- Service Details
- Case Study
What is Glycan Degradation?
Glycan degradation analysis is a sophisticated analytical technique utilized in the field of glycobiology to unravel the intricate structures and modifications of complex carbohydrates, known as glycans. Glycans play pivotal roles in numerous biological processes, including cell signaling, immunity, and pathogen recognition. They are composed of various sugar molecules, often arranged in elaborate branching patterns, making their structural elucidation a challenging task.
Glycan degradation analysis involves the systematic breakdown of these complex carbohydrate molecules into simpler units through enzymatic or chemical methods. This process facilitates the characterization and identification of individual sugar residues, glycosidic linkages, and branching patterns within the glycan structure.
The primary objective of glycan degradation analysis is to decode the glycan structures at a molecular level, unveiling their composition, sequence, and spatial arrangement. By deciphering the intricate glycan structures, researchers can gain valuable insights into their biological functions, interactions with other molecules, and roles in health and disease.
What We Can Provide?
At Creative Proteomics, we offer detailed analysis of glycan degradation modifications. We can customize our analyses to meet the specific analytical needs of our clients in therapeutic development and basic research.
Identification of Glycan Degradation Products: We employ advanced analytical techniques to identify various glycan degradation products, allowing for a detailed characterization of the glycan modification process.
Quantification of Glycan Degradation: Our methods enable accurate quantification of glycan degradation products, providing valuable insights into the extent of glycan modification under different conditions or treatments.
Glycan Degradation Profiling: We perform comprehensive profiling of glycan degradation products across different biological samples or experimental conditions, facilitating comparative studies and biomarker discovery.
Structural Analysis of Degraded Glycans: We utilize cutting-edge technologies to elucidate the structural changes occurring in degraded glycans, facilitating a deeper understanding of the underlying mechanisms of glycan degradation.
Glycan Degradation Pathway Analysis: We offer comprehensive pathway analysis services to unravel the intricate pathways involved in glycan degradation, aiding in the identification of key enzymes and regulatory factors.
Characterization of Degradation Enzymes: Our expertise allows for the characterization of enzymes involved in glycan degradation, including their substrate specificity, kinetics, and regulatory mechanisms.
Customized Analysis Services: We provide tailored analysis solutions to meet specific research needs or project requirements, ensuring high-quality and reliable results.
Technical Platform for Glycan Degradation Analysis
Glycan degradation analysis relies on advanced analytical techniques, with mass spectrometry (MS) serving as the cornerstone for elucidating glycan structures and modifications. Creative Proteomics employs state-of-the-art MS instruments, including:
Liquid Chromatography-Mass Spectrometry (LC-MS):
We utilize high-resolution LC-MS systems such as the Thermo Scientific™ Orbitrap series (e.g., Orbitrap Fusion, Orbitrap Exploris) or the Waters Xevo™ series (e.g., Xevo G2-XS QTOF) for comprehensive analysis of glycan degradation products. LC separation coupled with MS detection enables accurate identification and quantification of degraded glycans.
Matrix-Assisted Laser Desorption/Ionization Mass Spectrometry (MALDI-MS):
Our MALDI-MS platforms, such as the Bruker Daltonics ultrafleXtreme or the SCIEX™ TOF/TOF™ series, offer high-throughput analysis of glycan degradation products. MALDI facilitates direct analysis of glycan samples with minimal sample preparation, making it ideal for rapid screening and profiling studies.
Tandem Mass Spectrometry (MS/MS):
We utilize tandem mass spectrometers integrated into our LC-MS or MALDI-MS systems, including collision-induced dissociation (CID) and higher-energy collisional dissociation (HCD) capabilities. MS/MS enables structural elucidation of degraded glycans by fragmenting precursor ions and analyzing the resulting fragment ions.
