- Service Details
- Case Study
Introduce of Glycoprotein Secondary Structure
Glycoproteins represent a class of proteins that are fundamentally complex due to the bonding of carbohydrate moieties to the protein chain. They play pivotal roles in numerous biological processes, including cell-cell communication, protein folding, immune responses, and pathogenic mechanisms. Understanding their secondary structure can shed light on their function and behavior in biological systems.
As part of the protein structure hierarchy, the secondary structure refers to the spatial arrangement of adjacent amino acids. These arrangements create regular patterns, typically represented by alpha-helices and beta-sheets. These orderly structures arise from hydrogen bonding, particularly along the protein backbone, acting as the underpinning framework for the protein's three-dimensional configuration.
With glycoproteins, the secondary structure becomes even more critical owing to the attached carbohydrates, which can induce conformational changes or mediate different interactions. Thus, analysis of glycoprotein secondary structures unravels the impact of such modifications on protein function and stability, paving the way for the design of advanced therapeutics and comprehensive understanding of disease mechanisms.
Creative Proteomics' Services for Glycoprotein Secondary Structure Analysis
Structural Modeling and Prediction
Creative Proteomics leverages state-of-the-art computational algorithms and molecular modeling techniques to predict the secondary structure of glycoproteins with unparalleled accuracy. By integrating sequence-based predictions, homology modeling, and molecular dynamics simulations, our experts provide valuable insights into the folding patterns and conformational dynamics of glycoproteins.
Experimental Validation and Characterization
In addition to computational approaches, Creative Proteomics offers a wide range of experimental methods for validating and characterizing the secondary structure of glycoproteins. From X-ray crystallography and NMR spectroscopy to CD and FTIR spectroscopy, our experienced scientists employ cutting-edge technologies to elucidate the structural properties and functional implications of glycoproteins in diverse biological contexts.
Customized Solutions and Consultation
At Creative Proteomics, we understand that every research project is unique, and we are committed to providing tailored solutions and expert consultation to address specific challenges and objectives. Whether it's designing custom experiments, optimizing assay conditions, or interpreting complex data, our team works closely with clients to deliver actionable insights and maximize research outcomes.
Technology Platform for Glycoprotein Secondary Structure Analysis
Glycoprotein LC-MS Disulfide Bridge Mapping
Liquid Chromatography-Mass Spectrometry (LC-MS) stands as a cornerstone technique for elucidating the disulfide bridge connectivity within glycoproteins. By coupling high-performance liquid chromatography with mass spectrometry detection, this approach enables the precise identification and characterization of disulfide linkages, providing crucial structural information about the spatial arrangement of cysteine residues within the glycoprotein's tertiary structure.
Glycoprotein Circular Dichroism (CD)
Circular Dichroism (CD) spectroscopy serves as a powerful tool for probing the secondary structure of glycoproteins in solution. By measuring the differential absorption of left- and right-handed circularly polarized light, CD spectroscopy provides valuable insights into the helical content, beta-sheet formation, and overall fold of glycoproteins. This technique offers rapid and quantitative assessment of structural changes induced by environmental factors or ligand binding, making it indispensable for characterizing the conformational dynamics of glycoproteins.
Glycoprotein Fourier Transform Infrared (FTIR)
Fourier Transform Infrared (FTIR) spectroscopy offers a unique approach to analyzing the secondary structure of glycoproteins based on their vibrational modes. By measuring the absorption of infrared radiation by chemical bonds within the molecule, FTIR spectroscopy provides spectral fingerprints that reflect the composition and conformation of glycoproteins. This technique enables researchers to monitor structural alterations and conformational transitions induced by external stimuli or biochemical processes, thereby unraveling the intricacies of glycoprotein folding and stability.
Glycoprotein Intrinsic Tryptophan Fluorescence (ITF)
Intrinsic Tryptophan Fluorescence (ITF) spectroscopy serves as a sensitive probe for monitoring structural changes and conformational dynamics within glycoproteins. Tryptophan residues, abundant in many glycoproteins, exhibit characteristic fluorescence emission spectra that are sensitive to changes in their local environment. By measuring the intensity, wavelength, and lifetime of tryptophan fluorescence, researchers can gain valuable insights into the tertiary structure, ligand binding, and folding kinetics of glycoproteins, providing critical information about their functional properties and biological activities.
