- Service Details
- Case Study
What is Glycan Separation and Purification?
Glycan separation and purification are integral processes in glycobiology, addressing the complex nature of glycans, which are carbohydrate structures linked to proteins or lipids. The significance of these processes lies in the intricate heterogeneity of glycans, where variations in structure and composition play crucial roles in biological functions.
Glycans are involved in essential cellular processes such as cell signaling, immune response, and protein stability. However, their diverse and heterogeneous nature poses challenges for comprehensive analysis. Glycan separation and purification serve as crucial preparative steps, allowing researchers to isolate specific glycan structures from complex mixtures.
In the context of biomedical research, understanding the glycome—the complete set of glycans in an organism or a specific cell type—can provide insights into disease mechanisms, biomarker discovery, and therapeutic development. Glycan separation and purification enable researchers to obtain pure glycan samples, facilitating detailed analysis that contribute to our understanding of glycan structures and their functional roles.
These processes are particularly relevant in the study of glycoproteins, where the identification and characterization of glycans are essential for unraveling the complexity of cellular interactions and molecular mechanisms. The ability to separate and purify glycans enhances the precision and accuracy of downstream analytical techniques, ultimately advancing our knowledge of glycan-mediated biological processes.
Glycan Separation and Purification Services at Creative Proteomics
At Creative Proteomics, our glycan separation and purification services are designed to meet the demands of researchers and industries requiring precise and reliable glycan characterization. We offer the following technology platforms:
Chromatography:
- Size Exclusion Chromatography (SEC): Separates glycans based on size, with larger molecules eluting earlier.
- Ion Exchange Chromatography (IEC): Exploits charge differences for glycan separation.
- Affinity Chromatography: Utilizes specific interactions between glycans and affinity ligands.
- Reversed Phase Liquid Chromatography (RPLC): Separates glycans based on hydrophobicity.
- Hydrophilic Interaction Chromatography (HILIC): Utilizes polarity differences for glycan separation.
- Porous Graphitized Carbon (PGC): Offers a unique medium for glycan separation.
Electrophoresis:
- Capillary Electrophoresis (CE): Separates glycans based on electrophoretic mobility.
Mass Spectrometry (MS):
- Matrix-Assisted Laser Desorption/Ionization (MALDI) MS: Provides mass information for glycan identification.
- Electrospray Ionization (ESI) MS: Ionizes glycans for mass analysis.
Precipitation and Filtration:
- Ethanol Precipitation: Precipitates glycans for purification.
- Filtration: Uses size-based filtration for glycan separation.
Enzymatic Digestion:
- Glycanase Treatment: Enzymatic cleavage for glycan purification and analysis.
Lectin Enrichment:
- Lectin Enrichment: Exploits the specific binding of lectins to glycans for enrichment.
Centrifugation:
- Ultracentrifugation: Separates glycans based on density.
Sample Requirements for Glycan Separation and Purification
| Sample Type | Recommended Sample Amount |
|---|---|
| Serum or Plasma | 100-500 µL |
| Cell Culture Supernatants | 1-5 mL |
| Tissue Homogenates | 20-100 mg |
| Glycoprotein Solutions | 100-500 µg |
| Glycan Mixtures or Standards | 50-200 µg |
| Other Biological Samples | Varies |
Applications of Glycan Separation and Purification
Biomedical Research:
- Disease Biomarker Discovery: Identifying specific glycan biomarkers associated with diseases for early diagnosis and prognosis.
- Immunology: Understanding glycan profiles of antibodies and immune cells for research on immune responses and vaccine development.
Pharmaceutical Development:
- Biopharmaceuticals: Characterizing glycosylation patterns in therapeutic proteins for product quality and efficacy.
- Drug Development: Investigating glycan structures of cell surface receptors and signaling molecules to inform drug design.
Glycoengineering:
- Bioprocessing: Controlling and optimizing glycan structures in biopharmaceutical production for enhanced protein stability and bioactivity.
- Cell Line Development: Analyzing glycan structures to select cell lines with desired glycosylation patterns in bioproduction.
Food and Nutrition:
- Quality Control: Ensuring the quality and authenticity of food products, particularly in the dairy and beverage industries.
- Nutritional Studies: Investigating glycan structures in food products to understand their nutritional value and potential health benefits.
Glycan Biomaterials:
- Glycan Arrays: Creating glycan arrays for studying interactions with proteins, cells, and pathogens.