Sample Requirements for Glycan Degradation Analysis
| Sample Type | Detailed Description | Recommended Quantity |
|---|---|---|
| Glycoproteins/Glycopeptides | Proteins containing covalently attached glycans | 10-100 micrograms |
| Glycans (purified) | Isolated and purified carbohydrate molecules | 1-10 micrograms |
| Serum/Plasma | Blood components after clotting (Serum) or without (Plasma) | 100-500 microliters |
| Tissue Extracts | Extracts from various tissues or organs | 5-20 milligrams |
| Cell Culture Supernatant | Liquid surrounding cultured cells | 100-500 microliters |
| Urine | Liquid waste excreted by the kidneys | 1-5 milliliters |
| Cerebrospinal Fluid (CSF) | Fluid surrounding the brain and spinal cord | 500 microliters - 1 milliliter |
| Saliva | Fluid produced by the salivary glands | 100-500 microliters |
| Synovial Fluid | Fluid found in joints | 100-500 microliters |
| Amniotic Fluid | Fluid surrounding the fetus in the womb | 500 microliters - 1 milliliter |
| Breast Milk | Milk produced by mammary glands | 1-5 milliliters |
| Tears | Fluid secreted by the lacrimal glands | 100-500 microliters |
Sample Quantity: Adequate sample quantity is necessary for robust analysis, typically ranging from micrograms to milligrams of glycoproteins or glycan samples.
Sample Integrity: Preserve the integrity of glycan structures during preparation by avoiding conditions that may lead to degradation or modification, such as extremes of pH or temperature.
Buffer Composition: Use buffer solutions that maintain physiological pH and ionic strength to preserve glycan integrity. Common buffers include phosphate-buffered saline (PBS) and Tris-HCl.
Deglycosylation: Enzymatic or chemical deglycosylation may be required for glycoproteins or glycopeptides to isolate glycans. Choose appropriate deglycosylation agents based on the glycan composition and linkage types present.
Sample Storage: Store samples at recommended temperatures (-80°C for long-term storage or -20°C for short-term storage) to prevent degradation prior to analysis. Avoid repeated freeze-thaw cycles.
Case. Structural Glycoproteomics Reveals ER Stress Response Mechanisms in Plants under Chilling Stress
Background:
Understanding how plants respond to chilling stress is crucial for agricultural productivity. Endoplasmic reticulum (ER) stress is a significant component of this response, but its molecular mechanisms remain unclear. Glycoproteins play essential roles in ER function and stress responses, making them valuable targets for studying plant stress adaptation.
Samples:
The study utilized Arabidopsis thaliana seedlings grown under normal (22°C) and cold stress (4°C) conditions for varying periods to analyze the glycoproteome and ER stress response.
Technical Method:
Glycoproteome Analysis: Employed LC-MS/MS to analyze glycoproteins involved in plant defense responses to abiotic stresses. Identified proteins encoded by stress-related genes, including those involved in temperature and salt responses.
N-Glycosylation Analysis: Determined N-glycosylation patterns of glycoproteins, with ER-resident proteins primarily showing oligomannosidic glycoforms, while receptor-like protein kinases exhibited complex N-glycans.
GPI-Anchored Membrane Protein Identification: Utilized LC-MS/MS to identify glycopeptides of GPI-anchored membrane proteins, indicating possible subcellular location-specific proteins.
Quantitative Proteomics: Employed 15N-isotope metabolic labeling and LC-MS/MS to quantify ER-resident glycoproteins under normal and cold stress conditions. Revealed up-regulation of ER stress-responsive glycoproteins in response to chilling stress.
