- Service Details
- Case Study
Glycoproteins, characterized by the presence of glycans attached to peptide backbones, play crucial roles in numerous biological processes, including cell-cell recognition, immune response modulation, and protein folding. However, the structural diversity and heterogeneity of glycans present significant challenges for traditional analytical techniques. Glycopeptide mass spectrometry quantitative analysis addresses these challenges by offering high sensitivity, specificity, and comprehensive characterization of glycoproteins.
By leveraging advanced mass spectrometry techniques coupled with sophisticated data analysis algorithms, glycopeptide mass spectrometry quantitative analysis enables:
- Precise Quantification: Accurate measurement of glycopeptide abundance facilitates quantitative assessment of protein glycosylation patterns, aiding in the elucidation of glycan biosynthesis pathways and disease-related glycosylation changes.
- Structural Elucidation: Through fragmentation analysis, glycopeptide mass spectrometry provides detailed structural information about glycans, including glycan composition, linkage patterns, and positional isomer identification, thereby facilitating the characterization of glycoprotein structures.
- Biomarker Discovery: The ability to detect and quantify glycopeptides with high sensitivity makes glycopeptide mass spectrometry a powerful tool for biomarker discovery and validation in various diseases, including cancer, autoimmune disorders, and infectious diseases.
Creative Proteomics' Glycopeptide Mass Spectrometry Quantitative Analysis Services
Creative Proteomics offers a comprehensive suite of glycopeptide mass spectrometry analysis services tailored to meet the diverse needs of researchers across academia, biotechnology, and pharmaceutical industries. Our services include:
- Glycopeptide Quantification: Utilizing state-of-the-art mass spectrometry platforms, we offer accurate and reproducible quantification of glycopeptides, enabling quantitative comparison of glycosylation profiles across different biological samples.
- Glycan Structure Analysis: Our expert analysts employ advanced fragmentation techniques and bioinformatics tools to elucidate the detailed structure of glycans attached to peptide backbones, providing invaluable insights into glycoprotein composition and modification.
- Site-specific Glycosylation Profiling: Creative Proteomics specializes in site-specific glycosylation analysis, allowing precise mapping of glycosylation sites on target proteins and determination of site-specific glycan heterogeneity.
- Biomarker Discovery and Validation: Leveraging our extensive experience in biomarker research, we offer comprehensive solutions for biomarker discovery and validation using glycopeptide mass spectrometry, empowering clients to identify novel glycoprotein biomarkers for diagnostic and therapeutic applications.
Technical Platforms for Glycopeptide Mass Spectrometry Analysis
Enrichment Strategies: Techniques such as lectin affinity chromatography, hydrazide chemistry, and hydrophilic interaction liquid chromatography (HILIC) have been optimized to isolate glycopeptides from complex biological samples.
Liquid Chromatography Mass Spectrometry (LC-MS): LC-MS systems equipped with high-resolution mass spectrometers enable sensitive detection and accurate quantification of glycopeptides in complex biological samples.
Tandem Mass Spectrometry (MS/MS): By performing sequential fragmentation of glycopeptides, MS/MS analysis facilitates the elucidation of glycan structures and the identification of glycopeptide amino acid sequences.
Data Analysis Software: We utilize advanced data analysis software packages tailored for glycopeptide analysis, allowing for automated peak detection, glycan composition prediction, and comprehensive data interpretation.
Bioinformatics Tools: Our bioinformatics experts develop custom algorithms and databases for glycopeptide data analysis, enabling in-depth characterization of glycoprotein structures and glycosylation patterns.
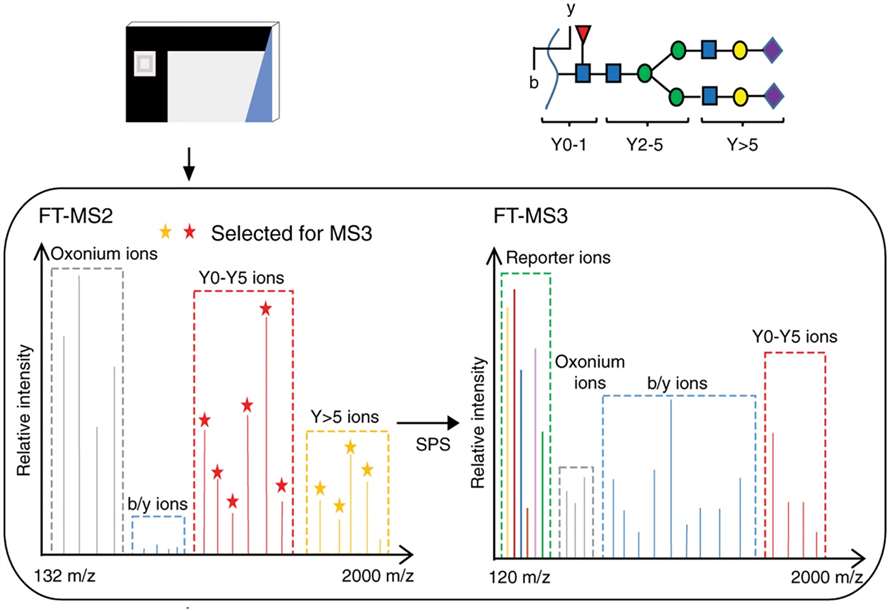 Quantitation of glycopeptides with TMT labeling by multi-notch MS3 acquisition on a Orbitrap Fusion Lumos mass spectrometer. Reprinted with permission from Fang et al. (2020).
