- Service Details
- Case Study
Introduction of Intact Glycoproteins
Intact glycoproteins, complex biomolecules characterized by the covalent attachment of oligosaccharide chains to protein backbones, embody a structural and functional diversity crucial to myriad biological processes. Their intricate glycan decorations dictate spatial organization and molecular interactions, influencing cellular recognition, signaling, and immune response modulation. Beyond their physiological roles, aberrant glycosylation patterns in intact glycoproteins are implicated in diseases, fueling interest in their analysis for biomarker discovery and therapeutic targeting.
Technology Platform for Intact Glycoproteins Analysis
To meet the challenges posed by intact glycoproteins, we have developed a technological platform for the thorough analysis of intact glycoproteins. Our technology relies on advanced equipment and techniques, exploiting the best of mass spectrometry, proteomics, and glycomics, assimilating them into a collective effort to truly understand these molecules.
We employ advanced mass spectrometry like LTQ Orbitrap XL, Q Exactive hybrid quadrupole-Orbitrap MS equipped with nano-LC system, MALDI-TOF/TOF MS, that allow for accurate m/z measurements and full MS scan capabilities, thereby enabling the identification and characterization of both the peptide backbone and glycan structures simultaneously. Furthermore, subsequent fragmentation via Higher Energy Collision Dissociation (HCD) or Electron Transfer Dissociation (ETD) techniques provide essential information for the localization of glycosylation sites that yields a more comprehensive picture of these highly complex biomolecules.
Intact Glycoproteins Analysis by Creative Proteomics
- Intact Glycoprotein Identification: We perform protein extraction and enrichment, followed by identification using advanced MS techniques for comprehensive analysis.
- Glycoprotein Quantitation: Creative Proteomics' intact glycoprotein analysis service can also accurately measure the concentrations of these glycoproteins within a biological sample.
- Glycoprotein Characterization: For a more complete understanding of glycoproteins, Creative Proteomics' technology can delineate the chemical characteristics of glycoproteins, ensuring a detailed understanding of their properties.
- Glycosylation Site Analysis: Creative Proteomics' technology can also adeptly pinpoint the specific sites of glycosylation on a given protein.
- Glycan Structure Analysis: We perform glycan release, labeling, and cleanup to analyze the glycan composition.
Sample Requirements for Intact Glycoproteins Analysis
| Sample Type | Sample Matrix | Sample Volume | Additional Information |
|---|---|---|---|
| Serum/Plasma | Blood | 100 μL - 500 μL | Commonly used for systemic glycoprotein analysis; suitable for biomarker discovery. |
| Cell Lysates | Cell culture | 1 mg - 10 mg | Reflect intracellular glycoprotein composition; useful for studying cell-specific glycosylation. |
| Tissue Homogenates | Tissue | 10 mg - 50 mg | Provide insights into tissue-specific glycoprotein expression; suitable for organ-level analysis. |
| Biofluids (Urine, CSF) | Urine, cerebrospinal fluid | 500 μL - 1 mL | Reflect systemic and localized glycosylation patterns; useful for biomarker discovery. |
| Cell Culture Supernatants | Cell culture | 1 mL - 5 mL | Secreted glycoproteins; valuable for biopharmaceutical production and disease-related studies. |
| Saliva | Oral cavity | 100 μL - 500 μL | Reflects oral health status and systemic diseases; potential biomarkers for various conditions. |
| Synovial Fluid | Joint cavities | 100 μL - 500 μL | Implicated in joint health and inflammatory diseases; aids in diagnosis and treatment monitoring. |
| Tears | Ocular surface | 50 μL - 100 μL | Essential for ocular surface protection and immune defense; informative for ophthalmic research. |
| Milk | Mammary glands | 100 μL - 500 μL | Rich source of glycoproteins; crucial for infant nutrition and breastfeeding-related health. |
Applications of Intact Glycoproteins Analysis
Biomarker Discovery
Identifying disease-specific glycan signatures for early diagnosis, prognosis, and therapeutic monitoring.
Drug Target Identification
Elucidating glycan-mediated interactions to identify and validate glycoprotein targets for drug development.
Biopharmaceutical Characterization
Ensuring product quality and regulatory compliance of therapeutic proteins through glycoprotein characterization.
Disease Mechanism Elucidation
Unraveling the molecular mechanisms of disease progression and treatment resistance.
Case. Glycosylation Patterns and Genetic Polymorphism in Human Alpha-1-Acid Glycoprotein (AGP) Using Mass Spectrometry
Background:
Human alpha-1-acid glycoprotein (AGP) is a highly glycosylated protein with significant genetic polymorphism. Variations in glycosylation patterns and genetic makeup of AGP have been linked to various physiological conditions and diseases. Therefore, understanding these variations is crucial for clinical research and healthcare applications.
Samples:
Samples included AGP isolated from pooled human plasma and individual plasma samples obtained from different donors. Genetic variants of AGP (AGP-1 and AGP-2) were analyzed to assess glycosylation and genetic polymorphism.
Technical Method:
AGP samples were prepared for mass spectrometry analysis by dissolving in a 1:1 mixture of deionized water and acetonitrile.
