- Service Details
- Case Study
What are Glycosides?
Glycosides are molecules consisting of a sugar moiety (glycone) bonded to a non-sugar moiety (aglycone) through a glycosidic linkage. They encompass a broad spectrum of compounds found in plants, animals, and microorganisms, exhibiting diverse chemical structures and biological activities. Glycosides serve multifaceted functions in living organisms, including energy storage, structural integrity, cell-cell recognition, and defense mechanisms against predators and pathogens.
The structural diversity of glycosides confers them with a wide array of pharmacological properties, making them invaluable targets for drug discovery and development. Additionally, glycosides play crucial roles in food chemistry, flavor enhancement, and natural product synthesis, underscoring their significance across various industries.
Glycoside analysis is critical in helping to elucidate the chemical composition, structural features and biological activities of these complex molecules. By employing advanced analytical techniques, we can unravel the intricacies of glycosidic bonds, identify specific sugars and glycosides, and determine the stereochemistry of glycosidic bonds.
Glycoside analysis also helps to characterize natural products, elucidate biosynthetic pathways and discover novel bioactive compounds with therapeutic potential. It enables quality control of herbal medicines, dietary supplements and pharmaceutical preparations to ensure their safety, efficacy and compliance.
Glycosides Analysis Services by Creative Proteomics
Creative Proteomics offers a one-stop shop for customized glycoside analysis to meet the diverse needs of researchers and industries worldwide.
Glycoside Identification
We use a combination of high-resolution mass spectrometry (HR-MS) and chromatographic techniques such as liquid chromatography (LC) and gas chromatography (GC) to identify and characterize glycosides in complex biological samples. By combining precise mass measurements, fragmentation analysis and database search algorithms, glycoside structures can be quickly and accurately identified, even in trace amounts.
Glycoside Quantification
Targeted glycosides in a variety of matrices, including plant extracts, foods and pharmaceutical preparations, are precisely quantified using validated LC-MS/MS methods. By using stable isotope-labeled internal standards and calibration curves, we ensure the accuracy, reproducibility, and sensitivity of quantitative measurements to facilitate quantitative structure-activity relationship (QSAR) studies and pharmacokinetic assessments.
Glycoside Profiling
We offer comprehensive glycoside analysis services, including systematic analysis of the composition, distribution and abundance of glycosides in complex biological samples. By using multi-dimensional chromatography coupled with high-resolution mass spectrometry, we provide in-depth analysis of glycoside subclasses, isomers and positional isomers, which provides valuable information for understanding the metabolic pathways, biosynthetic mechanisms and physiological functions of glycosides in organisms.
Technical Platforms for Glycoside Analysis
High-Performance Liquid Chromatography (HPLC):
HPLC is one of the most widely used techniques for glycoside analysis due to its high sensitivity, resolution, and versatility.
- Reverse-phase HPLC (RP-HPLC) is often employed, utilizing a non-polar stationary phase and a polar mobile phase for separation based on hydrophobic interactions.
- Normal-phase HPLC can also be utilized for glycoside analysis, separating compounds based on their polarity.
- Detection methods such as UV-Vis spectroscopy, fluorescence, or mass spectrometry (MS) can be coupled with HPLC for identification and quantification of glycosides.
Mass Spectrometry (MS):
MS, coupled with techniques like liquid chromatography (LC-MS) or gas chromatography (GC-MS), is invaluable for identifying and quantifying glycosides. MS provides molecular weight information and can elucidate glycoside structures based on fragmentation patterns. Various ionization techniques such as electrospray ionization (ESI) or matrix-assisted laser desorption/ionization (MALDI) can be utilized depending on the sample type and instrumentation available.
Nuclear Magnetic Resonance Spectroscopy (NMR):
NMR spectroscopy provides valuable structural information about glycosides, including the configuration of the glycosidic bond and the aglycone structure. NMR analysis requires relatively pure samples and is often used in conjunction with other techniques for comprehensive glycoside characterization.