Quantitation of glycopeptides with TMT labeling by multi-notch MS3 acquisition on a Orbitrap Fusion Lumos mass spectrometer. Reprinted with permission from Fang et al. (2020).
Sample Requirements for Glycopeptide Quantitative Analysis
| Sample Type | Sample Quantity | Application |
|---|---|---|
| Cell Culture | 1-10 million cells | Quantitative analysis of cellular glycoproteins |
| Tissue Biopsy | 5-50 mg | Profiling glycoprotein expression in tissues |
| Serum/Plasma | 100-500 µL | Biomarker discovery and validation |
| Urine | 1-5 mL | Identification of urinary glycoprotein biomarkers |
| Cerebrospinal Fluid | 100-500 µL | Detection of glycoprotein biomarkers in neurological disorders |
Guidelines for Sample Collection and Handling
- Cell Culture: Harvest cells using standard protocols, wash with PBS, and lyse cells in suitable lysis buffer. Avoid freeze-thaw cycles to maintain glycopeptide integrity.
- Tissue Biopsy: Snap-freeze tissue samples immediately after collection and store at -80°C until further processing. Homogenize tissues in appropriate lysis buffer to extract glycoproteins.
- Serum/Plasma: Collect blood samples in EDTA or heparin tubes to prevent coagulation. Centrifuge samples at 1000-2000 × g for 10 minutes to obtain cell-free serum or plasma.
- Urine: Collect urine samples in sterile containers and centrifuge at 1000-2000 × g for 10 minutes to remove cellular debris. Store supernatant at -80°C until analysis.
- Cerebrospinal Fluid: Collect CSF samples via lumbar puncture using aseptic techniques. Centrifuge samples at low speed to remove cells and debris, then aliquot and store at -80°C.
Quality Control Measures
To ensure reliable results, adhere to the following quality control measures:
- Maintain sample integrity during collection, handling, and storage to prevent degradation of glycopeptides.
- Include appropriate internal standards for quantification and normalization of glycopeptide abundance.
- Perform replicate analyses to assess measurement variability and ensure data reproducibility.
- Validate analytical methods using standard reference materials to verify accuracy and precision.
Applications of Glycopeptide Mass Spectrometry Quantitative Analysis
Biomedical Research: Glycopeptide mass spectrometry is used to study protein glycosylation patterns, which play crucial roles in various biological processes such as cell signaling, immune response, and disease progression. Quantitative analysis helps in understanding the dynamics of protein glycosylation in health and disease states.
Clinical Diagnostics: Glycoproteins are important biomarkers for various diseases including cancer, diabetes, and autoimmune disorders. Quantitative analysis of glycopeptides can aid in the discovery and validation of novel glycoprotein biomarkers for early disease detection, prognosis, and monitoring treatment response.
Pharmaceutical Development: Glycosylation is a critical quality attribute for therapeutic proteins such as monoclonal antibodies, which can affect their efficacy, stability, and immunogenicity. Quantitative analysis of glycopeptides is essential for characterizing and monitoring the glycosylation profiles of therapeutic proteins during the development and manufacturing process to ensure product quality and consistency.
Glycoprotein Engineering: Glycopeptide mass spectrometry quantitative analysis is used to study the effects of glycosylation on protein structure and function. This information can be applied to engineer glycoproteins with desired glycosylation patterns for improved therapeutic efficacy and pharmacokinetics.
Food and Environmental Science: Glycoproteins are present in various food products and environmental samples. Quantitative analysis of glycopeptides can be used to assess the glycosylation profiles of food proteins, detect food adulteration, and monitor environmental contaminants.
Title: Quantitative Analysis of Glycopeptides: Techniques and Applications
Background
Glycopeptides, characterized by the presence of glycans attached to peptide backbones, play crucial roles in various biological processes and are potential biomarkers for diseases. However, their quantitative analysis poses significant challenges due to the complexity of glycan structures and the lack of standardization. Several analytical techniques have been developed to address these challenges and enable accurate quantification of glycopeptides.
