- Service Details
- Case Study
What are Glycoconjugates?
Glycoconjugates are complex molecules found in living organisms, composed of a carbohydrate (sugar) moiety covalently attached to a non-carbohydrate moiety. These non-carbohydrate moieties can include proteins (forming glycoproteins), lipids (forming glycolipids), or other carbohydrates (forming proteoglycans or polysaccharides).
The carbohydrate portion of glycoconjugates, also known as glycans, consists of oligosaccharide chains with various monosaccharide units linked together through glycosidic bonds. These glycans can exhibit diverse structures, including linear or branched arrangements, and can be modified by various chemical groups such as sulfates, phosphates, or acetyl groups.
Glycoconjugates are ubiquitous in nature and are found on the surfaces of cells, in extracellular matrices, and in secreted fluids. They play essential roles in numerous biological processes, including cell-cell recognition, cell adhesion, signaling, immune response modulation, and pathogen-host interactions. The carbohydrate moiety of glycoconjugates often serves as recognition sites for other molecules, facilitating specific interactions and molecular recognition events crucial for biological function.
Understanding the structure, composition, and function of glycoconjugates is essential for elucidating their roles in physiology and pathology. Glycoconjugate analysis techniques, such as glycan profiling, glycosylation site mapping, and glycan structural analysis, are employed to characterize these molecules and uncover their biological significance.
Glycoconjugate Analysis Services by Creative Proteomics
Glycan Profiling
Creative Proteomics offers comprehensive glycan profiling services to characterize the diverse glycan structures present in glycoconjugates. Utilizing advanced analytical techniques such as liquid chromatography-mass spectrometry (LC-MS) and capillary electrophoresis (CE), our experts identify and quantify glycans with high sensitivity and accuracy. Through glycan profiling, clients gain insights into the glycan composition, linkage patterns, and heterogeneity of glycoconjugates, facilitating biomarker discovery, quality assessment, and glycoengineering strategies.
Glycosylation Site Mapping
Determining the glycosylation sites within glycoproteins is essential for understanding their structure-function relationships and biological activities. Creative Proteomics offers state-of-the-art glycosylation site mapping services using enzymatic digestion coupled with LC-MS/MS analysis. By selectively cleaving glycan moieties and identifying glycopeptides, we precisely locate glycosylation sites within protein sequences. Our comprehensive approach enables accurate characterization of glycoprotein glycosylation, aiding in drug development, biopharmaceutical analysis, and protein engineering endeavors.
Glycan Structural Analysis
Characterizing the structural features of glycans is crucial for elucidating their biological roles and therapeutic potentials. Creative Proteomics provides advanced glycan structural analysis services utilizing a combination of enzymatic digestion, chromatographic separation, and mass spectrometry techniques. By determining monosaccharide composition, glycosidic linkages, and branching patterns, we elucidate the detailed structures of glycans present in glycoconjugates. Our in-depth structural analysis facilitates the identification of disease-associated glycan biomarkers, development of glycan-based therapeutics, and optimization of glycoprotein expression systems.
Glycoconjugate Interaction Studies
Investigating the interactions between glycoconjugates and their binding partners is crucial for understanding molecular recognition events and designing targeted therapies. Creative Proteomics offers comprehensive glycoconjugate interaction studies using surface plasmon resonance (SPR), isothermal titration calorimetry (ITC), and other biophysical techniques. By assessing the binding kinetics, affinity, and specificity of glycan-protein interactions, we elucidate the molecular mechanisms underlying biological processes such as cell adhesion, immune recognition, and pathogen invasion. Our expertise in glycoconjugate interaction analysis facilitates drug discovery, vaccine development, and diagnostic assay design.
List of Glycoconjugates We Can Analyze
Protein Glycoconjugates:
| Glycoproteins | Glycopeptides | Glycosylated Enzymes |
| Mucins | Immunoglobulins | Glycosylated Hormones |
| Blood Proteins | Serum Glycoproteins | Extracellular Matrix Proteins |
| Cell Surface Receptors | Interferons | Growth Factors |
Lipid Glycoconjugates:
| Glycolipids | Glycosylated Lipoproteins | Glycosylated Phospholipids |
| Sphingolipids | Lipopolysaccharides (LPS) | Glycophosphatidylinositols |
Nucleic Acid Glycoconjugates:
| Glycosylated DNA | Glycosylated RNA | GlycoNucleotides |
| Modified Nucleosides | RNA Cap Structures |
Other Glycoconjugates:
| Proteoglycans | Glycosaminoglycans (GAGs) | Bacterial Capsular Polysaccharides |
| Glycosides | Peptidoglycan | Glycogen |
Additional Analysis Services:
Technical Platforms for Glycoconjugate Analysis
Liquid Chromatography (LC): LC systems, such as high-performance liquid chromatography (HPLC) or ultra-high-performance liquid chromatography (UHPLC), are employed for separating glycoconjugates or their components based on differences in polarity, size, or charge.
