- Service Details
- Case Study
What is Glycosylated Antibody?
Glycosylated antibodies are a subclass of monoclonal antibodies characterized by the presence of complex carbohydrate chains attached to their protein backbone. These carbohydrate moieties, known as glycans, play crucial roles in modulating the antibody's structure, stability, and biological activity. Glycosylation can occur at specific sites within the antibody's Fc region, impacting effector functions such as antibody-dependent cellular cytotoxicity (ADCC) and complement-dependent cytotoxicity (CDC).
The analysis of glycosylated antibodies is indispensable for elucidating their structure-function relationships and optimizing their therapeutic efficacy. Variations in glycan composition, such as changes in glycan branching, sialylation, or fucosylation, can profoundly influence the antibody's pharmacokinetics, immunogenicity, and interactions with cellular receptors. Therefore, comprehensive characterization of glycosylation patterns is essential for ensuring the safety, potency, and consistency of glycosylated antibodies -based therapeutics.
Glycosylated Antibody Analysis Services by Creative Proteomics
Glycan Profiling
Our glycan profiling services employ advanced analytical techniques to elucidate the composition, structure, and heterogeneity of glycans attached to glycosylated antibodies. Through high-resolution mass spectrometry and liquid chromatography methods, we accurately identify and quantify glycan species, including complex and high-mannose glycans, facilitating comprehensive glycosylation analysis.
Glycopeptide Mapping
Creative Proteomics employs cutting-edge proteomic approaches to perform glycopeptide mapping of glycosylated antibodies. By enzymatically digesting antibodies into glycopeptides and analyzing them via liquid chromatography-tandem mass spectrometry (LC-MS/MS), we map the precise glycosylation sites and characterize the glycan microheterogeneity with unparalleled accuracy and sensitivity.
Glycoform Quantification
Quantitative analysis of glycoforms is essential for assessing the relative abundance of specific glycan structures and monitoring glycosylation changes under different manufacturing conditions or biological contexts. Creative Proteomics offers robust glycoform quantification services utilizing label-free or stable isotope labeling strategies coupled with mass spectrometry-based detection, enabling precise quantitation of glycan variants in glycosylated antibodies.
Glycosylation Site Occupancy
Determining the occupancy of glycosylation sites within the antibody molecule is crucial for understanding glycan attachment dynamics and optimizing glycoengineering strategies. Through innovative analytical workflows combining enzymatic digestion, glycan release, and site-specific quantification methods, Creative Proteomics accurately assesses glycosylation site occupancy in glycosylated antibodies, providing valuable insights into their structural heterogeneity and functional implications.
Technical Platforms for Glycosylated Antibody Studies Services
Below are some of the key instrumental platforms employed in our analytical workflows:
Mass Spectrometry (MS)
a. High-Resolution Mass Spectrometry (HRMS): Creative Proteomics harnesses high-resolution mass spectrometers, such as Orbitrap and time-of-flight (TOF) instruments, to achieve accurate mass measurements of glycan species and glycopeptides. These instruments enable precise determination of glycan compositions, structures, and relative abundances, facilitating in-depth glycosylation analysis.
b. Liquid Chromatography-Mass Spectrometry (LC-MS): Coupling liquid chromatography with mass spectrometry allows for the separation and identification of glycan and glycopeptide species based on their retention times and mass spectra. Creative Proteomics utilizes state-of-the-art LC-MS systems, including nanoLC and ultra-high-performance liquid chromatography (UHPLC) platforms, to achieve high sensitivity and resolution in glycosylated antibody analysis.
Chromatography Systems
a. HPLC Systems: High-performance liquid chromatography (HPLC) systems equipped with various chromatographic columns, such as reverse-phase (RP), hydrophilic interaction chromatography (HILIC), and porous graphitic carbon (PGC) columns, are employed for the separation and purification of glycopeptides and glycans prior to mass spectrometric analysis. Creative Proteomics utilizes Agilent, Waters, and Thermo Fisher HPLC systems to achieve robust and reproducible chromatographic separations.
b. Capillary Electrophoresis (CE): Capillary electrophoresis systems, including capillary zone electrophoresis (CZE) and capillary electrophoresis-mass spectrometry (CE-MS) platforms, are utilized for the separation and quantification of charged glycan species based on their electrophoretic mobilities. Creative Proteomics leverages Agilent and SCIEX capillary electrophoresis systems to complement mass spectrometry-based glycosylation analysis.
