- Service Details
- Case Study
Why Choose Monosaccharide Composition Analysis?
Monosaccharides, often referred to as simple sugars, serve as the foundational units of carbohydrates, playing a pivotal role in biological systems. These small molecules are fundamental to the structure and function of complex carbohydrates, including glycoproteins and polysaccharides. In the realm of biochemistry and pharmaceuticals, understanding the composition of monosaccharides is paramount due to its far-reaching implications.
The need for monosaccharide analysis arises from the intricate nature of glycoproteins and carbohydrates, where the specific arrangement and types of monosaccharides determine their biological activities. Glycosylation, the process of attaching sugar molecules to proteins or lipids, significantly influences the stability, function, and immunogenicity of biotherapeutics. Therefore, a detailed analysis of monosaccharides becomes imperative in ensuring the quality, efficacy, and safety of glycoprotein-based drugs and other biopharmaceuticals.
Monosaccharide composition analysis provides a comprehensive understanding of the glycosylation patterns within a given sample. It allows researchers and pharmaceutical professionals to discern the types and quantities of monosaccharides present, identifying potential modifications such as sulfation and phosphorylation. By unraveling the intricate carbohydrate structures, scientists can gain insights into the glycan diversity, aiding in the development of therapeutic agents with enhanced properties.
Moreover, in drug development, monosaccharide analysis serves as a critical tool for characterizing and monitoring the glycosylation status of biopharmaceuticals. It aids in meeting stringent regulatory requirements by providing evidence of glycan composition and ensuring the consistency of glycosylation patterns across different batches. This analytical approach acts as a safeguard, mitigating risks associated with variations in glycosylation that could impact the safety and efficacy of therapeutic products.
Monosaccharide Profile in Creative Proteomics
- Qualitative and quantitative analysis of neutral sugars and amino sugars.
- Identification and quantification of specific monosaccharides in complex samples.
- Structural characterization of monosaccharides and their derivatives.
- Profiling of monosaccharide composition in biological samples.
- Investigation of monosaccharide metabolism and pathways.
- Biomarker discovery and validation studies related to monosaccharides.
- Analysis of monosaccharide interactions with other biomolecules.
- Customized experiments and consulting services tailored to specific research needs.
List of Detectable Monosaccharide
Aldoses:
| Monosaccharide | Chemical Formula |
|---|---|
| Glucose | C6H12O6 |
| Galactose | C6H12O6 |
| Mannose | C6H12O6 |
| Ribose | C5H10O5 |
| Arabinose | C5H10O5 |
| Xylose | C5H10O5 |
| Erythrose | C4H8O4 |
| Threose | C4H8O4 |
Ketoses:
| Monosaccharide | Chemical Formula |
|---|---|
| Fructose | C6H12O6 |
| Xylulose | C5H10O5 |
| Erythrulose | C4H8O4 |
| Dihydroxyacetone | C3H6O3 |
Amino Sugars:
| Monosaccharide | Chemical Formula |
|---|---|
| Glucosamine | C6H13NO5 |
| Galactosamine | C6H13NO5 |
| Mannosamine | C6H13NO5 |
| N-Acetylglucosamine | C8H15NO6 |
| N-Acetylgalactosamine | C8H15NO6 |
| N-Acetylneuraminic acid | C11H19NO9 |
Others:
| Monosaccharide | Chemical Formula |
|---|---|
| Fucose | C6H12O5 |
Technology Platform for Monosaccharide Composition Analysis
Creative Proteomics has established an advanced and efficient technical platform for monosaccharide composition analysis, employing state-of-the-art technologies to deliver accurate and insightful results.
High-Performance Anion-Exchange Chromatography with Pulsed Amperometric Detection (HPAE-PAD)
A key component of our technology platform is the HPAE-PAD, an advanced chromatographic technique specifically designed for glycoprotein glycosylation analysis. The HPAE-PAD separates carbohydrates based on their composition, size, linkage, charge and isomerism. It eliminates the need for derivatization of samples, thus reducing analysis time and costs. The released monosaccharides are acid-hydrolyzed and then analyzed by the HPAE-PAD, resulting in high-resolution results.
High-Performance Liquid Chromatography (HPLC)
HPLC, coupled with fluorescent labeling, ensures stable and accurate monosaccharide determination. Glycoprotein hydrolysates are labeled with 2-aminobenzoic acid (2AA) during mild acid hydrolysis, allowing for quantitative analysis by HPLC.
Mass Spectrometry (MS)
Our technical platform incorporates LC-ESI-MS and MALDI-TOF MS for powerful glycan analysis. MS enables accurate identification and quantification of monosaccharides, enhancing the precision of our analytical capabilities.