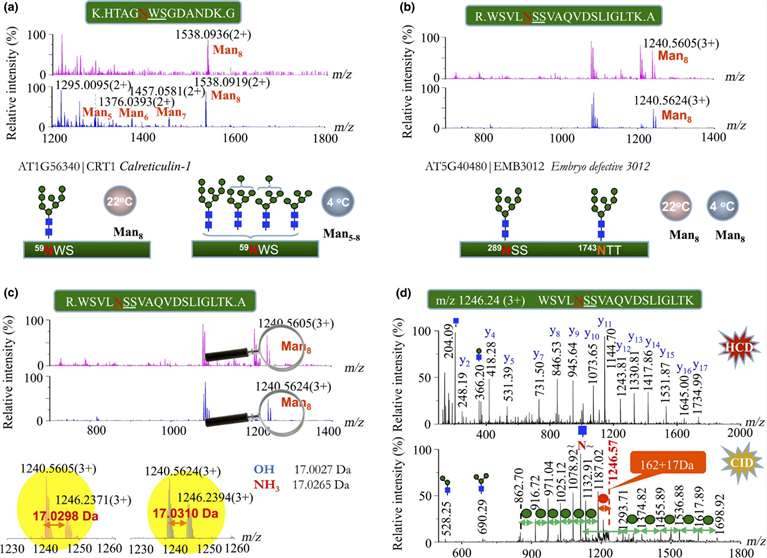 Discriminating the N-glycan profiles of endoplasmic reticulum (ER)-resident glycoproteins in plants under normal and cold-treated conditions for 18 d.
Discriminating the N-glycan profiles of endoplasmic reticulum (ER)-resident glycoproteins in plants under normal and cold-treated conditions for 18 d.
Results:
N-Glycan Degradation: Cold stress induced N-glycan degradation in ER-resident glycoproteins, observed through trimming of high-mannose N-glycans.
ER Stress Response: Up-regulation of ER-resident glycoproteins, particularly chaperones like protein disulfide isomerases (PDIs) and calreticulins (CRTs), in response to chilling stress.
Enhancement of Plant Tolerance: Findings suggest that ER-associated N-glycan degradation and up-regulation of ER-resident glycoproteins contribute to plant adaptation to chilling stress.
Quantitative Proteomics Validation: Quantitative analyses confirmed the correlation between cold-induced ER stress and up-regulation of ER-resident glycoproteins, providing molecular insights into plant stress responses.
Implications for Plant Breeding: Understanding ER stress response mechanisms could inform strategies to enhance plant tolerance to chilling stress and improve agricultural productivity.
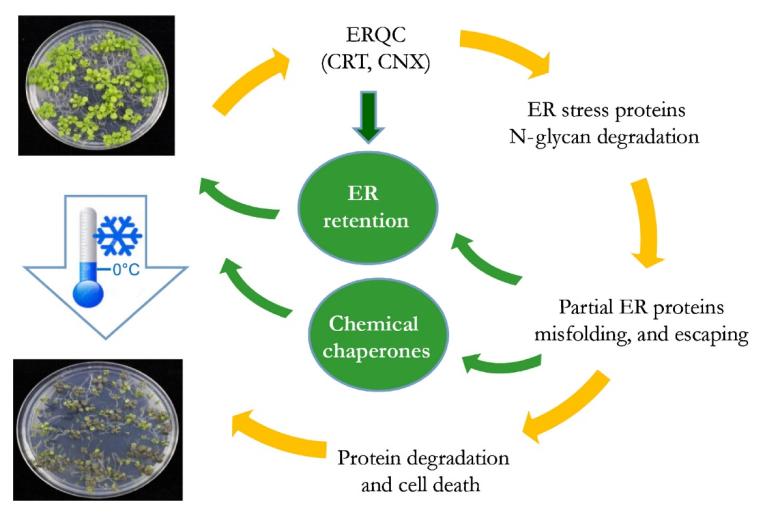 Diagram of the stress-related glycoproteins in the endoplasmic reticulum (ER) and the effects of chilling stress in Arabidopsis.
Diagram of the stress-related glycoproteins in the endoplasmic reticulum (ER) and the effects of chilling stress in Arabidopsis.
Reference
- Ma, Jun, et al. "Endoplasmic reticulum‐associated N‐glycan degradation of cold‐upregulated glycoproteins in response to chilling stress in Arabidopsis." New Phytologist 212.1 (2016): 282-296.