Glycan Analysis Software
a. Data Analysis Software: Creative Proteomics employs specialized software packages for the processing, interpretation, and visualization of mass spectrometry data generated from glycosylated antibody analysis. These software tools, including Thermo Xcalibur, Proteome Discoverer, and GlycoWorkbench, facilitate glycan identification, quantification, and structural elucidation, enhancing the efficiency and accuracy of glycosylation analysis workflows.
Sample Requirements for Glycosylated Antibody Studies Services
| Sample Type | Sample Volume | Recommended Amount |
|---|---|---|
| Monoclonal Antibodies | 100-500 µL | 5-10 mg |
| Polyclonal Antibodies | 100-500 µL | 10-20 mg |
| Recombinant Antibodies | 100-500 µL | 5-10 mg |
| Cell Culture Supernatant | 1-5 mL | 100-500 µg/mL |
| Serum or Plasma | 100-500 µL | 5-20 µg/mL |
| Tissue Homogenate | Variable | 10-50 mg |
| Biopharmaceutical Product | Variable | As per manufacturing specifications |
Applications of Glycosylated Antibody Studies Services
Bioprocessing and Quality Control: Glycosylation patterns on antibodies can affect their stability, efficacy, and immunogenicity. Analyzing glycosylation profiles helps in optimizing bioprocessing conditions to ensure consistent antibody quality and production.
Biopharmaceutical Development: Understanding the glycosylation profile of antibodies is crucial during the development of biopharmaceuticals. It enables us to engineer antibodies with desired glycosylation patterns to enhance therapeutic efficacy, pharmacokinetics, and reduce immunogenicity.
Vaccine Development: Glycosylated antibodies play a significant role in immune responses and are involved in the development of vaccines. Analyzing the glycosylation patterns of antibodies can provide insights into vaccine efficacy, immunogenicity, and potential adjuvant effects.
Biomarker Discovery: Glycosylation patterns on antibodies have been linked to various diseases and physiological states. Analyzing these patterns can help identify novel biomarkers for disease diagnosis, prognosis, and monitoring treatment response.
Immunotherapy Optimization: Glycosylation profiles influence the effector functions of therapeutic antibodies, such as antibody-dependent cellular cytotoxicity (ADCC) and complement-dependent cytotoxicity (CDC). By analyzing glycosylation, researchers can optimize immunotherapies for enhanced efficacy in cancer treatment and autoimmune diseases.
Comparative Studies: Comparing glycosylation patterns between different batches of antibodies, biosimilars, or between species can provide valuable information about structural differences and potential functional implications, aiding in regulatory approval processes and ensuring product consistency.
Drug Safety Assessment: Changes in glycosylation patterns of antibodies can sometimes lead to adverse effects, such as increased immunogenicity or altered pharmacokinetics. Glycosylated antibody analysis helps in assessing the safety profile of therapeutic antibodies during preclinical and clinical studies.
Research Tools Development: Glycosylated antibodies are widely used as research tools in various fields, including immunology, cell biology, and biochemistry. Analyzing their glycosylation patterns can improve the design and development of research reagents for better specificity and functionality.
Title: Middle-Up MS-Based Strategy for Subclass and Allotype-Specific Analysis of Polyclonal IgG Glycosylation
Background
Analyzing the glycosylation of polyclonal IgGs poses challenges due to their heterogeneity in subclasses, allotypes, and post-translational modifications (PTMs). Traditional methods, such as glycopeptide analysis, lack the resolution to differentiate between subclasses and allotypes, hindering comprehensive characterization.
Sample
Plasma samples from five independent donors were analyzed to investigate the glycosylation patterns of polyclonal IgGs. The donors exhibited heterozygosity in at least one subclass, resulting in a total of 12 identified allotypes. These samples provided a diverse representation of IgG glycosylation profiles within a population.
Technical Methods
Isolation of Fc Subunits: Fc subunits were isolated from plasma using selective IgG trapping and on-bead digestion with IdeS, allowing for the enrichment of Fc/2 subunits.
Orthogonal Separation Techniques: Two orthogonal separation techniques, hydrophilic interaction liquid chromatography (HILIC) and capillary electrophoresis (CE), were employed prior to mass spectrometry (MS) detection. HILIC provided resolution of allotypes and strong glycosylation profiles, while CE allowed separation of subclasses based on differences in charge or size.
High-Resolution MS Analysis: High-resolution MS enabled the identification of specific allotypes with low ppm error. Spectral deconvolution revealed oxidation of IgG1 and IgG2, with levels increasing over sample storage time.
