- Service Details
- Case Study
Glycans, or carbohydrates, are essential biomolecules involved in a myriad of biological functions. They play pivotal roles in cell communication, immune response, and various physiological processes. Glycan profiling aims to systematically analyze and characterize the diverse structures and compositions of glycans present in biological samples.
Glycoproteins, essential players in various physiological processes, pose a formidable challenge due to their low abundance and structural diversity. Our glycan profiling services are meticulously crafted to address this complexity, providing unparalleled insights into the glycosylation patterns of proteins.
Techniques Employed in Glycan Profiling
N-Glycan Profiling
N-Glycans are predominantly linked to asparagine residues in proteins, influencing protein folding, stability, and function. N-Glycan profiling involves the characterization of these glycans, allowing researchers to understand the glycosylation patterns associated with specific proteins. Techniques such as mass spectrometry and liquid chromatography are commonly employed for N-Glycan profiling, providing high-resolution data on glycan structures.
- MALDI-TOF MS: Matrix-assisted laser desorption/ionization time-of-flight mass spectrometry provides high-throughput structural characterization with sensitivity and robustness. This method is particularly advantageous for profiling mixtures of N-glycans.
- UPLC-FLD/MS: Ultra-high performance liquid chromatography coupled with fluorescence detection/mass spectrometry offers a powerful tool for N-glycan profiling. This fast and robust technique is ideal for analyzing larger sample numbers with known N-glycan profiles.
- HPAEC-PAD: High-performance anion-exchange chromatography with pulsed amperometric detection allows for monosaccharide composition analysis, providing insights into both total glycosylation and specific monosaccharide amounts.
O-Glycan Profiling
O-Glycans, attached to serine or threonine residues, are essential for modulating protein function and stability. O-Glycan profiling focuses on the analysis of these glycans, unveiling the diversity and complexity of O-glycosylation. Advanced methods, including glycan microarrays and lectin-based approaches, facilitate the comprehensive profiling of O-glycans.
- MALDI-TOF MS: The most common method for O-glycan profiling, it involves permethylation of O-glycans to enhance analysis in positive mode.
- HPAEC-PAD: Providing details about fucosylation, HPAEC-PAD complements MALDI-TOF MS, offering insights into O-linked glycans.
Glycan Profiling Solutions at Creative Proteomics
Glycoprotein Profiling of Intact Proteins
Our rapid workflow is tailored to unravel the intricate glycoform profiles of intact proteins, providing researchers with a comprehensive understanding of glycoprotein structures. This holistic approach contributes to a more profound exploration of glycoprotein functionalities.
Middle-up/Middle-down Protein Subunit Analysis
Designed for efficiency, this fast workflow adeptly separates heavy and light protein chains. Alternatively, the utilization of IdeS digestion sheds light on post-translational modifications, offering valuable insights into the dynamic world of glycoprotein subunits.
Glycopeptide Mapping
Utilizing UPLC HILIC/TUV/MS, our glycopeptide mapping workflow preserves glycan heterogeneity and site occupancy. This detailed analysis at the peptide level facilitates a deeper comprehension of glycoprotein structures, enriching the overall glycoprotein analysis process.
Released Glycan Analysis
Our workflow-based solutions for released N-glycans streamline sample preparation, paving the way for information-rich analysis. This approach represents the future of glycan analysis, ensuring researchers have a robust toolkit for monitoring and characterizing released N-glycans efficiently.
Monosaccharide Composition Analysis
Serving as a vital orthogonal technique, monosaccharide profiling is employed to confirm glycan composition. This additional layer of analysis provides researchers with nuanced insights, further enhancing the precision and depth of glycoprotein characterization.
Advantages of Glycan Profiling
High Precision Glycan Analysis: Employing advanced mass spectrometry and liquid chromatography techniques with unparalleled precision, we can achieve a resolution capability of up to 0.1 Da. This ensures the differentiation of closely related glycan structures, providing a nuanced understanding of the glycosylation landscape.
Comprehensive Coverage: Glycan profiling offers extensive coverage of glycan structures, analyzing a broad spectrum from high-mannose to complex sialylated structures. This captures the full complexity of glycan profiles, enabling researchers to explore the diversity inherent in biological samples.