Sample
The samples used for quantitative analysis of glycopeptides typically include biological fluids such as serum or plasma, where glycopeptides are derived from glycoproteins. These samples are enriched for glycopeptides using techniques such as lectin affinity chromatography or solid-phase extraction before analysis.
Technical Methods
Multiple Reaction Monitoring (MRM) Approach: MRM involves selecting specific precursor and fragment ions for quantification. It requires prior knowledge of precursor-transition ion pairs, optimal collision energies (CE), and retention times. MRM has been applied to quantify both truncated and intact glycopeptides, with optimizations to enhance sensitivity and specificity.
Parallel Reaction Monitoring (PRM) Approach: PRM selects precursor ions for fragmentation and scans all possible fragment ions using high-resolution mass analyzers. It simplifies method setup compared to MRM and offers enhanced specificity. PRM has been utilized for quantifying truncated and intact glycopeptides, with developments in data processing pipelines to improve throughput and accuracy.
Advances in Methodologies: Recent developments aim to enhance the throughput and sensitivity of glycopeptide quantitation. Dynamic MRM and Scheduled MRM modes allow for narrower monitoring windows and automatic dwell time assignment, improving efficiency. Additionally, PRM methodologies have been optimized for enhanced sensitivity through dynamic control of ion accumulation time and scan speed.
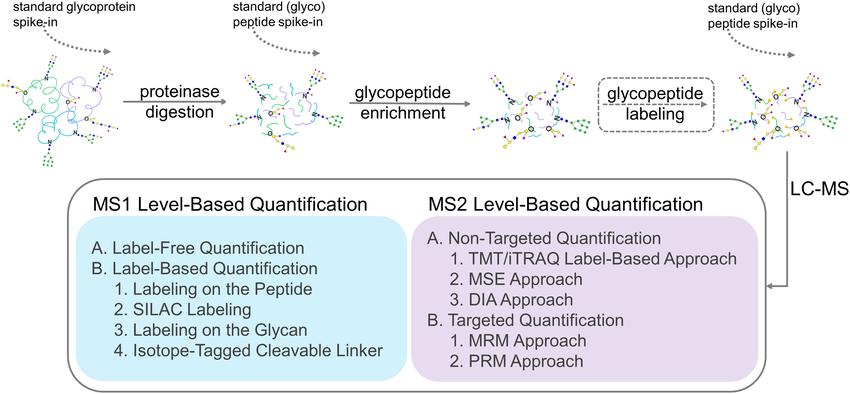 Strategies for glycopeptide quantification by mass spectrometry.
Strategies for glycopeptide quantification by mass spectrometry.
Results
Quantitative analysis of glycopeptides using MRM and PRM approaches has led to significant advancements in understanding glycoprotein dynamics in various biological contexts. These techniques have enabled the identification of glycopeptide biomarkers for diseases such as liver fibrosis and cancer, with improved sensitivity and specificity. Moreover, the development of calibration curves using glycopeptide standards has facilitated absolute quantitation, enhancing the reliability of quantitative glycopeptide analysis.
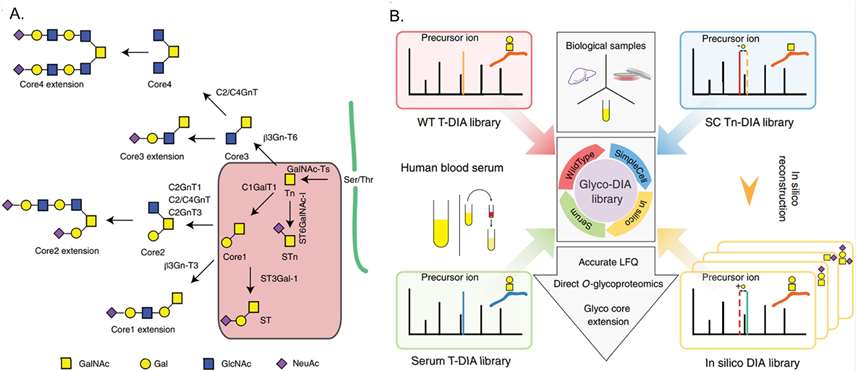 Library construction in DIA approach for O-glycopeptide quantitation using a silico-boosted glycopeptide library.
Library construction in DIA approach for O-glycopeptide quantitation using a silico-boosted glycopeptide library.
Reference
- Yin, Haidi, and Jianhui Zhu. "Methods for quantification of glycopeptides by liquid separation and mass spectrometry." Mass spectrometry reviews 42.2 (2023): 887-917.




