- Service Details
- Case Study
Glycosaminoglycans, frequently abbreviated GAGs, are unbranched polysaccharides that contain sulfated disaccharide repeating units. They are an integral component of the extracellular matrix; they interact with numerous proteins where they modulate significant biological functions such as cell adhesion, proliferation, differentiation, and morphogenesis.
When dealing with GAGs, problems arise as their structure and composition are highly complex and vary considerably from one tissue to another, and even from one cell type to another within the same tissue. Misregulation of GAG expression or modifications can contribute to the development and progression of various diseases including cancer, rheumatoid arthritis, atherosclerosis, and fibrotic diseases. Accurate glycosaminoglycan analysis can assist in understanding the fundamental mechanisms that drive these diseases, providing insight for potential therapeutic targets.
Given that changes in glycosaminoglycan expression and structure often correlate with disease progression, glycosaminoglycans have great potential for the development of diagnostic and prognostic tools, and Creative Proteomics offers a one-stop shop for glycosaminoglycan analysis to help you with your research.
Glycosaminoglycans Analysis Services in Creative Proteomics
- GAG Structure Elucidation: Comprehensive characterization of GAG structure, including disaccharide composition, sulfation patterns, and linkage analysis.
- GAG Quantification: Accurate quantification of GAGs in biological samples using state-of-the-art analytical techniques such as liquid chromatography-mass spectrometry (LC-MS) and enzyme-linked immunosorbent assay (ELISA).
- GAG-Protein Interaction Studies: Investigation of GAG-protein interactions through surface plasmon resonance (SPR), isothermal titration calorimetry (ITC), and other biophysical assays.
- GAG Biomarker Discovery: Identification and validation of GAG biomarkers for various diseases, including cancer, cardiovascular disorders, and inflammatory conditions.
Technical Platforms for Glycosaminoglycans Analysis
- Liquid Chromatography-Mass Spectrometry (LC-MS): High-resolution LC-MS for the characterization and quantification of GAGs, allowing for precise identification of structural modifications and quantitative analysis of GAG species.
- Capillary Electrophoresis (CE): CE-based methods for rapid separation and analysis of GAGs, offering excellent resolution and sensitivity for complex samples.
- Enzyme-Linked Immunosorbent Assay (ELISA): ELISA kits and custom assay development for the quantitative detection of specific GAGs in biological matrices, providing a cost-effective solution for large-scale screening.
- Surface Plasmon Resonance (SPR): SPR biosensors for real-time monitoring of GAG-protein interactions, enabling kinetic analysis and affinity determination with high precision.
Sample Requirements for Glycosaminoglycans Analysis
| Sample Type | Sample Quantity Required |
|---|---|
| Tissue Biopsy | 5-20 mg |
| Cell Culture Supernatant | 1-5 mL |
| Serum/Plasma | 50-200 μL |
| Urine | 100-500 μL |
| Synovial Fluid | 100-500 μL |
| Cerebrospinal Fluid | 100-500 μL |
| Saliva | 100-500 μL |
| Tears | 50-200 μL |
| Skin Swab | Swab saturated with buffer |
| Cartilage | 5-20 mg |
| Bone | 5-20 mg |
Advantages of Glycosaminoglycans Assay in Creative Proteomics
Expertise and Experience: Our team of experienced scientists possesses extensive expertise in glycosaminoglycan analysis, ensuring reliable results and interpretation.
Customized Solutions: We tailor our analysis services to accommodate specific research objectives and sample types, providing personalized solutions for each project.
State-of-the-Art Facilities: Creative Proteomics is equipped with state-of-the-art instrumentation and facilities, enabling advanced analytical capabilities and rapid turnaround times.
Quality Assurance: We adhere to stringent quality control measures throughout the analysis process, ensuring accuracy, precision, and reproducibility.
Comprehensive Support: From project design to data analysis and interpretation, our dedicated support team offers comprehensive assistance at every stage of your research.
Applications of Glycosaminoglycans Analysis
Biomedical Research: Understanding the structure and function of GAGs is essential for studying various biological processes such as cell signaling, tissue development, and inflammation. Analyzing GAGs helps researchers elucidate their roles in health and disease, including cancer, neurodegenerative disorders, and cardiovascular diseases.
Drug Development: GAGs serve as targets for drug development and therapy. Analyzing GAGs can aid in the discovery and development of drugs that modulate GAG-related pathways, such as anticoagulants, anti-inflammatory agents, and treatments for diseases involving abnormal GAG metabolism.
Diagnostics: Abnormalities in GAG metabolism are associated with several diseases, including lysosomal storage disorders (e.g., mucopolysaccharidoses) and osteoarthritis. GAG analysis can be used for diagnostic purposes, helping clinicians identify and monitor these conditions through biomarker detection and quantification.
Biotechnology: GAGs are valuable biomolecules used in various biotechnological applications, including tissue engineering, drug delivery systems, and wound healing. Analyzing GAGs enables researchers to optimize their properties for specific applications and develop novel GAG-based materials with improved functionality.
Quality Control: GAG analysis is essential for quality control in industries producing GAG-based products such as cosmetics, pharmaceuticals, and biomedical devices. Ensuring the purity, consistency, and structural integrity of GAGs is critical for maintaining product efficacy and safety.
Glycobiology: GAG analysis contributes to the broader field of glycobiology, which focuses on the study of carbohydrates and their biological roles. By characterizing GAG structures and interactions with proteins and other molecules, researchers gain insights into the complex glycocode and its significance in cellular communication and physiology.
Regenerative Medicine: GAGs play crucial roles in tissue regeneration and repair processes. Analyzing GAG composition and distribution in native tissues and engineered constructs helps researchers design strategies for enhancing tissue regeneration and developing advanced therapies for tissue injuries and degenerative diseases.
