- Service Details
- Case Study
What is Glycan Sequencing?
Glycan Sequencing plays a pivotal role in the scientific investigation of glycans, intricate carbohydrate structures deeply integrated into biological processes. These structures, inherent within glycoproteins, assume critical functions in cellular processes, such as cell signaling and immune responses. The imperative for glycan sequencing arises from the realization that even subtle variations in carbohydrate structures can exert significant impacts on the bioactivity of glycoproteins.
Understanding the biological relevance of a specific glycan within a glycoconjugate necessitates the decoding of its primary sequence and subsequent structure. This process gains significance due to the intricate nature of glycosylation, where minor changes manifest notable effects. At Creative Proteomics, we acknowledge the intricacies of this task and provide specialized glycan sequencing services, utilizing advanced technologies, including mass spectrometry techniques such as capillary electrophoresis (CE), liquid chromatography-mass spectrometry (LC-MS), and high-performance liquid chromatography (HPLC). These methodologies, coupled with exoglycosidase digestion techniques, ensure a thorough analysis of glycan structures.
Technology Platform for Glycan Sequencing
Enzymatic Digestion and Glycosidase Treatment: Enzymatic methods involve breaking down complex glycans into smaller fragments using enzymes. This process aids in the structural elucidation of glycans.
MALDI-TOF (Matrix-Assisted Laser Desorption/Ionization Time-of-Flight): Ideal for glycan profiling, providing information about the mass and composition of intact glycans.
ESI-MS (Electrospray Ionization Mass Spectrometry): Useful for identifying glycan structures and determining branching patterns.
HPLC (High-Performance Liquid Chromatography): Coupled with MS, HPLC separates and analyzes glycans, improving sensitivity and resolving glycan isomers.
CE-LIF (Capillary Electrophoresis with Laser-Induced Fluorescence): Separates and analyzes glycans based on charge and size, known for high resolution.
Nuclear Magnetic Resonance (NMR) Spectroscopy: Provides valuable structural information about glycans, particularly useful for elucidating linkage positions between sugar residues.
Ion Mobility Spectrometry (IMS): Analyzes glycan isomers based on size, shape, and charge, contributing to structural characterization.
High-Throughput Glycan Microarrays: Facilitates high-throughput screening of glycan-protein interactions, valuable for studying glycan-binding proteins.
Applications of Glycan Sequencing
Biopharmaceutical Development: Glycan sequencing is essential for ensuring the quality and safety of therapeutic glycoproteins. By characterizing glycosylation patterns, researchers can monitor and control the bioactivity and immunogenicity of these proteins, impacting biopharmaceutical efficacy.
Disease Biomarker Identification: In the realm of disease research, glycan sequencing is instrumental in identifying disease-specific glycosylation changes. Aberrant glycan structures are associated with various diseases, and glycan sequencing aids in discovering potential biomarkers for early disease detection and prognosis.
Immunology Research: The study of glycan structures on immune system molecules is enhanced through glycan sequencing. This detailed analysis contributes to a better understanding of immune function, facilitating the development of novel immunotherapies and advancements in immunology research.
Microbial Glycobiology: Glycan sequencing is employed to study the surface glycomes of microorganisms. This is particularly relevant for vaccine development and understanding host-pathogen interactions, contributing to the design of effective vaccines against infectious diseases.
Glycoengineering: In the field of glycoengineering, glycan sequencing plays a crucial role in modifying glycan structures for various applications. This includes engineering glycosylation patterns in cell lines to produce recombinant proteins with specific glycan profiles, impacting the therapeutic properties of biologics.
Sample Requirements for Glycan Sequencing
| Sample Type | Recommended Sample Size | Additional Considerations |
|---|---|---|
| Glycoproteins | 50-100 micrograms | Ensure high purity; consider the glycoprotein source (e.g., antibodies, enzymes). |
| Tissue Samples | Depends on tissue type; ensure sufficient material for glycoprotein extraction | Different tissues may require varying amounts; maintain sample integrity during processing. |
| Cell Culture Supernatants | Milliliters of culture supernatant | Volume depends on the glycoprotein concentration; consider the specificity of the glycan analysis method. |
| Serum/Plasma Samples | Microliters to Milliliters | Study glycosylation patterns in circulating proteins; collect and handle samples with anti-coagulants. |
| Urine Samples | Milliliters to Liters | Investigate urinary glycoproteins; consider variations related to kidney function or diseases. |
| Saliva Samples | Milliliters | Non-invasive; suitable for studying glycan structures related to oral health and diseases. |
| CSF (Cerebrospinal Fluid) | Microliters to Milliliters | Important for neurological research; consider low abundance of glycoproteins in CSF. |
| Synovial Fluid | Microliters to Milliliters | Relevant for studying glycan changes in joint-related diseases; consider potential variations in glycosylation. |
| Mucin Samples | Micrograms to Milligrams | Heavily glycosylated; provides insights into mucin-type O-glycans. |
| Cell Lysates | Micrograms to Milligrams | Analyze intracellular glycoproteins; lysing cells and extracting glycoproteins for sequencing. |
Case. Comprehensive Characterization of Glycan Isomers in Monoclonal Antibodies Produced in Murine Myeloma Cells
Background:
The study aims to provide a thorough understanding of the structural diversity and distribution of glycan isomers in monoclonal antibodies (mAbs) produced in murine myeloma cells. Glycan structures play a crucial role in the quality and functionality of therapeutic mAbs, and non-human glycans can pose challenges in biotherapeutics production.
