- Service Details
- Case Study
What is Oligosaccharide Population Analysis?
Oligosaccharide population analysis refers to the study and characterization of the composition, structure, and distribution of oligosaccharides within a given biological or chemical sample. Oligosaccharides are short chains of sugar molecules, typically composed of 3 to 20 monosaccharide units. Analyzing the population of oligosaccharides is important in various fields such as glycobiology, biochemistry, and pharmaceutical research.
This analysis involves techniques such as chromatography and mass spectrometry. Researchers aim to understand the diversity of oligosaccharide structures present in a sample, identify specific oligosaccharide species, and determine their relative abundance.
Oligosaccharide population analysis is particularly relevant in the study of glycoproteins, glycolipids, and other glycoconjugates, as oligosaccharides play crucial roles in various biological processes, including cell signaling, immune response, and protein folding. By gaining insights into the oligosaccharide composition, researchers can better understand the functional implications of these molecules in biological systems.
In the field of drug development, oligosaccharide population analysis reveals the overall profile of N-glycans or O-glycans present in glycoproteins. It is crucial at different stages of drug development and allows a detailed examination of the glycan profile. In addition, the analysis ensures consistency between batches during the manufacturing process.
In addition to manufacturing applications, oligosaccharide population analysis plays a key role in identifying aberrant glycosylation profiles, helping to ensure the safety and efficacy of biopharmaceuticals; it also detects potentially immunogenic glycan structures, helping to improve the overall quality and safety standards of pharmaceuticals.
Technology Platform for Oligosaccharide Population Analysis
Our oligosaccharide population analysis service employs a sophisticated technology platform, utilizing precise techniques to unravel glycans' intricacies. The analysis includes:
Matrix Assisted Laser Desorption Ionization Mass Spectrometry (MALDI-MS): Provides molecular ion data for a precise understanding of glycan mass-to-charge ratios.
Electrospray Mass Spectrometry (ES-MS): Offers fragment ion information, enhancing analysis by providing insights into ionization-produced fragments.
High Performance Anion Exchange Chromatography with Pulsed Amperometric Detection (HPAEC-PAD): Utilizes chromatographic profiles for the separation and detection of glycan species based on charge.
Hydrophilic Interaction Liquid Chromatography with Fluorescence Detection (HILIC-FLD): Enhances precision with fluorescence detection, contributing to a comprehensive understanding of the oligosaccharide population.
List of Detectable Oligosaccharide
| Maltose | Fructooligosaccharides (FOS) | Galactooligosaccharides (GOS) | Xylooligosaccharides (XOS) | Mannooligosaccharides (MOS) |
| Isomaltooligosaccharides (IMO) | Chitooligosaccharides (COS) | Manno-oligosaccharides (MnOS) | Xylitol oligosaccharides | Oligofructose |
| Stachyose | Raffinose | Gluco-oligosaccharides (GlcOS) | Laminarioligosaccharides | Chondroitin oligosaccharides |
| Pectic oligosaccharides | Neooligosaccharides (NOS) | Arabinooligosaccharides (AOS) |
Applications of Oligosaccharide Population Analysis
Pharmaceutical Development:
- Screening Glycoproteins: Meticulous differentiation of glycoproteins from various cell lines.
- Biosimilar Product Development: Guiding optimization and creation of biosimilar pharmaceuticals.
- Immunogenicity Assessment: Critical detection of potentially immunogenic glycan structures.
- Consistency Confirmation: Ensuring uniformity between batches for regulatory compliance.
Biotechnology and Enzyme Engineering:
- Optimization of biotechnological processes through understanding and manipulation of glycan structures.
- Enhancing efficiency in enzyme engineering applications.
Clinical Research:
- Exploring glycan variations in biological samples for a deeper understanding of disease mechanisms.
- Identifying potential glycan-based biomarkers in clinical research.
Food and Beverages Industry:
- Unveiling glycan profiles to ensure the quality and authenticity of food products.
- Contributing to quality control and regulatory compliance in the food and beverages sector.
Environmental Monitoring:
- Studying glycan compositions in environmental samples for insights into ecological changes.
- Contributing to environmental monitoring and identifying potential contaminants.
