibody (mAb).
Profiling and subsequent assignment of N-glycans released from a monoclonal antibody (mAb).
Reference
- Huang, Chuncui, et al. "Multistage mass spectrometry with intelligent precursor selection for N-glycan branching pattern analysis." Carbohydrate polymers 237 (2020): 116122.




