- Service Details
- Case Study
As the recognition of the significance of protein glycosylation deepens in the biological field, so does the need for sophisticated glycomic analysis services.
Creative Proteomics offers glycan composition analysis, allowing the identification and precise quantification of glycan structures in biomolecules, especially proteins. Glycans are carbohydrate structures attached to proteins and lipids that play a key role in a variety of biological functions such as protein folding, immune response regulation, and cell-cell interactions.
Glycan composition analysis involves not only detecting the presence or absence of glycans in a protein sample, but also the relative quantification of glycans and information on potential linkages, thus providing a holistic view of the glycan profile of a biological sample. Changes in glycan structure can affect protein function and may lead to significant biological changes and diseases, including cancer and infectious diseases.
Technology Platform for Glycan Composition Analysis
Sample Preparation and Glycan Extraction
The process commences with meticulous sample preparation to extract glycans from complex biological matrices. Creative Proteomics employs enzymatic digestion and chemical extraction methods to liberate glycans from glycoproteins or glycolipids while preserving their structural integrity. Enzymes like PNGase F and Endo H are utilized to cleave glycan moieties from protein backbones, ensuring the isolation of free glycans for subsequent analysis.
Glycan Labeling and Derivatization
To enhance the detection sensitivity and selectivity of glycans, Creative Proteomics employs various labeling and derivatization strategies. Fluorescent tags such as 2-aminobenzamide (2-AB) and 2-aminobenzoic acid (2-AA) are commonly used to label glycans prior to analysis, enabling fluorescence detection and quantification. Additionally, derivatization methods such as permethylation and reductive amination can be applied to modify glycan structures, facilitating their characterization by MS analysis.
High-Performance Liquid Chromatography (HPLC)
With strengths in separation efficiency and resolution, HPLC is best suited for detailed glycan profiling, including identification and quantification. Creative Proteomics utilizes advanced HPLC platforms coupled with fluorescence detection for enhanced glycan detection sensitivity and quantification accuracy. Whether it's normal-phase HPLC (NP-HPLC) for separation based on hydrophilicity or reverse-phase HPLC (RP-HPLC) for hydrophobic interaction, Creative Proteomics ensures precise and comprehensive analyses.
Mass Spectrometry (MS)
Mass Spectrometry is a powerful and versatile tool used for glycan composition analysis. It provides sensitive and comprehensive profiling of complex glycan mixtures, yielding detailed information about glycan structure, composition, sequence, and even linkage. Creative Proteomics leverages MS-based methodologies, including MALDI-TOF MS and ESI MS, in conjunction with specific glycan release methods and derivatization, to facilitate the characterization of diverse glycan structures and compositions.
Bioinformatics and Data Analysis
Creative Proteomics utilizes advanced software algorithms and databases to process and interpret mass spectrometry data, identifying glycan species, annotating glycan structures, and quantifying their abundance in biological samples. Customized data processing pipelines enable the integration of chromatographic and spectral information, facilitating comprehensive glycan characterization.
Glycan Composition Analysis by Creative Proteomics
- Glycan Profiling: Delve into the abundance and structures of glycans within your glycome, offering a comprehensive snapshot of glycosylation status.
- Glycan Sequencing: Unravel the precise sequence of monosaccharide units in your glycans, including linkage analysis and overall architecture insights.
- Comprehensive Glycosylation Site Analysis: Pinpoint the exact attachment sites of glycans on proteins using cutting-edge mass spectrometry techniques, ensuring precise localization.
Sample Requirements for Glycan Composition Analysis
| Sample Type | Recommended Sample Quantity |
|---|---|
| Serum/plasma | 100-500 μL |
| Tissue (e.g., liver, brain) | 10-50 mg |
| Cell culture supernatant | 1-5 mL |
| Biopsy specimens | 5-20 mg |
| Saliva | 100-500 μL |
| Urine | 1-5 mL |
| Cerebrospinal fluid | 100-500 μL |
| Glycoprotein/ glycolipid extracts | 5-20 μg |
Applications of Glycan Composition Analysis
Disease Biomarker Discovery: Identifying disease-specific glycan biomarkers for early detection and prognosis assessment.
Biopharmaceutical Characterization: Assessing glycosylation patterns of therapeutic proteins to ensure product consistency and compliance with regulatory standards.
Glycoprotein Engineering: Modifying glycan profiles of proteins to enhance their therapeutic efficacy and reduce immunogenicity.
Biomolecular Interactions: Studying glycan-protein interactions to elucidate molecular recognition mechanisms and identify therapeutic targets.
Structure-Function Relationships: Investigating how glycan structures influence biological functions, aiding in drug development and diagnostics.
Personalized Medicine: Profiling individual glycan signatures for personalized disease risk assessment and treatment optimization.
Glycan-based Vaccines: Designing vaccines that target specific glycan epitopes to induce immune responses against pathogens or cancer cells.
Quality Control in Food and Agriculture: Analyzing glycan profiles in food products and agricultural crops to assess quality, authenticity, and nutritional value.
Glycan-mediated Drug Delivery: Engineering glycan-based drug delivery systems for targeted delivery of therapeutic agents to specific tissues or cell types.
Stem Cell Research: Studying glycan expression patterns during stem cell differentiation and development, providing insights into cell fate determination and tissue regeneration.
Glycan Profiling in Microorganisms: Characterizing glycan structures in microorganisms for microbial identification, pathogen detection, and vaccine development.
