- Service Details
- Case Study
At Creative Proteomics, we understand the potential of FTIR spectroscopy and leverage this robust technique for comprehensive glycoprotein analysis. Our team of skilled biochemical and analytical scientists combines their expertise with cutting-edge technology, to extract the most reliable and detailed spectral data. From protein identification and characterization to elucidating post-translational modifications, our FTIR analysis service provides a complete package to cater to all your glycoprotein analysis needs.
Principles of FTIR Spectroscopy
Fourier Transform Infrared (FTIR) spectroscopy is a non-destructive analytical technique used to identify functional groups in molecules based on the absorption of infrared radiation. When infrared light interacts with a molecule, it causes molecular vibrations, leading to characteristic absorption bands in the infrared spectrum. By analyzing these absorption bands, researchers can deduce information about the molecular structure of the sample.
Glycoproteins contain carbohydrate moieties that exhibit characteristic infrared absorption bands due to the presence of functional groups such as hydroxyl (OH), carbonyl (C=O), and amide (C-N) groups. By subjecting glycoprotein samples to FTIR spectroscopy, researchers can obtain spectra that provide insights into the composition, conformation, and interactions of the carbohydrate and protein components within the molecule.
Why Choose FTIR Spectroscopy?
- High Sensitivity: Even minute quantities of a substance can be analyzed using FTIR, enabling detection of subtle changes in the glycoprotein structure and composition. This high level of sensitivity proves advantageous when limited samples are available for experimentation.
- Non-Destructive Technique: FTIR is a non-destructive technique, implying that the sample remains intact following analysis. This allows the potential for re-analysis or further testing if required without the need for additional samples.
- Speed and Simplicity: One of the most compelling reasons for choosing FTIR spectroscopy is its speed and simplicity. With FTIR, rapid data acquisition is possible, reducing the amount of time between initiation and culmination of the experiment. Additionally, the fact that sample preparation is largely straightforward and uncomplicated further boosts its efficiency.
- Structural Information: FTIR provides valuable information about the structural composition of a substance. In the case of glycoproteins, this includes information regarding the presence and orientation of various functional groups, which can be of immense value in understanding their biological function.
Technical Platform for Glycoprotein FTIR Spectroscopy Analysis
FTIR Spectrometer: Equipped with a high-performance Fourier-transform infrared spectrometer with a spectral resolution of up to 0.1 cm^-1, allowing for precise characterization of glycoproteins.
Sample Preparation Equipment: Utilizing advanced sample preparation techniques including purification, concentration, and desalting to ensure high-quality spectra.
ATR Accessory: Leveraging state-of-the-art Attenuated Total Reflection (ATR) accessories for direct analysis of liquid and solid samples, minimizing sample handling and preparation time.
Software for Data Analysis: Employing industry-leading software packages such as OMNIC and Origin for data acquisition, processing, and spectral analysis, enabling accurate interpretation of glycoprotein spectra.
Reference Materials: Calibration and validation are performed using certified reference materials with known spectral signatures, ensuring the accuracy and reliability of results.
Quality Control Standards: Strict adherence to quality control standards, including regular calibration and performance verification of instruments, to guarantee the consistency and reproducibility of analysis.
Specific Analytical Capabilities Offered by Creative Proteomics
Glycoprotein Composition Analysis
We offer detailed analysis of glycoprotein composition, including identification of carbohydrate and protein constituents, quantification of glycan moieties, and determination of glycosylation patterns.
Conformational Analysis
Our platform enables conformational analysis of glycoproteins, allowing for the characterization of secondary and tertiary structures. By monitoring changes in infrared spectra under various conditions, we assess the stability, folding, and unfolding of glycoprotein molecules.
Interaction Studies
We conduct interaction studies to investigate the binding affinity and specificity of glycoproteins with ligands, receptors, or other biomolecules. By monitoring spectral changes upon complex formation, we elucidate the molecular mechanisms underlying glycoprotein interactions.
Sample Requirements for Glycoprotein Fourier Transform Infrared Analysis
| Sample Type | Sample Volume | Sample Concentration | Sample Preparation |
|---|---|---|---|
| Purified Protein | 10-50 µL | 1-10 mg/mL | Dialysis or buffer exchange to remove interfering agents |
| Cell Culture Supernatant | 100-500 µL | Concentrated as needed | Protein precipitation or ultrafiltration |
| Tissue Homogenate | 10-50 mg | N/A | Homogenization in appropriate buffer |
| Serum/Plasma | 50-100 µL | Diluted as needed | Protein precipitation or ultrafiltration |
| Biopsy Specimen | Variable | N/A | Homogenization or extraction in appropriate solvent |
Case. Effect of Pre-Soaking Peanut Seeds in Bulk and Nano-Fe2O3 Suspension on Leaf Protein Structure
Background:
Micronutrients, particularly iron, play a crucial role in plant growth and development. Iron deficiency can lead to various symptoms such as leaf yellowing, impacting crop productivity. Nanotechnology offers potential solutions to enhance nutrient uptake in plants. In this study, the effects of pre-soaking peanut seeds in bulk and nano-Fe2O3 suspensions on leaf protein structure were investigated.