Glycoprotein Differential Scanning Calorimetry (DSC)
Differential Scanning Calorimetry (DSC) offers a thermodynamic approach to studying the secondary and tertiary structure of glycoproteins. By measuring the heat absorbed or released during thermal denaturation or folding transitions, DSC provides valuable information about the stability, folding kinetics, and conformational changes of glycoproteins. This technique is particularly useful for assessing the effects of temperature, pH, and ligand binding on the structural integrity and stability of glycoproteins, offering insights into their biophysical properties and behavior under physiological conditions.
Integrated Approach for Comprehensive Analysis
By integrating these complementary techniques, Creative Proteomics offers a holistic approach to glycoprotein secondary structure analysis, enabling researchers to unravel the complexities of protein folding, stability, and function with unprecedented accuracy and precision. From disulfide bridge mapping and circular dichroism spectroscopy to Fourier transform infrared spectroscopy, intrinsic tryptophan fluorescence, and differential scanning calorimetry, our advanced technology platform empowers scientists to explore the structural intricacies of glycoproteins and unlock new insights into their biological roles and therapeutic potential.
Sample Requirements for Glycoprotein Secondary Structure Analysis
| Sample Type | Sample Volume | Additional Requirements |
|---|---|---|
| Purified Glycoprotein | 100 µg - 500 µg | Concentration: ≥ 0.5 mg/mL |
| Serum or Plasma | 50 µL - 200 µL | Removal of interfering components (e.g., albumin) recommended |
| Cell Culture Supernatant | 1 mL - 5 mL | Centrifugation to remove cell debris recommended |
| Tissue Homogenate | 10 mg - 50 mg | Homogenization in suitable buffer solution |
| Saliva | 100 µL - 500 µL | Centrifugation to remove debris and mucins |
| Urine | 1 mL - 5 mL | Centrifugation to remove cells and debris |
| Cerebrospinal Fluid (CSF) | 100 µL - 500 µL | Centrifugation to remove cells and debris |
| Cell Lysate | Depends on cell density | Ensure efficient cell lysis |
| Biopsy Samples | Variable | Homogenization or extraction in appropriate buffer |
| Body Fluids (e.g., synovial fluid, peritoneal fluid) | Varies | Centrifugation to remove debris and cells |
| Other | Depends on the source | Please contact us for specific recommendations |
Applications of Glycoprotein Secondary Structure Analysis
Drug Discovery and Development
Glycoproteins serve as attractive targets for drug discovery and development due to their involvement in key cellular processes and disease pathways. Secondary structure analysis provides insights into the structural dynamics and conformational changes of glycoproteins, aiding in the design and optimization of therapeutic agents targeting these molecules. By elucidating the effects of ligand binding on glycoprotein structure, researchers can identify potential drug candidates and optimize their pharmacological properties.
Biopharmaceutical Production
In the biopharmaceutical industry, glycoproteins are widely used as therapeutic agents, diagnostic tools, and vaccine components. Secondary structure analysis plays a crucial role in ensuring the quality, efficacy, and safety of glycoprotein-based biopharmaceuticals. By monitoring glycoprotein folding, stability, and post-translational modifications, manufacturers can optimize production processes, control product quality, and meet regulatory requirements.
Biomedical Research
Glycoproteins are involved in diverse biological processes, including cell signaling, immune response, and protein-protein interactions. Secondary structure analysis provides valuable insights into the structure-function relationships of glycoproteins, facilitating biomedical research in areas such as cancer biology, infectious diseases, and neurodegenerative disorders. By elucidating the structural basis of glycoprotein function, researchers can uncover novel therapeutic targets and develop innovative treatment strategies.
Biotechnology and Protein Engineering
Glycoproteins have emerged as versatile tools in biotechnology and protein engineering, serving as scaffolds for protein design, enzyme immobilization, and biocatalysis. Secondary structure analysis enables researchers to engineer glycoproteins with tailored properties and functionalities, such as enhanced stability, altered substrate specificity, or improved therapeutic efficacy. By manipulating glycoprotein structure, researchers can create novel biotechnological applications with potential implications in fields ranging from biocatalysis to biomaterials.
Structural Biology and Protein Engineering
Glycoproteins exhibit intricate folding patterns and structural dynamics that govern their biological functions and interactions. Secondary structure analysis, coupled with advanced structural biology techniques such as X-ray crystallography and nuclear magnetic resonance (NMR) spectroscopy, provides detailed insights into the three-dimensional architecture of glycoproteins. By elucidating the structural determinants of glycoprotein-ligand interactions, researchers can design targeted interventions and modulate biological pathways for therapeutic purposes.