Glycosylation Analysis: The middle-up approach permitted subclass and allotype-specific glycosylation analysis, overcoming limitations of standard glycopeptide methods. The relative abundances of different glycoforms were determined for each specific subclass and allotype, highlighting variability among donors and subclasses.
Results
High Variability in Glycosylation Profiles: Analysis of the five donors revealed high variability in IgG glycosylation profiles within the same population, with differences potentially associated with donor and subclass.
Clear Distinctions Between Subclasses: Differences in glycosylation patterns were observed between subclasses, with IgG1 showing higher galactosylation and sialylation compared to other subclasses.
Limited Differences Between Allotypes: While no obvious differences in glycosylation were observed between allotypes within donors, some variation was noted between donors, emphasizing the importance of studying glycosylation in a subclass and allotype-specific manner.
Future Directions: Further research will focus on analyzing a larger number of samples from different populations to detect all possible allotypes and explore potential differences in glycosylation patterns.
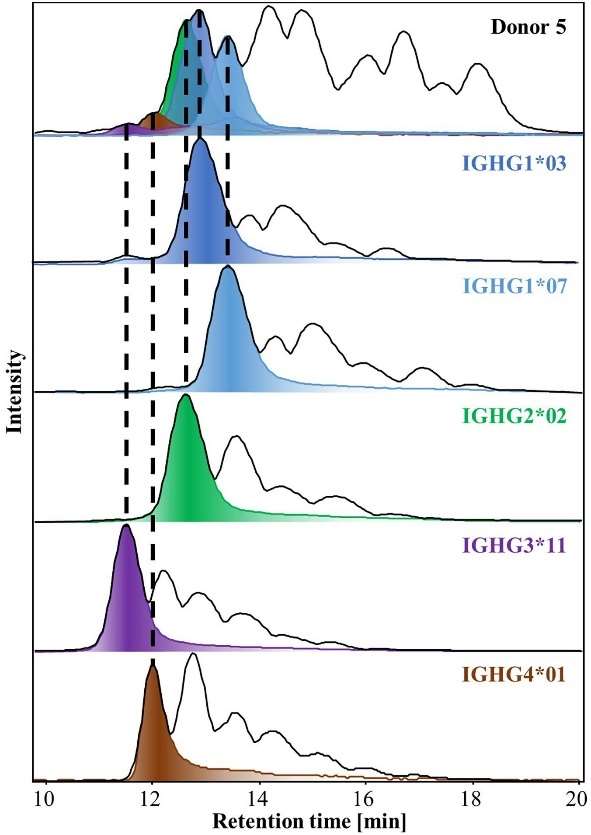 BPCs and EICs of G0F obtained by HILIC-MS of IgG-Fcs isolated from Donor 5 (top trace) and from recombinant IgGs of the relevant allotypes (bottom traces).
BPCs and EICs of G0F obtained by HILIC-MS of IgG-Fcs isolated from Donor 5 (top trace) and from recombinant IgGs of the relevant allotypes (bottom traces).
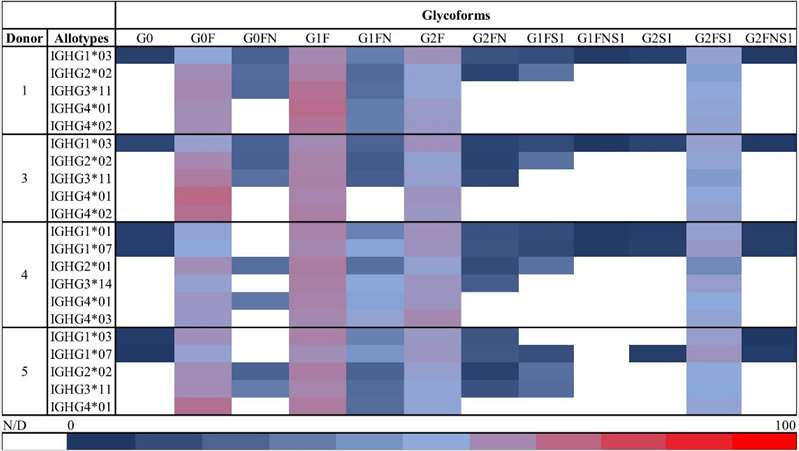 Overview of the relative abundances of the glycoforms detected by HILIC-MS for all the allotypes in each of the five assessed donors.
Overview of the relative abundances of the glycoforms detected by HILIC-MS for all the allotypes in each of the five assessed donors.
Reference
- Sénard, Thomas, et al. "MS-based allotype-specific analysis of polyclonal IgG-Fc N-glycosylation." Frontiers in immunology 11 (2020): 565585.