Sample Requirements for Glycosides Analysis
| Sample Type | Recommended Sample Amount |
|---|---|
| Plant Extracts | 10-50 mg |
| Herbal Materials | 100-500 mg |
| Food Products | 5-20 g |
| Biological Fluids | 0.1-1 mL |
| Pure Compounds | 1-10 mg |
| Tissue Samples | 10-50 mg |
| Fermentation Broths | 1-10 mL |
| Environmental Samples | Varies (Dependent on matrix) |
Applications of Glycosides Analysis
Pharmaceutical Industry:
- Drug Discovery: Glycosides are abundant in medicinal plants and play crucial roles in traditional medicine. Analyzing glycosides can help identify bioactive compounds with potential therapeutic properties.
- Quality Control: Analyzing glycosides ensures the consistency and quality of herbal medicines and botanical extracts.
Food Science:
- Food Authentication: Detection and quantification of glycosides can be used for authentication and quality control of food products, especially those derived from plants.
- Flavor and Aroma: Glycosides contribute to the flavor and aroma of many foods. Analyzing glycosides helps in understanding flavor development and enhancing food products.
Natural Product Chemistry:
- Chemical Ecology: Glycosides are involved in plant defense mechanisms and ecological interactions. Analyzing glycosides aids in studying plant-insect interactions and chemical ecology.
- Isolation and Structural Elucidation: Glycosides are often isolated from natural sources for structural elucidation and bioactivity studies.
Environmental Monitoring:
- Bioremediation: Some glycosides are involved in the metabolism and detoxification of environmental pollutants. Analyzing glycosides can provide insights into bioremediation processes.
- Phytoremediation: Plants containing glycosides may be used for phytoremediation of contaminated soils. Glycoside analysis helps assess the efficiency of this process.
Biotechnology:
- Enzyme Engineering: Glycoside analysis is essential for studying enzyme kinetics and substrate specificity, which is valuable for enzyme engineering and biocatalysis.
- Glycoengineering: Understanding glycoside structures and biosynthesis pathways is crucial for glycoengineering applications, such as the production of therapeutic glycoproteins.
Phytochemical Studies:
- Bioactivity Screening: Glycoside analysis facilitates the identification of bioactive compounds from natural sources for various applications, including drug development and nutraceuticals.
- Structure-Activity Relationship (SAR) Studies: Analyzing the structure-activity relationships of glycosides helps in optimizing their biological activities and pharmacological properties.
Title: Investigation of Neuroprotective and Anti-inflammatory Effects of Chestnut Extracts
Background
Chestnut extracts have shown potential neuroprotective and anti-inflammatory properties in various studies. However, a comprehensive investigation into the effects of different chestnut extracts on cell viability and inflammatory markers is lacking.
Sample
Samples were collected from chestnut leaves and spiny burs obtained from an experimental grove in Granaglione, Italy. The leaves were freeze-dried, ground, and stored at -80°C, while spiny burs were dried and ground before analysis.
Technical Methods
1. Chemicals and Materials: Deuterated solvents, cell culture media, antibodies, and standards were sourced from reputable suppliers.
2. Sampling and NMR Metabolomics: Samples were prepared and subjected to NMR metabolomics analysis after extraction using a MeOH-d4/H2O-d2 solvent system.
3. Pre-Purification of Flavonoids from Leaves: A liquid-liquid partition method followed by MPLC fractionation was employed to purify flavonoids from chestnut leaves.
4. NMR and MS Spectra Measurement: NMR and MS spectra were acquired using appropriate instruments and parameters.
5. NMR Processing and Multivariate Data Treatment: Data processing involved Fourier transformation, phase correction, baseline correction, and calibration. Multivariate analysis using OPLS-DA was performed.