Sample Requirements for Oligosaccharide Population Analysis
| Sample Type | Sample Size Recommendations |
|---|---|
| Biological Samples | |
| Blood (Whole Blood) | 100-200 μL |
| Serum/Plasma | 100-200 μL |
| Biopsy Specimens | 20-50 mg |
| Tissue Homogenates | 20-50 mg |
| Cell Culture Supernatants | 5-10 mL |
| Cell Lysates | 20-50 mg |
| Cerebrospinal Fluid (CSF) | 50-100 μL |
| Feces | 50-100 mg |
| Milk | 50-100 μL |
| Plant Extracts | 20-50 mg |
| Saliva | 100-200 μL |
| Synovial Fluid | 50-100 μL |
| Urine | 50-100 μL |
| Miscellaneous | |
| Biopharmaceutical Products | Consult with our experts |
| Synthesized Compounds | Consult with our experts |
| Environmental Samples | |
| Environmental Samples | Varies based on source and complexity |
Case. Analytical Methods for Human Milk Oligosaccharides (HMOs)
Background:
Human Milk Oligosaccharides (HMOs) are complex carbohydrates found in human breast milk, playing a crucial role in infant health and development. Analyzing and characterizing these biomolecules is essential for understanding their structure-function relationships and developing high-quality infant formulas.
Samples:
The analysis focused on various samples, primarily human milk obtained from different donors and, in some cases, milk from domestic animals. The goal was to determine the composition and variations in HMO expression.
Technical Method:
Several analytical methods were employed for the characterization of HMOs:
High-Performance Liquid Chromatography (HPLC):
- Reversed-phase Chromatography (RPC): Used for analyzing neutral oligosaccharides in milk after pre-column derivatization. It showed sensitivity but involved time-consuming sample preparation.
- High-pH Anion-exchange Chromatography (HPAEC): Initially used for quantitative determination, but its significance decreased over time due to emerging techniques like porous graphitic carbon and hydrophilic interaction chromatography.
