- Service Details
- Case Study
Glycoproteins, a class of proteins covalently linked to oligosaccharide chains, play pivotal roles in numerous biological processes. Understanding their three-dimensional structures is crucial for elucidating their functions and designing targeted therapeutics. Among the various techniques available for structural analysis, Circular Dichroism (CD) spectroscopy stands out as a powerful tool for probing the conformational intricacies of glycoproteins.
Principles of CD Spectroscopy
CD spectroscopy exploits the differential absorption of left and right circularly polarized light by chiral molecules. When circularly polarized light interacts with a chiral molecule, such as a glycoprotein, the absorption of photons induces transitions between energy states, resulting in characteristic CD spectra. The magnitude and shape of the CD spectrum provide information about the secondary and tertiary structures of the molecule, as well as its dynamics and environment.
Why Choose CD Spectroscopy?
Glycoprotein CD analysis offers several advantages for structural characterization:
- Non-destructive Analysis: CD spectroscopy requires minimal sample preparation and can be performed under near-physiological conditions, preserving the native structure of glycoproteins.
- Sensitivity to Structural Changes: CD spectra are sensitive to changes in protein conformation induced by glycan moieties, making it ideal for studying glycoprotein folding, stability, and interactions.
- Quantitative Analysis: Through spectral deconvolution and computational modeling, CD spectra can be quantitatively analyzed to determine the relative abundance of secondary structural elements and infer tertiary structural features.
Technical Platform for Glycoprotein CD Analysis
High-Performance CD Spectrometers: State-of-the-art CD spectrometers equipped with variable temperature control, precise wavelength selection, and automated data acquisition capabilities enable high-resolution analysis of glycoprotein structures.
Advanced Data Analysis Software: Specialized software packages for spectral deconvolution, curve fitting, and computational modeling facilitate the interpretation of CD spectra and the extraction of structural parameters.
Expertise in Glycoprotein Analysis: A team of experienced scientists with expertise in structural biology, protein chemistry, and computational modeling ensures the accurate interpretation of CD data and the generation of actionable structural insights.
Analytical Offerings for Glycoprotein CD Analysis
At Creative Proteomics, we offer a wide range of glycoprotein CD analysis services, including:
1. Secondary Structure Determination: Quantification of α-helix, β-sheet, β-turn, and random coil content in glycoproteins based on CD spectra analysis.
2. Tertiary Structure Prediction: Inference of spatial arrangements of glycoprotein domains and glycan-binding sites from CD spectra using computational modeling techniques.
3. >Thermal Stability Profiling: Assessment of glycoprotein stability and unfolding kinetics as a function of temperature using temperature-dependent CD spectroscopy.
4. Glycan-Protein Interaction Studies: Investigation of conformational changes induced by glycan binding and elucidation of the molecular basis of glycoprotein recognition and signaling events.
5. Comparative Analysis: Comparative CD spectroscopy to identify structural differences between glycoprotein variants and assess the impact of glycosylation on protein structure and function.
Sample Requirements for Glycoprotein Circular Dichroism (CD) Analysis
| Sample Type | Sample Volume (µL) | Concentration (mg/mL) | Purity (%) | Glycan Composition |
|---|---|---|---|---|
| Monoclonal Antibody | 200 | 1-5 | >90 | N-glycan |
| Recombinant Protein | 100 | 2-10 | >95 | O-glycan |
| Serum/Plasma | 50 | 1-20 | >85 | N-glycan |
| Cell Culture Supernatant | 100 | 2-15 | >80 | N-glycan |
| Glycoprotein Standard | 50-100 | 1-10 | >98 | Various |
| Tissue Extract | 100-200 | 2-20 | >80 | N-glycan, O-glycan |
| Urine | 50-100 | 1-5 | >90 | N-glycan, O-glycan |
| Saliva | 50-100 | 1-10 | >85 | N-glycan, O-glycan |
| Cerebrospinal Fluid (CSF) | 50-100 | 1-15 | >90 | N-glycan, O-glycan |
Case. Characterization and Anticancer Activity of a Novel Glycoprotein from C. decorticatum
Background:
Previous research by Thangam et al. (2014a) identified a glycoprotein (GLP) from C. decorticatum with potential therapeutic properties. This study aimed to further characterize this GLP and investigate its anticancer activity against various cancer cell lines.
Samples:
The sample used in this study was a purified glycoprotein extracted from C. decorticatum.
Technical Method:
Chemical Composition Analysis: The GLP was extracted and purified, with its purity confirmed using SDS-PAGE and PAS staining. Carbohydrate and protein content were determined using phenol-sulfuric acid and Lowry's methods, respectively. Monosaccharide composition was analyzed, and amino acid composition was determined by HPLC.
Isoelectric Point Determination: The GLP's isoelectric point was determined using two-dimensional gel electrophoresis.
Conformation Analysis: Infrared (IR) spectroscopy, nuclear magnetic resonance (NMR) spectroscopy, and circular dichroism (CD) analysis were performed to study the conformation of GLP.
Mass Spectrometry and Protein Identification: Mass spectrometry analysis was conducted to identify the GLP, and peptide mass fingerprinting was performed for protein identification.
Anticancer Activity Evaluation: The cytotoxicity of GLP against MCF-7, Siha, and A549 cancer cell lines was assessed using MTT reduction assays. Cell membrane damage and induction of apoptosis were evaluated through LDH release assays and AO/EtBr staining, respectively.