6. Cell Culture: BV-2 murine microglial cells were cultured in DMEM supplemented with FBS, glutamine, and antibiotics.
7. Cell Viability: Cell viability was assessed using the MTT assay after treatment with chestnut extracts and LPS.
8. RT-PCR Analysis: Transcriptional levels of inflammatory markers (IL-1β and TNF-α) were evaluated using RT-PCR.
9. Western Blot Analysis: Protein expression of NF-kB was determined by Western blotting.
Results
The study revealed distinct metabolomic profiles between chestnut leaves ('Marrone' and 'Castagna'), with differences in flavonoid content. Chestnut extracts from both leaves and spiny burs demonstrated cytoprotective and anti-inflammatory effects on microglial cells, as evidenced by cell viability assays and decreased expression of inflammatory markers (IL-1β and TNF-α). Additionally, the extracts showed a reduction in NF-kB expression, indicating their potential to modulate pro-inflammatory pathways. These findings highlight the therapeutic potential of chestnut extracts in combating neuroinflammation and support their utilization in waste valorization strategies.
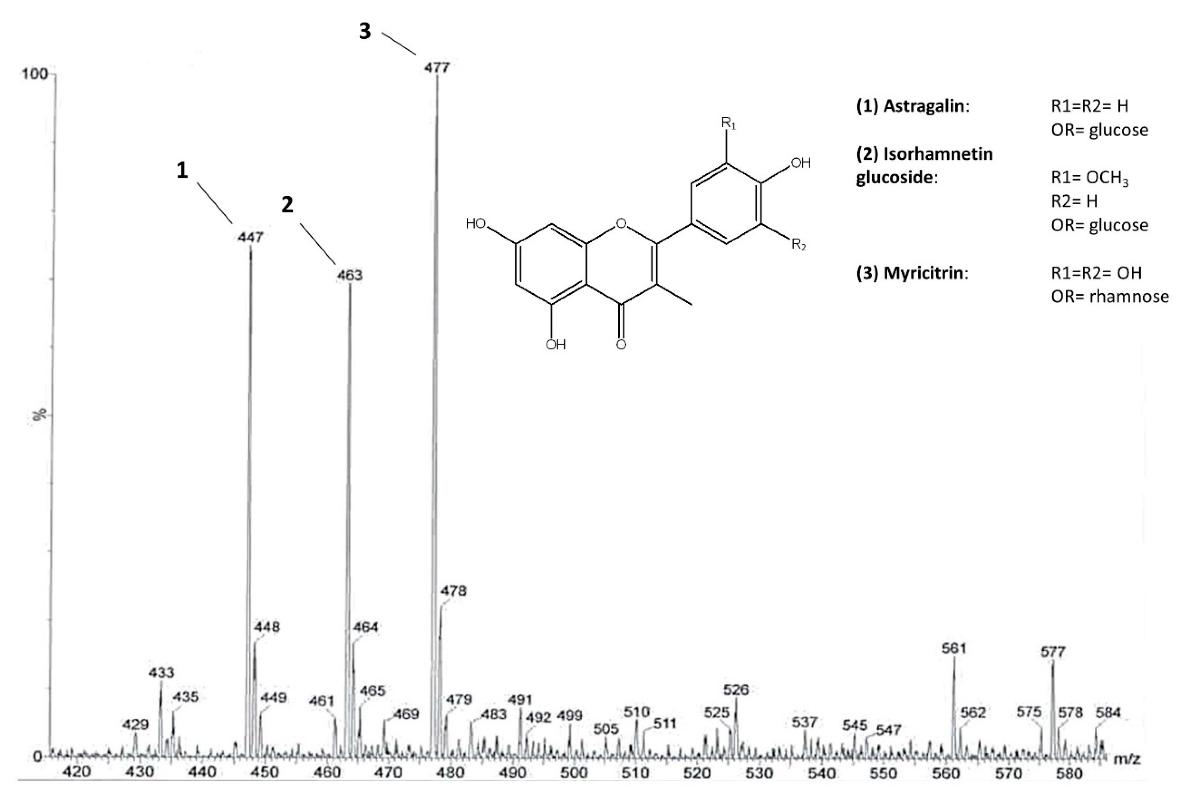 The main flavonoid glycosides identified in C. sativa leaves. ESI-MS spectrum of the flavonoid fraction obtained after direct infusion.
