- Service Details
- Case Study
What is Glycan Release?
Glycan release refers to the process of liberating glycans from glycoproteins or glycolipids. Glycans are complex carbohydrate structures that are covalently attached to proteins (glycoproteins) or lipids (glycolipids). The release of glycans is a crucial step in the analysis of glycoproteins, as it allows for the examination and characterization of the glycan structures.
After glycan release, the liberated glycans can be further analyzed using various techniques, including mass spectrometry, liquid chromatography, and capillary electrophoresis. These methods help in identifying the composition, structure, and heterogeneity of glycans attached to specific proteins.
Understanding glycan structures is essential in the study of glycoproteins because glycans play crucial roles in various biological processes, including cell signaling, immune response, and protein stability. Glycan release and subsequent analysis contribute to our understanding of glycoprotein function and can have implications in areas such as disease diagnosis and therapeutic development.
Why is Glycan Release Necessary?
Structural Insights: Glycans contribute significantly to the structural diversity of glycoproteins. Understanding the intricate arrangements of these sugar molecules provides insights into the functional roles of glycoproteins. Glycan release facilitates the isolation of glycans for detailed structural analysis, including mass spectrometry and nuclear magnetic resonance (NMR) spectroscopy.
Functional Understanding: The biological activities of glycoproteins are often intricately linked to their glycan moieties. By releasing glycans, researchers gain a deeper understanding of the functional roles played by these sugar molecules. This knowledge is crucial for deciphering processes such as cell adhesion, immune response, and signal transduction.
Disease Biomarker Discovery: Glycan alterations are associated with various diseases, including cancer and autoimmune disorders. Glycan release services enable the identification and quantification of disease-specific glycan changes. This has implications for biomarker discovery, diagnostics, and the development of targeted therapies.
Glycan Release Services at Creative Proteomics
In the dynamic landscape of glycoprotein analysis, the effectiveness of glycan release methods is paramount. Creative Proteomics, a pioneer in the field, offers unparalleled glycan release services, harnessing advanced methodologies to unlock the intricate details of N-glycan and O-glycan structures.
N-Glycan Liberation
Creative Proteomics employs a multifaceted approach to N-glycan liberation, recognizing the critical role of the chosen method in preserving glycan integrity. Our methods include enzymatic cleavage using PNGase F, chemical methods such as hydrazinolysis, and innovative combination approaches.
- Enzymatic Methods: PNGase F
Utilizing the powerful endoglycosidase PNGase F, we achieve selective cleavage of the glycan chain at the asparagine residue. This method ensures the liberation of intact N-glycans, offering high specificity, minimal side reactions, and suitability for a broad range of glycoproteins.
- Chemical Methods: Hydrazinolysis
Incorporating hydrazine as a reactive agent, hydrazinolysis enables the cleavage of the glycan from the asparagine residue. This chemical method is versatile, effective for diverse glycoproteins, and compatible with downstream analyses. Careful handling is advised due to the reactive nature of hydrazine.
O-Glycan Liberation
Our techniques encompass enzymatic cleavage using PNGase F, as well as chemical approaches such as beta-elimination and hydrazinolysis.
- Enzymatic Cleavage: PNGase F
Through a meticulous process, glycoproteins undergo incubation with PNGase F, resulting in the specific cleavage of the glycosidic bond and the subsequent liberation of O-glycans. This method boasts high specificity for N-glycosidic bonds, ensuring minimal impact on O-glycan structures, and is compatible with various glycoproteins.
- Chemical Approaches: Beta-Elimination
In this method, glycoproteins are treated with a beta-elimination reagent, initiating the chemical cleavage of O-glycans. The released O-glycans are then ready for subsequent analysis. Beta-elimination demonstrates applicability to diverse glycoprotein structures and offers versatility in cleaving various types of O-glycans.
- Chemical Approaches: Hydrazinolysis
The hydrazinolysis process involves the incubation of glycoproteins with hydrazine, facilitating the chemical cleavage of O-glycans. This results in the effective liberation of O-glycans with minimal side reactions, making it compatible with diverse glycoprotein structures.
Why Choose Our Glycan Release Service?
Customized Solutions: Recognizing the uniqueness of each research project, our services are customizable to meet your specific requirements, tailoring liberation methods based on your glycoprotein's structural features.
Versatility in Methods: Whether through enzymatic cleavage, chemical approaches, or a combination of both, our versatile methods adapt to a diverse range of glycoprotein structures.
Precision and Specificity: Enzymatic methods, such as using PNGase F, offer high specificity for glycosidic bonds, preserving the integrity of N-glycans, while chemical approaches are executed with precision for effective O-glycan liberation.
Optimization for Minimal Impact: Our processes are optimized to minimize side reactions, ensuring released glycans accurately represent the native structures in your samples.