Mass Spectrometry (MS): Mass spectrometry instruments, including electrospray ionization mass spectrometry (ESI-MS) or matrix-assisted laser desorption/ionization mass spectrometry (MALDI-MS), are crucial for detecting and characterizing glycoconjugates based on their mass-to-charge ratio.
Capillary Electrophoresis (CE): Capillary electrophoresis systems, such as capillary zone electrophoresis (CZE) or capillary isoelectric focusing (CIEF), enable the separation and analysis of glycoconjugates based on their charge and size.
High-Performance Anion Exchange Chromatography (HPAEC): HPAEC systems are specialized chromatography platforms used for the analysis of carbohydrates, including the glycan portions of glycoconjugates.
Nuclear Magnetic Resonance (NMR) Spectroscopy: NMR spectroscopy instruments provide valuable structural information about glycoconjugates by analyzing their nuclear magnetic resonance properties.
Glycan Array Technology: Glycan array platforms facilitate high-throughput screening of glycan-protein interactions by immobilizing diverse glycan structures on a solid support and probing their interactions with biomolecules.
Surface Plasmon Resonance (SPR) Spectroscopy: SPR instruments enable real-time monitoring of biomolecular interactions, including glycan-protein interactions, by measuring changes in refractive index upon binding.
Isothermal Titration Calorimetry (ITC): ITC instruments measure heat changes accompanying biomolecular interactions, providing insights into the thermodynamics of glycan-protein binding events.
Enzymatic Assays: Various enzymatic assays, utilizing glycosidases, glycosyltransferases, or glycan-binding proteins, are employed for specific glycoconjugate analysis tasks, such as glycan profiling or glycosylation site mapping.
Sample Requirements for Glycoconjugate Analysis
| Sample Type | Sample Amount Recommended |
|---|---|
| Serum/Plasma | 100-200 μL |
| Urine | 200-500 μL |
| Tissue Homogenate | 20-50 mg |
| Cell Culture | 1-5 x 106 cells |
| Saliva | 100-200 μL |
| Cerebrospinal Fluid (CSF) | 100-200 μL |
Applications of Glycoconjugate Analysis
Biomedical Research: Essential for understanding glycoconjugates' roles in cellular processes, immunity, and disease mechanisms.
Disease Biomarkers: Identifying glycan alterations linked to diseases, aiding in early diagnosis and monitoring.
Vaccine Development: Crucial for characterizing glycan antigens in vaccines, ensuring immunogenicity and efficacy.
Drug Development: Integral for characterizing glycosylated therapeutics, ensuring product consistency and efficacy.
Glycomics and Glycoproteomics: Used for comprehensive profiling of glycan and glycoprotein repertoires, advancing glycoscience research.
Glycoengineering: Facilitates modification of glycan structures to improve therapeutic properties or biological functions.
Food and Nutrition: Applied to study glycosylation of food components, detect contaminants, and assess nutritional value.
Title: Comparative Glycan Analysis of hGGT1 Using ALSA Platform
Background
Glycosylation plays a crucial role in modulating the function and stability of proteins, including human gamma-glutamyl transferase 1 (hGGT1), which is involved in various physiological and pathological processes. Understanding the glycan composition of hGGT1 from different sources can provide valuable insights into disease mechanisms and potential diagnostic markers.
Sample
The study utilized hGGT1 samples derived from normal human kidney and liver tissues, as well as hGGT1 expressed in Pichia pastoris, a yeast system known for producing high-mannose type N-glycans. These samples represent distinct glycosylation profiles reflecting physiological and pathological conditions.