Results:
Chemical Composition: GLP was found to contain 36.24% carbohydrate and 63.76% protein, with predominant monosaccharides being rhamnose and galactose. Amino acid analysis revealed 13 amino acids, with cystathionine being the most abundant.
Isoelectric Point: The GLP exhibited an acidic isoelectric point of approximately 4.
Conformation Analysis: IR, NMR, and CD analyses confirmed GLP's composition of protein and carbohydrate components, along with its secondary structural elements.
Protein Identification: Mass spectrometry identified GLP as a protein homologous to 50S ribosomal protein L11 from Pyropia yezoensis.
Anticancer Activity: GLP demonstrated time-dependent cytotoxicity against cancer cell lines, inducing cell membrane damage and apoptosis, while sparing normal cells.
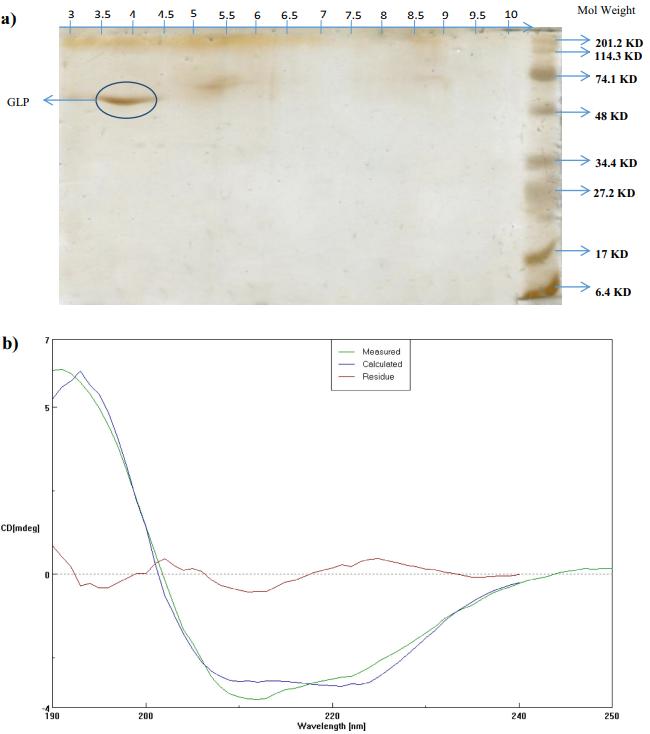 The purified glycoprotein (GLP): (a) two-dimensional gel electrophoresis analysis of GLP from C. decorticatum and (b) circular dichroism spectrum of purified GLP for the secondary structure analysis.
The purified glycoprotein (GLP): (a) two-dimensional gel electrophoresis analysis of GLP from C. decorticatum and (b) circular dichroism spectrum of purified GLP for the secondary structure analysis.
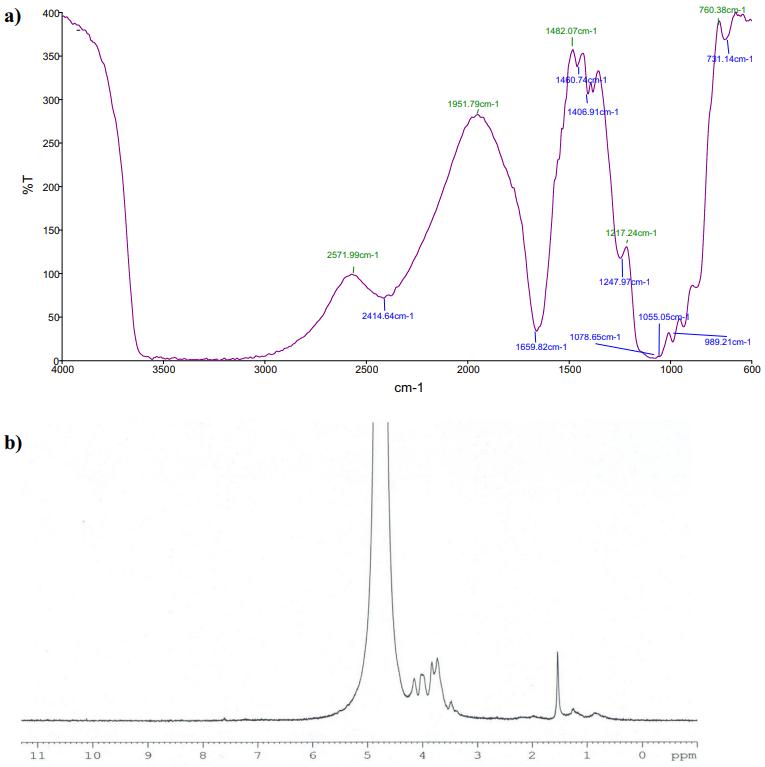 Conformation analysis GLP from C. decorticatum: (a) FTIR spectrum of GLP for functional group analysis and (b) 1 H NMR spectrum of GLP for chemical structure.
Conformation analysis GLP from C. decorticatum: (a) FTIR spectrum of GLP for functional group analysis and (b) 1 H NMR spectrum of GLP for chemical structure.
Reference
- Senthilkumar, Dharmaraj, and Sivaraman Jayanthi. "Partial characterization and anticancer activities of purified glycoprotein extracted from green seaweed Codium decorticatum." Journal of Functional Foods 25 (2016): 323-332.