Case. Comprehensive Structural Analysis of Gel-Forming Mucin MUC5B Using Raman Spectroscopic Techniques
Background:
Gel-forming mucins, such as MUC5B, play crucial roles in protecting epithelial surfaces from dehydration, injury, and pathogen colonization. Despite their significance, studying the structural architecture of mucins presents challenges due to their size, polydispersity, and dense glycosylation. Conventional structural techniques are inadequate for investigating these complex glycoproteins.
Samples:
Polymeric MUC5B was isolated from human whole saliva using density gradient centrifugation. MUC5B subdomains, including the heavily O-glycosylated central region and less glycosylated N- and C-terminal regions, were generated for detailed analysis.
Technical Method:
Mucin Purification: MUC5B isolation involved pooling saliva samples from healthy volunteers, followed by solubilization and density gradient centrifugation.
Mucin Subdomain Preparation: O-glycan-rich regions of MUC5B were obtained through reduction/alkylation and trypsin digestion, while N-terminal, C-terminal, and cysteine-rich domains were generated as recombinant proteins.
Raman and ROA Data Acquisition: Spectra were obtained using a ChiralRaman spectrometer, with Raman and ROA measurements focusing on the amide I region for secondary structure analysis.
CD Data Acquisition: Circular dichroism measurements were conducted to assess secondary structure characteristics.
TERS Sample Preparation and Data Acquisition: Tip-enhanced Raman spectroscopy was performed to investigate mucin structure at the single molecule level. Glass substrates were prepared, and AFM was used to visualize individual mucin molecules, followed by TERS measurements along their length.
Results:
Raman and ROA spectra revealed that polymeric MUC5B primarily exhibits an unordered polypeptide backbone due to its heavily O-glycosylated regions, while less glycosylated subdomains displayed a β-sheet structure.
TERS spectra provided insights into mucin structure at the single molecule level, tentatively identifying regions of secondary structure and glycosylation along the molecule's length.
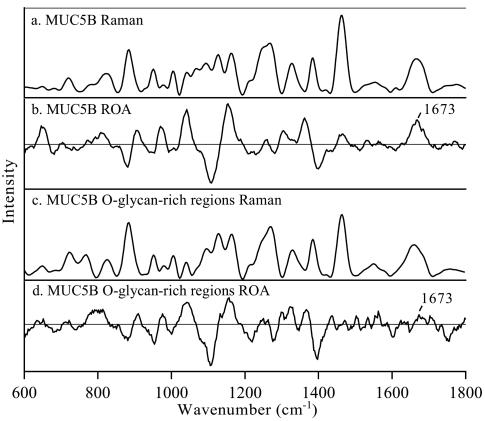 Raman and ROA spectra of MUC5B and MUC5B subdomains.
Raman and ROA spectra of MUC5B and MUC5B subdomains.
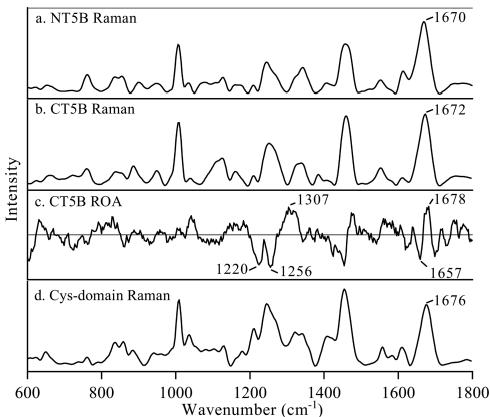 Raman and ROA spectra of MUC5B subdomains.
Raman and ROA spectra of MUC5B subdomains.
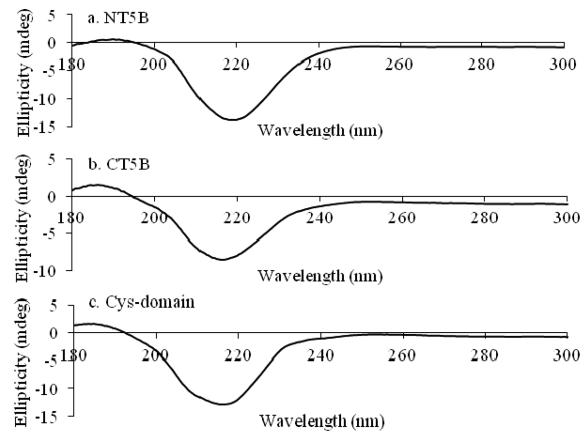 CD spectra of MUC5B subdomains.
CD spectra of MUC5B subdomains.
Reference
- Davies, Heather S., et al. "Secondary structure and glycosylation of mucus glycoproteins by Raman spectroscopies." Analytical chemistry 88.23 (2016): 11609-11615.