Case. Comprehensive Analysis of Rat Brain Membrane N-Glycoproteome
Background:
Understanding the complex landscape of protein glycosylation is crucial for elucidating various biological processes. The glycoproteome, particularly membrane-associated proteins, plays significant roles in cell signaling, adhesion, and recognition. However, due to the heterogeneity and dynamic nature of glycosylation, comprehensive characterization of the glycoproteome remains challenging.
Samples:
The study focuses on the rat brain membrane N-glycoproteome, a model system chosen for its relevance to neurological functions and disease processes. Membrane-associated proteins from the rat brain were utilized for detailed glycomic and glycoproteomic analyses.
Technical Method:
Peptide Preparation and Glycopeptide Enrichment: Membrane-associated proteins were prepared, digested with trypsin, and enriched for glycopeptides using a combination of chemical and enzymatic treatments. The enriched glycopeptides were subjected to solid-phase extraction, followed by zwitterionic hydrophilic interaction liquid chromatography (HILIC) for further purification.
Orthogonal Fractionation of Glycopeptides Using RPLC: Glycopeptides were fractionated offline using reversed-phase liquid chromatography (RPLC) at pH 7.9, followed by online RPLC at pH 2.7. This orthogonal fractionation strategy reduced peptide complexity in each fraction and facilitated subsequent analysis.
N-Glycomic Analysis of the Rat Brain: N-Glycans released from membrane-associated proteins were analyzed by porous graphitized carbon liquid chromatography-tandem mass spectrometry (PGC-LC-MS/MS) to identify unique monosaccharide compositions and N-glycan structures.
Analysis of N-Glycopeptides from Rat Brain: Enriched glycopeptides were analyzed using nano-reversed-phase liquid chromatography-mass spectrometry (nanoRPLC-MS/MS) with collision-induced dissociation (CID) and higher energy collision dissociation (HCD) fragmentation techniques. This analysis enabled the identification of formerly glycosylated peptides and intact glycopeptides, providing site-specific glycan structure assignments.
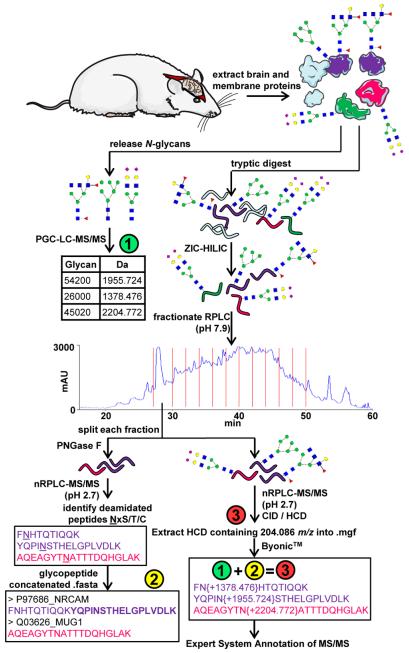 Site-specific N-glycosylation heterogeneity analysis of rat brain membrane-associated proteins by combining glycomics, glycoproteomics, and complementary mass spectrometry (MS) fragmentation techniques.
Site-specific N-glycosylation heterogeneity analysis of rat brain membrane-associated proteins by combining glycomics, glycoproteomics, and complementary mass spectrometry (MS) fragmentation techniques.
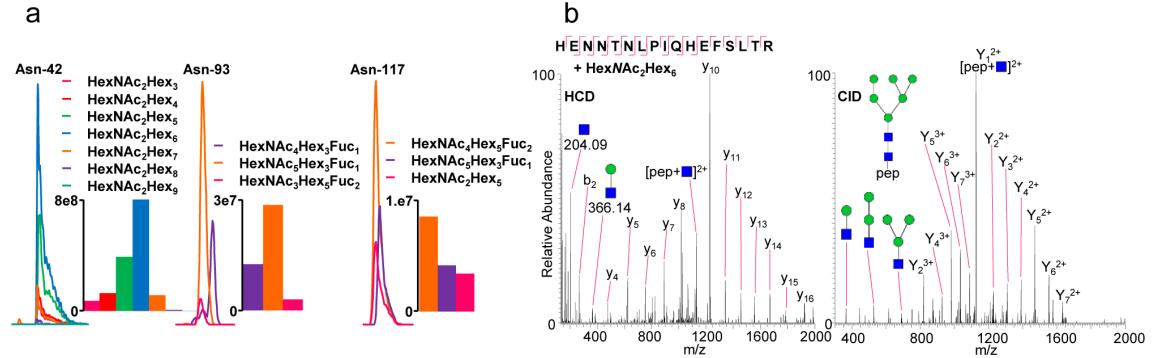 Site-specific N-glycosylation analysis of Thy-1 membrane glycoprotein (P01830) from rat brain.
Site-specific N-glycosylation analysis of Thy-1 membrane glycoprotein (P01830) from rat brain.
Results:
The comprehensive analysis provided insights into the heterogeneity of the rat brain membrane N-glycoproteome, identifying numerous glycosylation sites and structural isomers. Notably, the study expanded the glycoprotein landscape by characterizing novel glycosylation sites on trans-membrane receptor proteins, shedding light on their potential roles in neurological processes. Additionally, the analytical strategy employed demonstrated its effectiveness in unraveling the complex glycoproteome, paving the way for further investigations into glycosylation-related mechanisms in neurological disorders.
Reference
- Parker, Benjamin L., et al. "Site-specific glycan-peptide analysis for determination of N-glycoproteome heterogeneity." Journal of proteome research 12.12 (2013): 5791-5800.