Pharmacokinetics and Pharmacodynamics: Understanding the pharmacokinetics and pharmacodynamics of GAG-based drugs requires accurate analysis of GAG metabolism, distribution, and clearance in vivo. Analytical techniques such as mass spectrometry and chromatography enable researchers to track GAG dynamics in biological systems and optimize drug efficacy and safety profiles.
Title: In vivo Characterization of Asporin-Deficient Mouse Skin Reveals Altered Extracellular Matrix Composition and Biomechanical Properties
Background
Asporin, a small leucine-rich proteoglycan (SLRP), plays a crucial role in modulating collagen fibrillogenesis in vitro. However, its in vivo functions, particularly in skin extracellular matrix (ECM) biology, remain poorly understood. This study aims to investigate the impact of asporin deficiency on skin ECM composition and biomechanical properties in mice.
Sample
The study utilized skin samples from two-month-old mice, including wild-type and Aspn-/- (asporin knockout) mice. Each genotype comprised four samples for analysis, ensuring robustness and reliability in the results.
Technical Methods
Protein Extraction: Proteins were extracted from mouse skin using a dissociating buffer, followed by purification on DE-52 anion-exchange resin.
Protein Quantification: Protein concentrations were determined using the Bradford method.
Proteoglycan Analysis: Total proteoglycans were eluted from DE-52 resin and analyzed by SDS-PAGE and immunoblotting after digestion with chondroitinase ABC.
Skin Histology and Electron Microscopy: Histological examinations and transmission electron microscopy were performed to assess skin structure and collagen fibril morphology.
Gene Expression Analysis: qPCR was used to quantify the expression of extracellular matrix-related genes.
Glycosaminoglycan (GAG) Analysis: GAG content and structure were analyzed using HPLC quantification and identification of disaccharides obtained after enzymatic digestion.
Biomechanical Testing: Mechanical properties of the skin were assessed, including toughness and stress-strain curves.
Results
Skin Histology: Aspn-/- skin showed no gross abnormalities in histological examinations compared to wild-type.
Collagen Fibril Phenotype: Minor changes in collagen fibril diameter were observed in Aspn-/- skin, but overall collagen structure appeared normal.
Gene Expression: Aspn-/- skin exhibited increased expression of collagen-related genes and enzymes involved in ECM turnover.
GAG Composition: Aspn-/- skin showed elevated chondroitin/dermatan sulfate (CS/DS) content and reduced heparan sulfate (HS), indicating altered GAG composition.
Biomechanical Properties: Aspn-/- skin demonstrated increased toughness, possibly attributed to thinner collagen fibrils and changes in GAG composition.
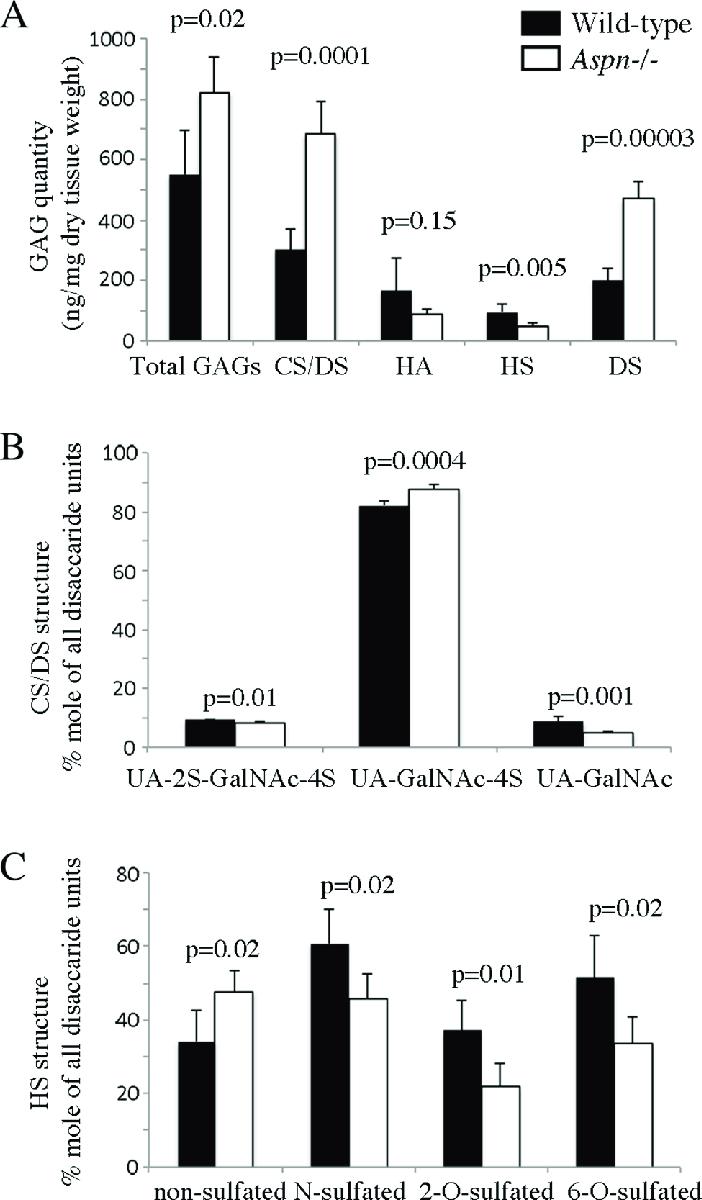 Skin glycosaminoglycan (GAG) analysis
Skin glycosaminoglycan (GAG) analysis
Reference
- Maccarana, Marco, et al. "Asporin-deficient mice have tougher skin and altered skin glycosaminoglycan content and structure." PLoS One 12.8 (2017): e0184028.