Samples:
Peanut seeds obtained from the Regional Agricultural Research Institute, Virudhachalam, India, were used in the study. Leaf samples collected after 30 days of growth were analyzed.
Technical Method:
Synthesis of Fe2O3 Nanoparticles: Chemical precipitation method was employed using ferric nitrate precursor material and ammonium hydroxide solution. Fe2O3 nanoparticles were annealed at 400 °C for 4 hours.
Characterization: Fe2O3 nanoparticles were characterized using X-ray diffraction (XRD), scanning electron microscopy (SEM), and atomic force microscopy (AFM) to confirm phase purity, particle size, and uniformity.
Seed Preparation and Pre-soaking: Peanut seeds were sterilized and treated with bulk and nano-Fe2O3 suspensions at different concentrations (500 and 4000 ppm) for 10 hours via ultrasonification. Seeds were then sowed in separate pots, and leaf samples were collected after 30 days.
FT-IR Spectral Analysis: Leaf samples were oven-dried, ground to fine powder, and analyzed using Fourier-transform infrared (FT-IR) spectroscopy in the 4000–400 cm−1 region. Spectra were recorded using the KBr pellet technique, and spectral analysis was performed to assess changes in protein structure.
Results:
Fe2O3 Nanoparticle Characterization: XRD confirmed phase purity, with an average particle size of 21 nm. SEM and AFM images demonstrated uniform nanophase distribution.
FT-IR Analysis: Increased carbohydrate and nucleic acid content of amide A and B proteins was observed in all samples, while amide I and II proteins showed slight decreases. Variation in secondary protein structure, particularly an increase in β-sheet and β-turn structures, was noted.
PCA Analysis: Principal component analysis revealed that control, L2, and L3 samples explained the majority of the variance in leaf protein structure, indicating the influence of Fe2O3 treatment.
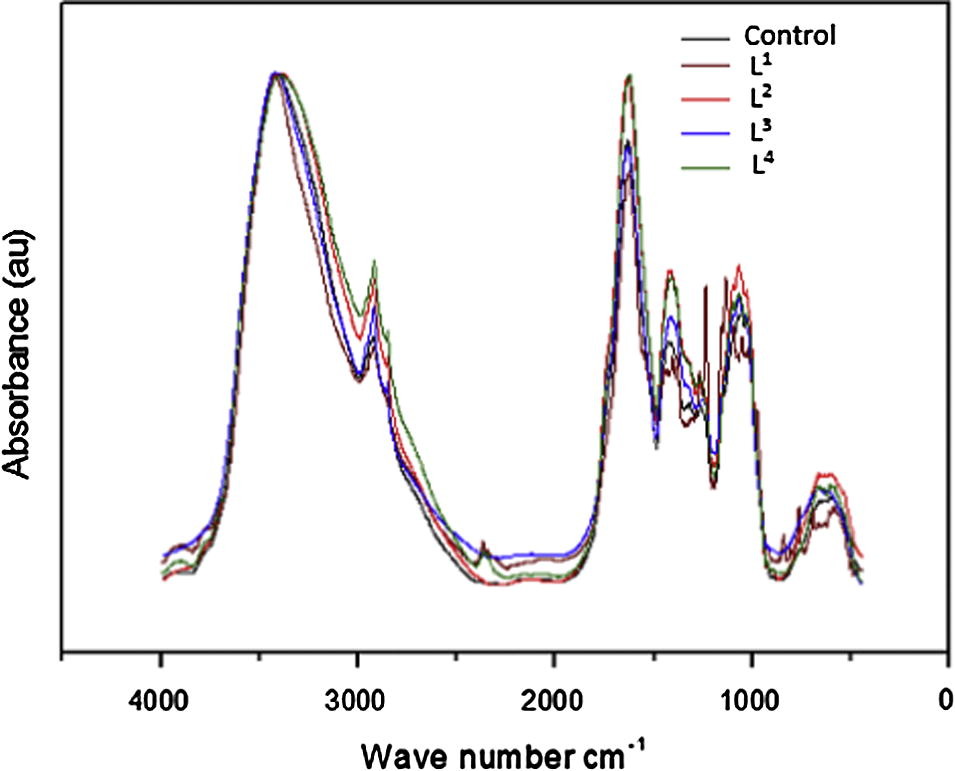 FT-IR spectra for the peanut leaf samples collected after 30 days.
FT-IR spectra for the peanut leaf samples collected after 30 days.
Reference
- Suresh, S., S. Karthikeyan, and K. Jayamoorthy. "Effect of bulk and nano-Fe2O3 particles on peanut plant leaves studied by Fourier transform infrared spectral studies." Journal of Advanced Research 7.5 (2016): 739-747.