Compatibility Across Glycoproteins: Designed to be compatible with a broad range of glycoproteins, our methods cater to various samples, from well-known glycoproteins to those with unique structures.
Sample Requirements for Glycan Release
| Sample Type | Recommended Sample Amount |
|---|---|
| Serum/Plasma | 20-100 μL |
| Cell Culture Supernatant | 1-5 mL |
| Tissue Homogenate | 10-50 mg |
| Urine | 20-100 μL |
| Saliva | 20-100 μL |
| CSF (Cerebrospinal Fluid) | 10-50 μL |
| Cell Lysate | 1-5 x 10^6 cells |
| Biopsy Specimen | 5-20 mg |
| Glycoprotein Isolated Fraction | 1-5 mg |
Note:
- The recommended sample amounts are general guidelines and may vary based on the specific characteristics of your samples.
- For precise requirements and to discuss special cases, please contact our team for personalized assistance.
- Ensure proper storage conditions (e.g., -80°C for tissues) to maintain sample integrity before submission.
- If your sample type is not listed or if you have unique requirements, feel free to reach out to our experts for tailored guidance.
Case. Oxidative Release of Natural Glycans for Functional Glycomics
Background:
Glycomics, particularly the analysis of glycan structures, faces challenges in obtaining sufficient amounts of natural glycans from complex biological samples. This limitation hinders progress in understanding protein-glycan interactions and glycan functions. Traditional methods involve expensive enzymes or corrosive chemicals, making large-scale analyses difficult.
Samples
The study utilized diverse biological samples, including kilogram quantities of animal tissues, store-bought chicken eggs, and mouse gastrointestinal tract tissues. These samples were chosen for their representation of various glycoproteins and glycoconjugates.
Technical Method
The Oxidative Release of Natural Glycans (ORNG) method was introduced as an innovative and cost-effective approach. It involves controlled treatment of biological samples with sodium hypochlorite (NaClO), the active ingredient in commercial bleach. The method selectively releases intact N-, O-, and GSL-glycans, allowing for their subsequent analysis and functional studies.
Results
N-glycans Release: NaClO treatment effectively released free reducing N-glycans from glycoproteins. This process was applicable to various common glycoproteins, preserving labile sialic acid residues.
O-glycan Derivatives: Unexpectedly, ORNG proved useful for preparing O-glycan derivatives, particularly O-glycan acids. This was achieved by selectively degrading the aglycon portion of native glycoconjugates.
GSLs Degradation: ORNG demonstrated the ability to degrade glycosphingolipids (GSLs), a challenging class of glycoconjugates. The method resulted in the release of glycan nitriles from GSLs, offering a novel approach to study this complex class of glycans.
Large-Scale Glycan Libraries: ORNG facilitated the preparative release of N- and O-glycans, allowing for the generation of gram-scale crude N-glycans and major O-glycan-acids. This enabled the creation of comprehensive glycan libraries for subsequent analyses.
Functional Tagging: The study introduced methods for functional tagging of released glycans, including derivatization with fluorescent linkers. This facilitated downstream applications such as chromatographic separation, microarray preparation, and glycan recognition studies.
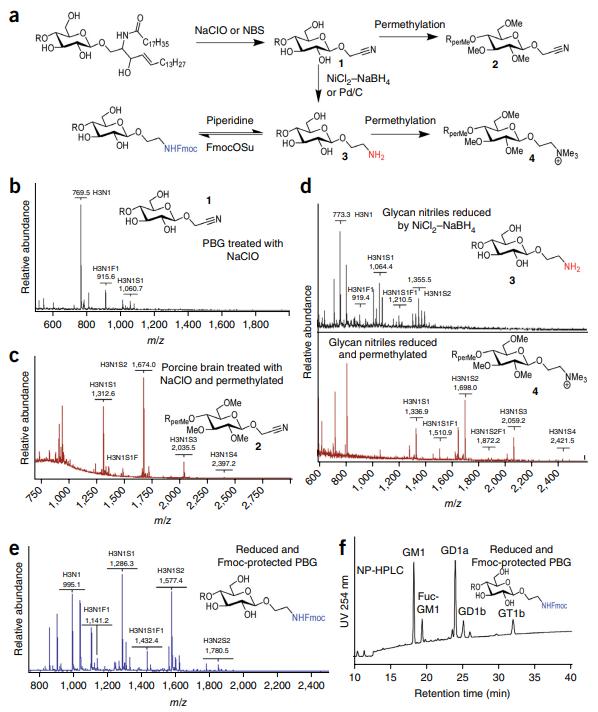 Release and tagging of glycans from GSLs by NaClO
Release and tagging of glycans from GSLs by NaClO
Reference
- Song, Xuezheng, et al. "Oxidative release of natural glycans for functional glycomics." Nature methods 13.6 (2016): 528-534.