Cutting-Edge Instrumentation: At the core of glycan profiling is advanced instrumentation, including high-resolution mass spectrometers and liquid chromatography systems. The Q-Exactive HF-X mass spectrometer delivers exceptional sensitivity and accuracy for N-Glycan profiling, while the Acquity UPLC system ensures high-throughput and reproducibility in O-Glycan profiling.
Customization for Diverse Research Needs: We offer customization, allowing researchers to tailor analysis parameters, chromatographic conditions, and detection methods to align with specific research goals. This flexibility ensures that the glycan profiling approach extracts maximum relevant information from diverse biological samples.
Stringent Quality Control Measures: Prioritizing quality control at every stage, glycan profiling ensures reproducibility and reliability. Rigorous quality assurance protocols result in a low coefficient of variation (CV) of less than 5%, signifying consistency and accuracy across multiple analysis.
Applications of Glycan Profiling
Biomarker Discovery: Glycan profiling facilitates the identification of glycan-based biomarkers associated with diseases, paving the way for early detection and diagnostic advancements.
Biopharmaceuticals: In the biopharmaceutical industry, glycan profiling ensures the quality control of glycoprotein therapeutics, optimizing their efficacy and safety.
Cancer Research: Understanding aberrant glycosylation patterns in cancer cells through glycan profiling contributes to targeted therapeutic strategies and precision medicine.
Immunology: Glycan profiling enhances our understanding of immune responses, aiding vaccine development and immunotherapy research.
Sample Requirements for Glycan Profiling
| Sample Type | Recommended Quantity |
|---|---|
| Serum/Plasma | ≥ 50 μL |
| Tissues (Biopsies) | 10-20 mg |
| Cell Cultures | 1 million cells |
Case. Glycan Profiling of Mesenchymal Stem Cell-Derived Exosomes Reveals Sialic Acid-Mediated Interactions with Target Cells
Background:
Breast cancer is a significant global health concern, with millions of cases diagnosed annually. While mammography is commonly used for screening, its limitations in sensitivity and specificity have prompted the exploration of alternative diagnostic biomarkers. Protein glycosylation, particularly changes in N-glycan profiles, has gained attention as a potential avenue for improving breast cancer diagnostics.
Samples:
The study involved 159 female patients with breast cancer and 173 healthy female volunteers. Serum samples were collected according to a standardized protocol, processed, and stored for subsequent analysis. The patient cohort was age-matched with controls, and detailed patient characteristics were recorded, including cancer staging.
Technical Method:
Sample Collection and Processing: Blood specimens were collected in 8.5 cc vacutainer serum separator tubes. After centrifugation (10 min at 1000 g), serum was divided into 5 mL polystyrene tubes. Serum samples were stored at -80 °C within 4 hours of collection. Samples underwent one freeze-thaw cycle for aliquoting in eight 60-µl tubes.
Chemicals Used: Various chemicals were sourced, including Nonidet P-40 substitute, potassium dihydrogenphosphate, sodium chloride, sodium hydroxide, 1-hydroxybenzotriazole, super-DHB, etc.
Enzymatic N-Glycan Release: 6 µL serum sample was incubated with 12 µL 2% SDS at 60 °C for 10 min. Release mixture (4 µL NP40, 5× PBS, 0.6 µL PNGase F) was added, and samples were incubated overnight at 37 °C. Released glycans were stored at -20 °C before further preparation.
Ethyl Esterification and Glycan Purification: Ethyl esterification for sialic acid stabilization was performed. Purification utilized cotton HILIC SPE microtips with specific conditioning steps. Elution was carried out with MQ, and samples were spotted onto MALDI targets.
MALDI-FTICR-MS Measurement: MALDI-FTICR-MS experiments were performed using a Bruker 15T solariX XR FTICR MS. Spectra were recorded in the m/z-range from 1011.86 to 5000.00, with 1 M data points. Average spectra contained ten acquired scans, and the system was operated using ftmsControl software.
Data Preprocessing and Statistical Analysis: Serum N-glycosylation profiles were obtained for 159 breast cancer patient samples and 173 healthy volunteer samples. Batch correction was performed to account for MALDI-target batch effects. Logistic regression was applied for each glycan, and multivariate analysis was performed for individual glycans and derived traits. Standardized values were normalized, and statistical analyses were conducted using R version 3.3.2 and RStudio.