Capillary Electrophoresis (CE)
CE, suitable for both derivatized and underivatized carbohydrates, is employed for its brief analysis time, high sensitivity, and capacity to resolve isomeric glycans. Our team of technical experts is adept at addressing challenges in CE-based monosaccharide analysis.
Applications of Monosaccharide Composition Analysis
Biopharmaceuticals and Drug Development: Characterization and monitoring of glycoproteins, quality control in drug development, exploration of monosaccharide modifications for thorough glycosylation analysis.
Biopharmaceutical Manufacturing: Monitoring changes in glycosylation during manufacturing, ensuring consistency between batches for release, compliance with regulatory standards, including ICH Q6B requirements.
Quality Assurance and Batch Consistency: Identification of carbohydrate impurities, initial investigations before detailed glycosylation analysis, screening glycoproteins from different cell lines.
Food and Nutrition Sciences: Determination of sugar content in food products, contribution to nutritional labeling, research on dietary impacts and metabolic processes.
Environmental Sciences: Study of microbial activity and decomposition processes, understanding carbon cycling and soil health.
Material Science and Industry: Characterization of polysaccharide-based materials, tailoring properties of materials in various industries.
Cosmetics and Personal Care: Assessment of carbohydrate components in cosmetic formulations, ensuring quality and safety of cosmetic products.
Disease Biomarker Discovery: Identification of glycosylation aberrations as disease biomarkers, insights into disease mechanisms and diagnostic possibilities.
Research and Academic Studies: Contribution to studies in glycobiology and biochemistry, unraveling complexities of glycan structures for academic research.
Sample Requirements for Monosaccharide Composition Analysis
| Sample Type | Sample Size Recommendations |
|---|---|
| Biological Fluids | Minimum volume of 100 μL for effective results. |
| Tissue Samples | Several milligrams, accounting for tissue heterogeneity. |
| Cell Culture Supernatants | Minimum volume of 500 μL for reliable analysis. |
| Glycoproteins | Minimum of 1-5 μg for accurate analysis. |
| Carbohydrate Polymers | 5-10 mg, depending on the complexity. |
| Food Products | Variable, based on the specific product. |
| Environmental Samples | Typically several grams for meaningful data. |
| Polysaccharide Materials | 10-20 mg, considering material properties. |
Case. Development of an HILIC-CAD Method for Comprehensive Monosaccharide Profiling in Plant-Derived Oligo- and Polysaccharides
Background:
This study aimed to establish a highly efficient and convenient method for the analysis of monosaccharide compositions in plant-derived oligo- and polysaccharides. The focus was on developing a technique that offers simplicity, reliability, and the ability to simultaneously separate and determine common neutral monosaccharides.
Samples:
The samples used in this study included E. prostrasta L. polysaccharide (EAPW) and stachyose, which served as representatives of plant-derived oligo- and polysaccharides. EAPW was obtained from E. prostrasta L. pieces through extraction and further processing.
Technical Method:
- Materials and Reagents: E. prostrasta L. pieces were provided and authenticated. Various chemicals and solvents, including monosaccharide standards, were procured. The DEAE cellulose column, BSA, and water purification system were utilized.
- Preparation of Standard Solution: Standard solutions of monosaccharide mixtures were prepared in acetonitrile-water solvent. Sodium chloride and sodium sulfate standard solutions were also prepared.
- Chromatographic Analysis: Chromatographic experiments were conducted using an Ultimate 3000 HPLC system coupled with a Corona CAD Ultra. The BEH amide column was used with an isocratic elution of 22 mM ammonium acetate in 88% ACN. Chromatographic conditions were optimized for retention and separation.
- Validation Study: The analytical methodology was validated for linearity, limit of detection (LOD), limit of quantification (LOQ), accuracy, and precision. Calibration curves were established, LOD and LOQ were determined, and precision was evaluated through intra- and inter-day variations. Accuracy was assessed using spike recovery tests.
- Sample Preparation and Analysis: E. prostrasta L. polysaccharide (EAPW) was prepared through extraction, precipitation, and column chromatography. Hydrolysis of carbohydrate polymers was carried out, and the resulting solutions were analyzed using the optimized HPLC method.
Results:
Optimization of Chromatographic Conditions: The study optimized column type, temperature, pH, and additives to achieve satisfactory separation of monosaccharides within 25 minutes.
Validation of the Analytical Method: The developed method exhibited good linearity, sensitivity, precision, and accuracy. LODs and LOQs were determined, and spike recovery tests showed acceptable accuracy. Intra- and inter-day precision demonstrated excellent reproducibility.