- Biosensors: Using glycan-modified surfaces in biosensors for detecting specific biomolecules with high sensitivity.
Glycan Profiling in Diseases:
- Cancer Research: Characterizing aberrant glycosylation in cancer cells for understanding tumor progression and developing targeted therapies.
- Infectious Diseases: Studying host-pathogen interactions and developing antiviral strategies through glycan analysis.
Structural Biology:
- Protein-Glycan Interactions: Investigating the role of glycans in protein folding, stability, and interactions for insights into structural biology.
- NMR and Mass Spectrometry: Conducting high-resolution structural analysis using techniques like NMR and mass spectrometry with purified glycans.
Environmental and Forensic Sciences:
- Biomarker Identification: Using glycan analysis of environmental samples to identify glycan-based biomarkers for pollution and exposure studies.
- Forensic Analysis: Applying glycan profiling for forensic analysis, such as the identification of biological samples.
Case. Glycan Separation Strategies in Analytical Chemistry - Exploring Reversed-Phase Chromatography and Beyond
Background:
Efficient glycan separation is vital for glycomics research, prompting the exploration of diverse chromatographic techniques. Reversed-phase chromatography stands out as a common method, but challenges arise from the selection of labeling reagents, experimental conditions, and sample complexities.
Samples
The discussion encompasses native and labeled glycans, focusing on hydrophobicity and structural attributes. Sample complexity ranges from simple glycan standards to intricate biological samples, influencing the success of glycan separation.
Technical Method
Extensively investigated as a glycan separation technique, reversed-phase chromatography is examined. Various labeling reagents, including PA, AB, ABEE, ABBE, HOA, AMAC, and PMP, are explored for their impact on hydrophobicity and separation efficiency. The text elucidates the influence of structural features like sialic acids, fucoses, and bisecting GlcNAc on retention. Additionally, alternative chromatographic methods, such as hydrophilic interaction chromatography (HILIC) and porous graphitized carbon (PGC) chromatography, are highlighted for improved separation, especially of glycan isomers.
Results
The results emphasize the significant impact of labeling tag hydrophobicity on glycan retention and elution order. PA labeling offers structural insights but may complicate direct coupling with ESI-MS. Different hydrophobic labels exhibit varied elution orders, influenced by factors like ion-pairing agents, column types, gradients, and eluent compositions. The presence of sialic acids and fucoses substantially affects retention, with linkage and structure specificity playing a crucial role. Sample complexity influences peak capacity, and longer run times or two-dimensional separations are suggested for improved resolution. While reversed-phase chromatography is commonly employed, HILIC and PGC chromatography are considered superior in separating glycan species and isomers, albeit with lower robustness and reproducibility.
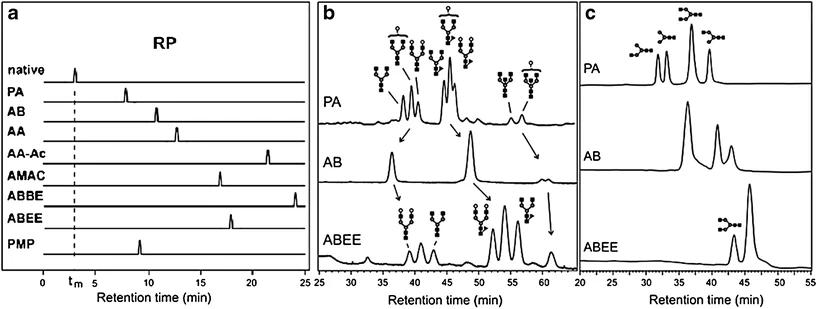 a Overview of the chromatograms of various reducing end derivatized N-glycans and a native glycan that is eluted at the void volume (dashed line). b Reversed-phase chromatograms of desialylated immunoglobulin G N-glycans derivatized with PA, AB, or ABEE. c Chromatograms of the separation of a mixture of four glycans.
a Overview of the chromatograms of various reducing end derivatized N-glycans and a native glycan that is eluted at the void volume (dashed line). b Reversed-phase chromatograms of desialylated immunoglobulin G N-glycans derivatized with PA, AB, or ABEE. c Chromatograms of the separation of a mixture of four glycans.
Reference
- Vreeker, Gerda CM, and Manfred Wuhrer. "Reversed-phase separation methods for glycan analysis." Analytical and bioanalytical chemistry 409 (2017): 359-378.