Results:
A total of 101 N-glycans were relatively quantified, and logistic regression analysis revealed significant differences in specific glycan compositions between breast cancer patients and healthy controls. Notably, H6N5F1L3, H6N5L2E1, and H7N6F1L1E3 exhibited distinct patterns in breast cancer patients. However, the study did not identify a consistent glycomic signature distinguishing breast cancer cases from controls. The observed variability in glycosylation profiles may be attributed to the heterogeneity of breast cancer subtypes and the small size of patient cohorts.
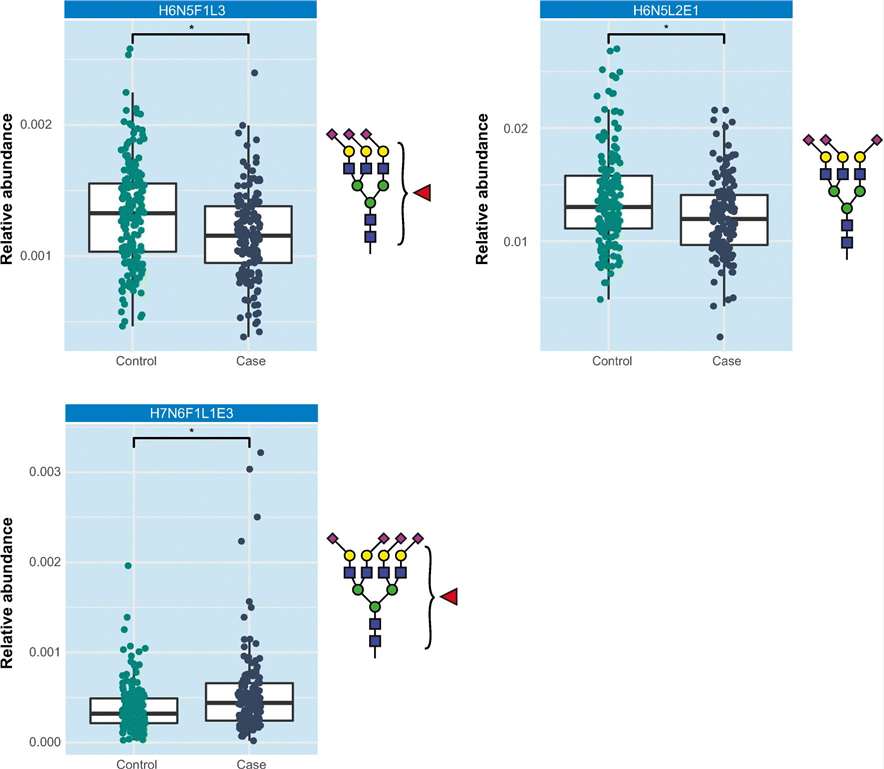 Association of H6N5F1L3, H6N5L2E1 and H7N6F1L1E3 with breast cancer
Association of H6N5F1L3, H6N5L2E1 and H7N6F1L1E3 with breast cancer
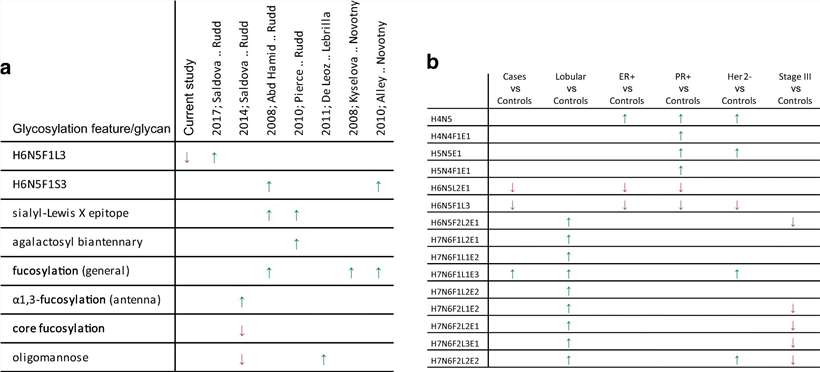 a Comparison of previously reported data and results of the current study. b Significant direct traits (glycan compositions) for specific breast cancer subtypes and stages as determined in a Student's t-test
a Comparison of previously reported data and results of the current study. b Significant direct traits (glycan compositions) for specific breast cancer subtypes and stages as determined in a Student's t-test
Reference
- Vreeker, Gerda CM, et al. "Serum N-glycan profiles differ for various breast cancer subtypes." Glycoconjugate Journal 38 (2021): 387-395.