Mass spectrometry experiments were performed using an ESI/qTOF instrument in negative ion mode.
De-sialylation of AGP samples was achieved using non-specific sialidase treatment.
Chromatographic separation was performed using a C8 column, followed by online MS detection.
Spectral data were analyzed using Bruker software and MATLAB-based scripts.
Isotopic distributions were calculated to determine glycan compositions and quantify glycan structures.
Genetic variants were identified and characterized based on amino acid sequences and post-translational modifications.
Partial separation of genetic variants was achieved on a standard reversed-phase column.
Results:
Significant heterogeneity in glycosylation patterns was observed among AGP samples, with variations in fucosylation and number of antennae.
Genetic polymorphism was evident, with differences in genetic variants (ORM1F1, ORM1S, ORM2) among individual plasma samples.
The degree of sialylation was determined for intact AGP, revealing glycan heterogeneity and genetic polymorphism.
Multiple glycan compositions were identified simultaneously for different genetic variants, providing insights into AGP diversity.
The analytical method showed improved ionization properties for de-sialylated AGP compared to fully sialylated AGP in positive ion mode, enabling comprehensive analysis of glycosylation patterns and genetic variability.
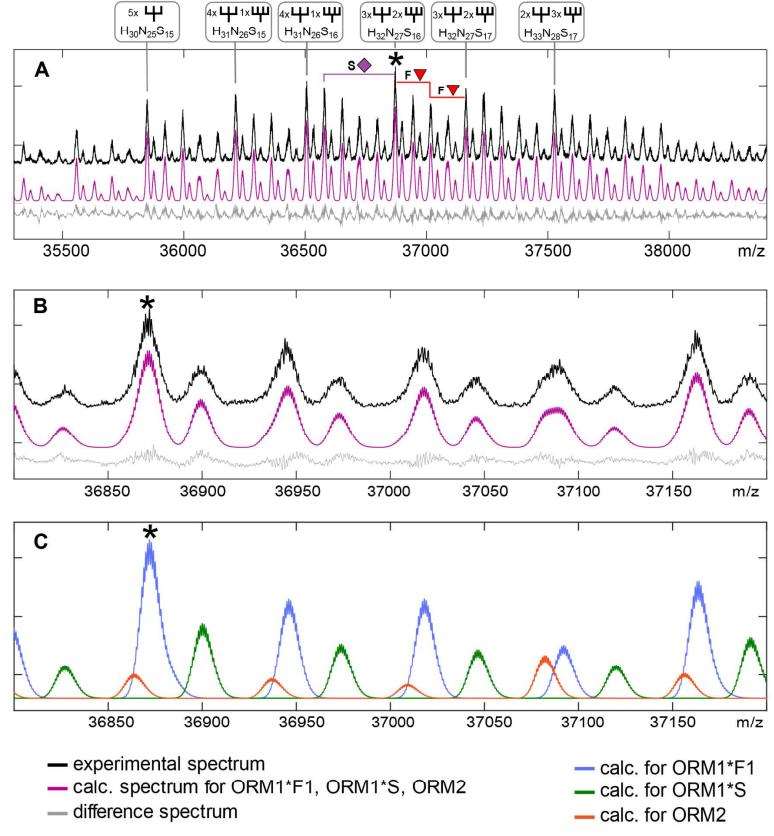 ESI-qTOF-MS spectrum of the highly sialylated alpha-1-acid glycoprotein from pooled plasma in negative ion mode
ESI-qTOF-MS spectrum of the highly sialylated alpha-1-acid glycoprotein from pooled plasma in negative ion mode
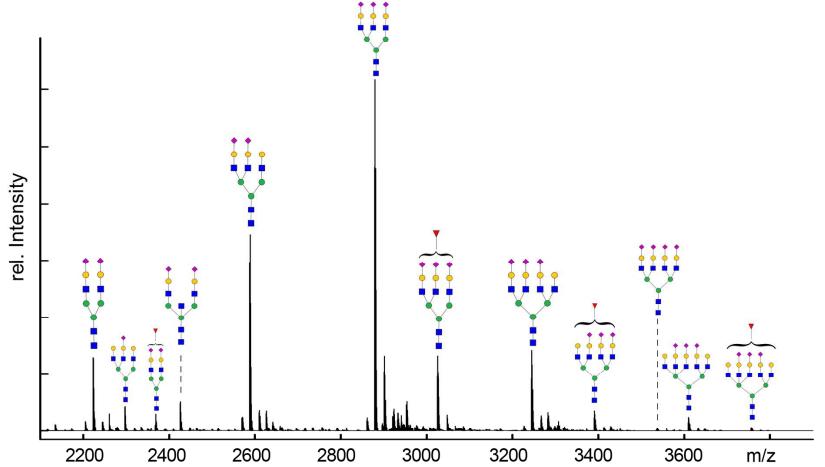 Deconvoluted ESI-MS spectrum of PNGase F released glycans of AGP from pooled human plasma
Deconvoluted ESI-MS spectrum of PNGase F released glycans of AGP from pooled human plasma
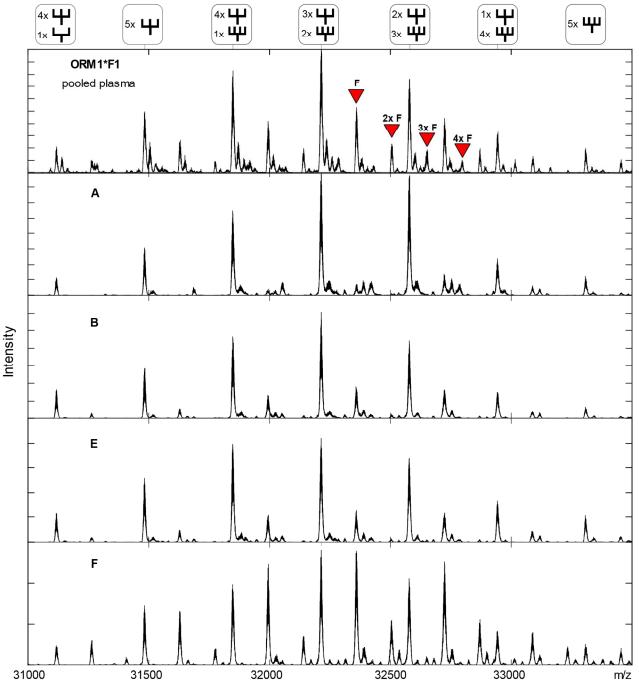 Spectra of the de-sialylated AGP-1 for variant ORM1*F1 from different samples.
Spectra of the de-sialylated AGP-1 for variant ORM1*F1 from different samples.
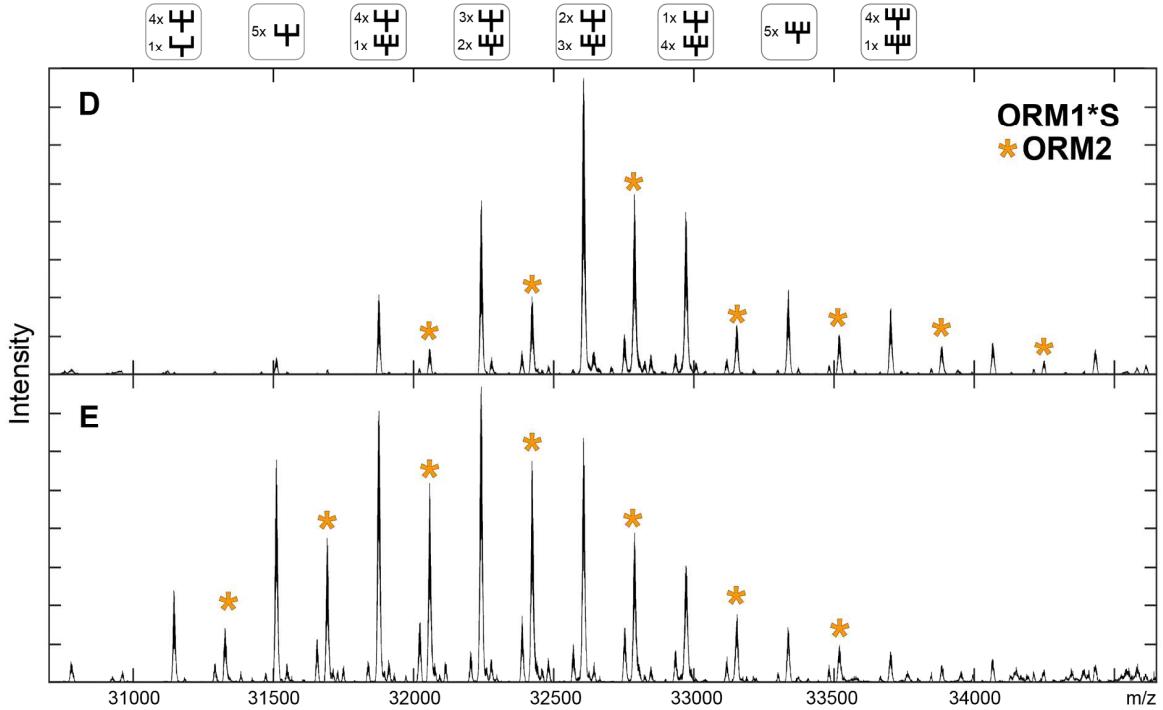 Spectra of the de-sialylated AGP for variant ORM1*S and ORM2 for sample D and E. Most abundant signals for ORM2 are marked with an orange asterisk with a mass shift of + 182 Da
Spectra of the de-sialylated AGP for variant ORM1*S and ORM2 for sample D and E. Most abundant signals for ORM2 are marked with an orange asterisk with a mass shift of + 182 Da
Reference
- Baerenfaenger, Melissa, and Bernd Meyer. "Intact human alpha-acid glycoprotein analyzed by ESI-qTOF-MS: simultaneous determination of the glycan composition of multiple glycosylation sites." Journal of proteome research 17.11 (2018): 3693-3703.