- Porous Graphitic Carbon (PGC) Chromatography: Showed improved separation of isomeric HMO species, providing high resolution for both neutral and acidic forms.
Gas Chromatography (GC): A novel GC–MS method was developed for the separation and quantification of neutral isomeric HMOs, demonstrating potential advantages in terms of peak capacities and low vapor pressure.
Electromigration Techniques: Capillary Electrophoresis (CE): Applied for both quantitative determination and investigating interactions with carbohydrate-binding molecules. CE-LIF-MSn analysis showed promise for profiling and structural characterization.
Results:
The studies yielded valuable insights into HMO composition, variations between donors, and differences in HMO profiles among populations. The use of advanced chromatographic and electrophoretic techniques provided improved sensitivity, resolution, and efficiency in the analysis of HMOs. The methodologies outlined in the review serve as a comprehensive guide for analysts dealing with the challenges of working with these complex biomolecules, offering both established and novel approaches for HMO characterization.
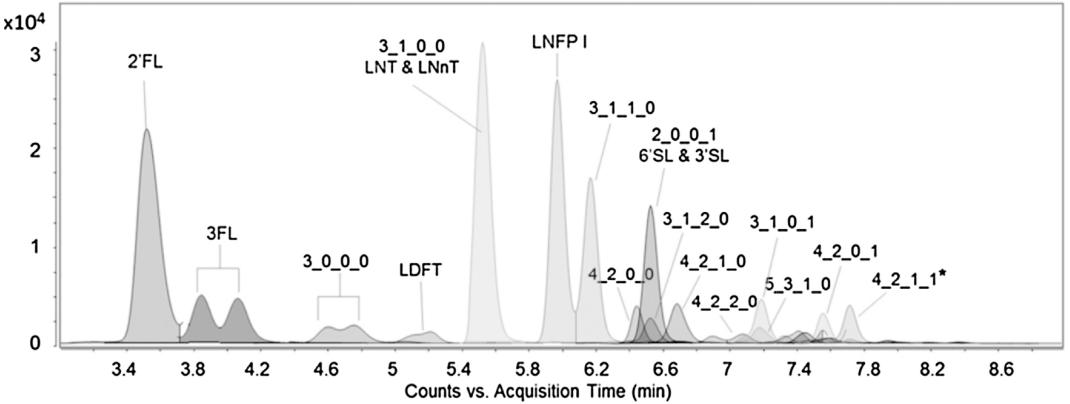 The extracted dynamic MRM chromatogram monitored for a pooled human milk sample
The extracted dynamic MRM chromatogram monitored for a pooled human milk sample
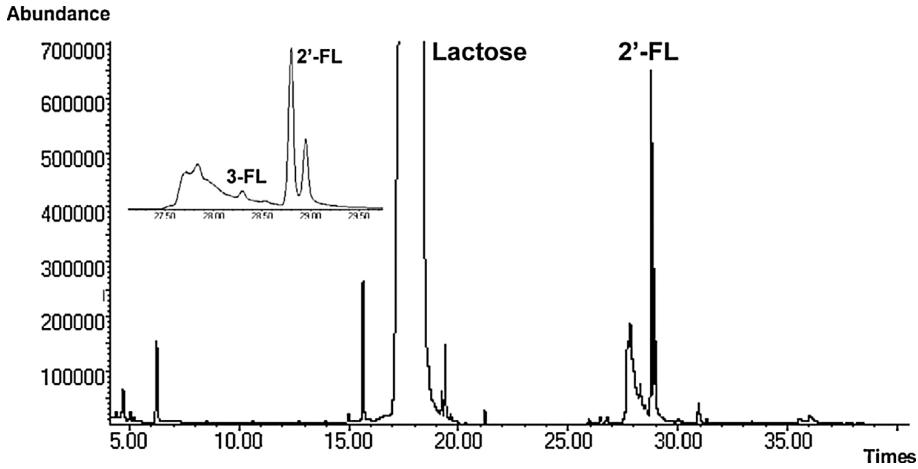 GC–MS TIC trace of a derivatized human milk sample.
GC–MS TIC trace of a derivatized human milk sample.
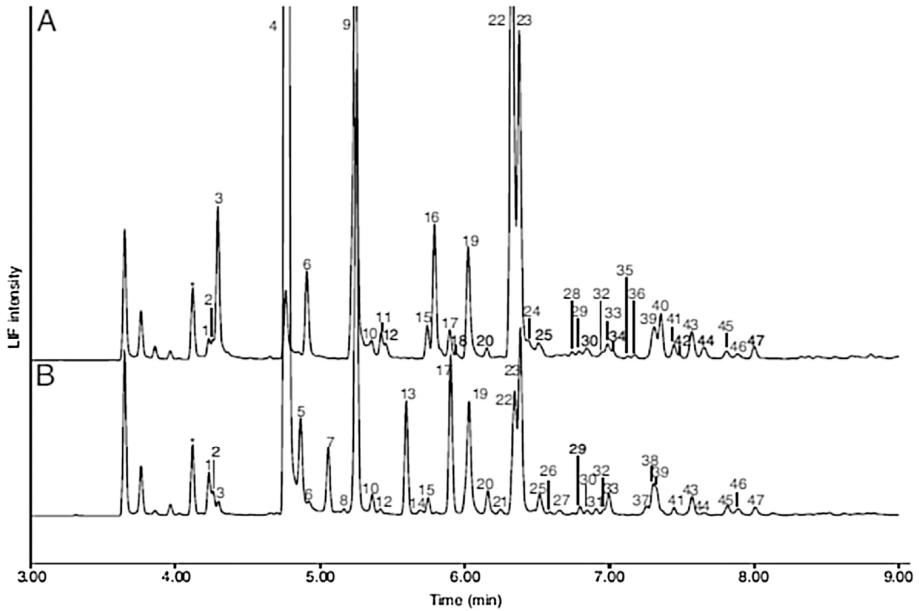 Off-line CE-LIF electropherograms of APTS-derivatized HMOs extracted from (A) feces of a breast-fed baby and (B) breast milk.
Off-line CE-LIF electropherograms of APTS-derivatized HMOs extracted from (A) feces of a breast-fed baby and (B) breast milk.
Reference
- Grabarics, Márkó, et al. "Analytical characterization of human milk oligosaccharides–potential applications in pharmaceutical analysis." Journal of Pharmaceutical and Biomedical Analysis 146 (2017): 168-178.