Application to Plant-Derived Oligo- and Polysaccharides: The method was successfully applied to determine the monosaccharide compositions of stachyose and EAPW. Stachyose was found to contain fructose, glucose, and galactose in a molar ratio consistent with theoretical values. EAPW was identified as a complex polysaccharide with specific monosaccharide compositions.
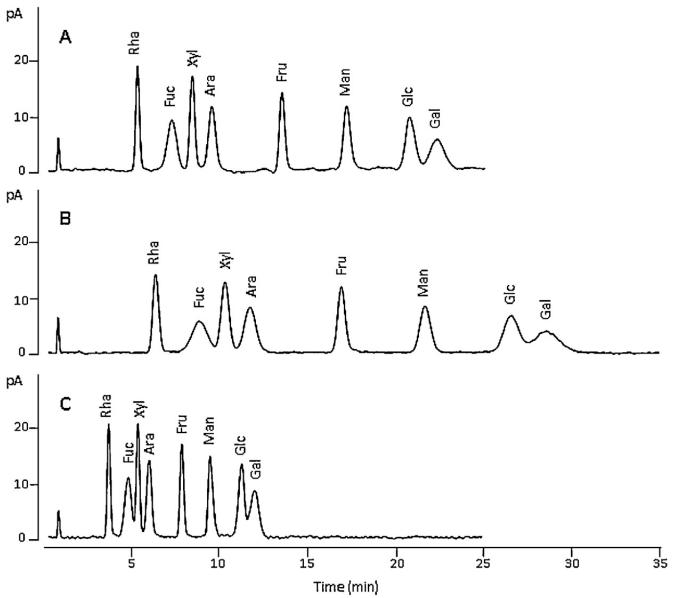 HILIC-CAD chromatogram of monosaccharide standard solution mixture obtained with the mobile phases of (A) 23 mM ammonium acetate (pH 10.8 adjusted with ammonium hydroxide) in 88% ACN; (B) 23 mM ammonium acetate (pH 9.8 adjusted with ammonium hydroxide) in 88% ACN; and (C) 23 mM ammonium acetate (pH 10.8 adjusted with ammonium hydroxide) in 86% CAN.
HILIC-CAD chromatogram of monosaccharide standard solution mixture obtained with the mobile phases of (A) 23 mM ammonium acetate (pH 10.8 adjusted with ammonium hydroxide) in 88% ACN; (B) 23 mM ammonium acetate (pH 9.8 adjusted with ammonium hydroxide) in 88% ACN; and (C) 23 mM ammonium acetate (pH 10.8 adjusted with ammonium hydroxide) in 86% CAN.
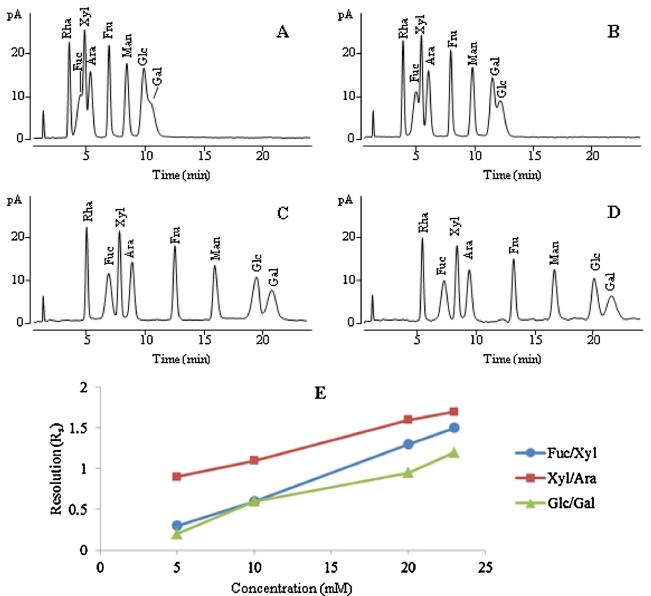 HILIC-CAD chromatogram of monosaccharide standard solution mixture obtained with the ammonium acetate concentration of (A) 5 mM; (B) 10 mM; (C) 20 mM; (D) 23 mM; (E) resolution (Rs) of the critical separation pairs at different concentrations of ammonium acetate.
HILIC-CAD chromatogram of monosaccharide standard solution mixture obtained with the ammonium acetate concentration of (A) 5 mM; (B) 10 mM; (C) 20 mM; (D) 23 mM; (E) resolution (Rs) of the critical separation pairs at different concentrations of ammonium acetate.
Reference
- Yan, Jun, et al. "Neutral monosaccharide composition analysis of plant-derived oligo-and polysaccharides by high performance liquid chromatography." Carbohydrate polymers 136 (2016): 1273-1280.