Samples:
Monoclonal antibodies derived from murine myeloma cells were used as the primary samples for analysis. The study focused on glycan isomers containing α-Gal residues and explored their structural variations, including Neu5Gc-sialylated glycans.
Technical Method:
1. Glycoproteomic Analyses of mAb Glycopeptides using RP C18 Column:
- Reduction and alkylation of mAb samples.
- Dialysis and trypsin digestion of proteins.
- Analysis of resulting peptides using RP LC MS/MS with an Orbitrap Fusion Lumos system.
2. Glycomic Analyses of Released mAb Glycans using PGC LC MS/MS:
- In-gel digestion of alkylated proteins with PNGase F.
- Extraction and purification of released glycans.
- Additional cleavage with glycosidases for specific glycan modifications.
- Analysis of glycans using nanospray PGC LC MS/MS on an Orbitrap Fusion mass spectrometer.
3. Construction of a mAb N-glycan Library for Glycopeptide Identification:
- Construction of an in-house N-glycan library based on common glycan structures from literature and LC MS/MS analyses.
- Identification of glycopeptides using Byonic software and manual inspection.
- MS/MS sequencing for unknown glycopeptides.
4. Elucidation of Structural Glycan Isomers:
- Assignment of isomeric glycans based on glycan biosynthetic pathway knowledge, diagnostic ions, accurate masses, and MS/MS fragment patterns.
- Validation of glycan structures through LC elution order and exoglycosidase sequencing.
5. Synthesis of G1 Standard Glycans:
- Isolation of sialoglycoprotein (SGP) from egg yolk.
- Release and purification of G1 glycans.
- Degalactosylation and purification of G1(6) glycans.
Results:
The study revealed a comprehensive characterization of glycan isomers in murine myeloma cell-derived mAbs. It identified α-Gal-containing glycans, confirmed structural consistency between α-galactosylated and Neu5Gc-sialylated glycans, and explored the distribution of non-human epitopes. The methods employed demonstrated high sensitivity and accuracy in glycan sequencing, providing valuable insights for quality control in future biotherapeutics production. The findings contribute to the understanding of structural diversity and cellular distributions of non-human glycans in recombinant mAbs.
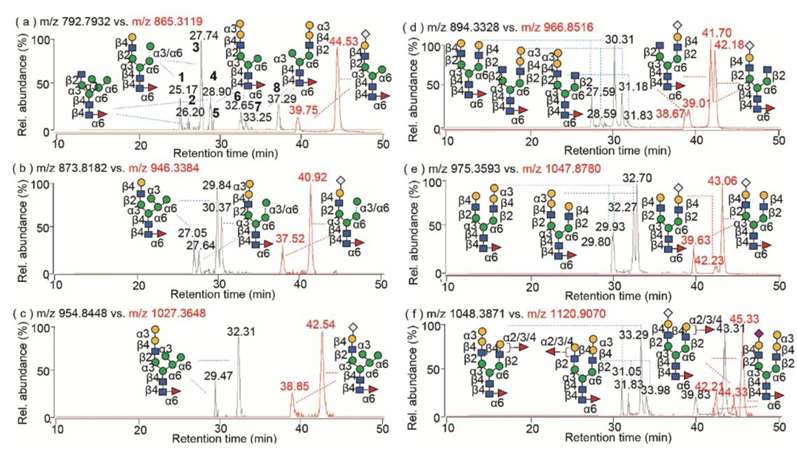 Structural comparison of the non-human glycan isomers between terminal residues of α-Gal and Neu5Gc
Structural comparison of the non-human glycan isomers between terminal residues of α-Gal and Neu5Gc
Reference
- She, Yi-Min, Shaojun Dai, and Roger Y. Tam. "Highly sensitive characterization of non-human glycan structures of monoclonal antibody drugs utilizing tandem mass spectrometry." Scientific Reports 12.1 (2022): 15109.