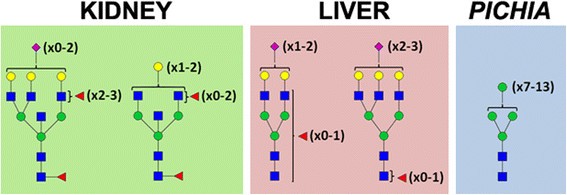 Source-specific composition of N -glycans on human GGT1 (hGGT1).
Source-specific composition of N -glycans on human GGT1 (hGGT1).
Technical Methods
Biotinylated MVN Development: Microcystis aeruginosa lectin (MVN) was biotinylated to specifically interact with high-mannose type N-glycans, particularly the Manα(1–2)Man disaccharide motif.
ALSA Platform Implementation: The Antibody-Lectin Sandwich Assay (ALSA) platform was employed to miniaturize and compare glycoanalyses of hGGT1 from different sources. This involved immobilizing a polyclonal antibody against hGGT1 on a solid support and subsequently probing with lectins to detect glycan differences.
Lectin Binding Pattern Analysis: Various lectins, including MVN, Concanavalin A (ConA), Galanthus nivalis lectin (GNL), Euonymus europaeus lectin (EEL), Griffonia simplicifolia Lectin 1 (GSL-1), Datura stramonium lectin (DSL), and Phaseolus vulgaris-Erythroagglutinin (Pha-E), were utilized to probe for specific glycan motifs.
Validation Through Western Blot: The ALSA results were validated using Western blot analysis of immunoprecipitated hGGT1 samples, confirming the lectin binding patterns observed.
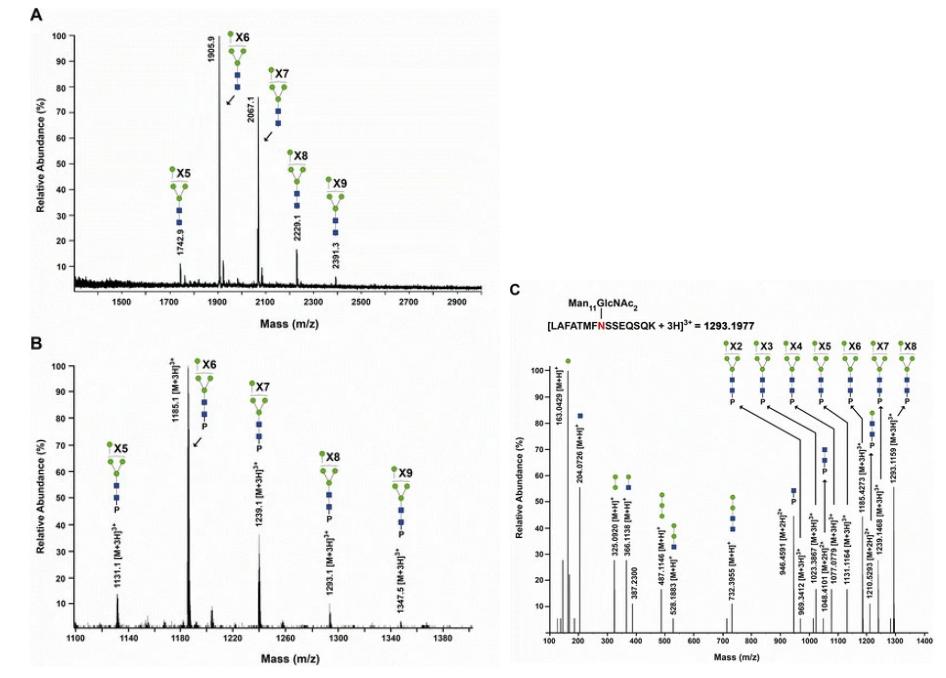 Glycoconjugate analysis of hGGT1 expressed in Pichia pastoris.
Glycoconjugate analysis of hGGT1 expressed in Pichia pastoris.
Results
The ALSA platform successfully identified differential lectin binding patterns on hGGT1 from different sources. MVN exhibited preferential binding to Pichia pastoris-expressed hGGT1 with high-mannose type N-glycans, while other lectins like Pha-E and DSL showed specificity for kidney- and liver-derived hGGT1, respectively. These findings were consistent with known glycan compositions and were validated through Western blot analysis.
Reference
- West, Matthew B., et al. "Detection of distinct glycosylation patterns on human γ-glutamyl transpeptidase 1 using antibody-lectin sandwich array (ALSA) technology." BMC biotechnology 14 (2014): 1-16.