The main flavonoid glycosides identified in C. sativa leaves. ESI-MS spectrum of the flavonoid fraction obtained after direct infusion.
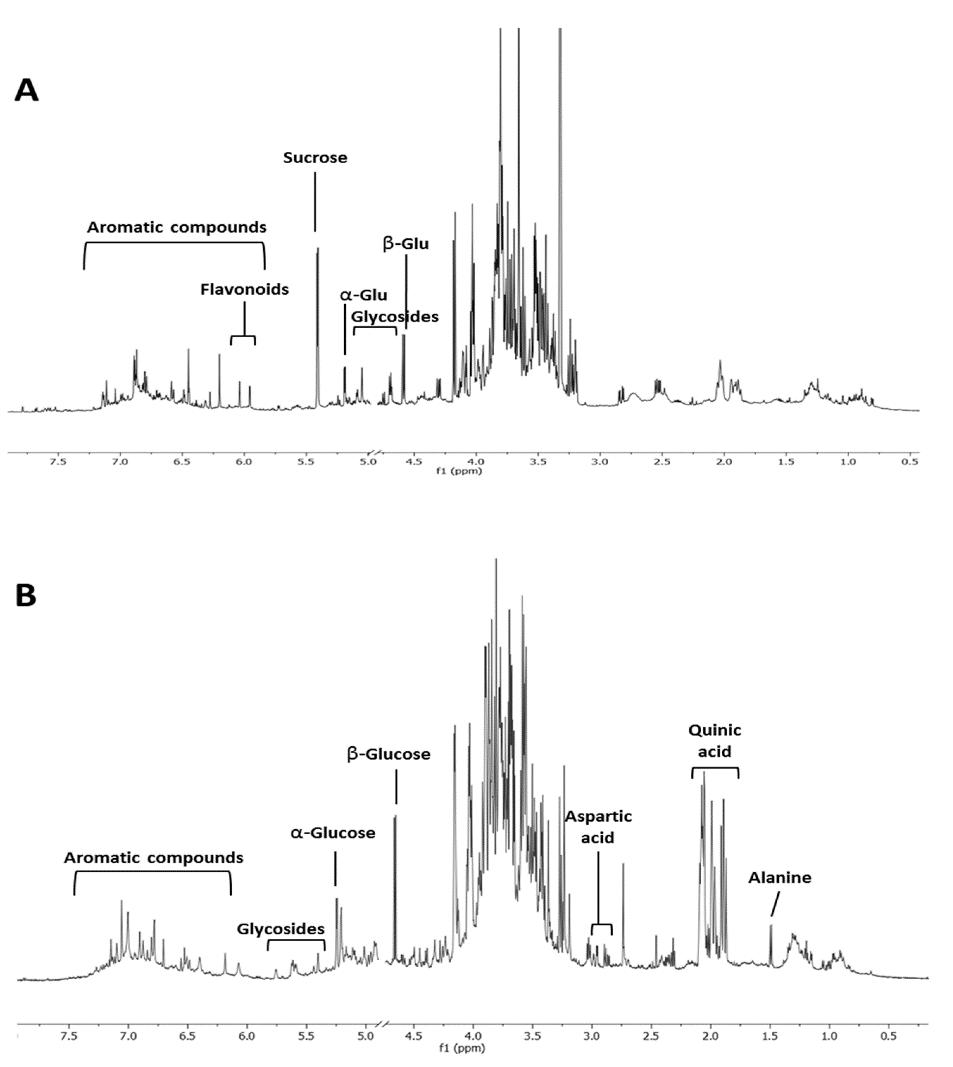 1H NMR profiling of C. sativa of representative leaves (A) and spiny burs (B) hydroalcoholic extract.
1H NMR profiling of C. sativa of representative leaves (A) and spiny burs (B) hydroalcoholic extract.
Reference
- Chiocchio, Ilaria, et al. "Leaves and spiny burs of Castanea Sativa from an experimental chestnut grove: Metabolomic analysis and anti-neuroinflammatory activity." Metabolites 10.10 (2020): 408.




