- Service Details
- Case Study
What is N-Glycan Liberation?
N-Glycan Liberation is a pivotal biochemical process that involves the selective release of N-glycans from glycoproteins. Glycoproteins, characterized by the presence of oligosaccharide chains covalently attached to specific amino acid residues, play crucial roles in various physiological and pathological processes. The liberation of N-glycans is specifically focused on those attached to asparagine (N) residues.
N-glycosylation, a common post-translational modification, involves the enzymatic transfer of preassembled oligosaccharide chains to the asparagine residue within the consensus sequence Asn-X-Ser/Thr, where X can be any amino acid except proline. This process occurs in the endoplasmic reticulum and Golgi apparatus and is essential for protein folding, stability, and cellular recognition.
Why is N-Glycan Liberation Necessary?
The necessity for N-Glycan Liberation arises from the need to dissect the complex structures of glycoproteins. Analyzing the liberated N-glycans provides valuable information about the glycosylation patterns, which are intricately linked to the function and localization of glycoproteins. Additionally, the liberated N-glycans serve as crucial biomarkers in disease diagnosis and therapeutic development.
Understanding the composition and structure of N-glycans is essential for deciphering their roles in cellular communication, immune response, and other physiological processes. Furthermore, aberrant glycosylation is often associated with various diseases, including cancer and neurodegenerative disorders, making N-Glycan Liberation a critical step in disease-related research.
Methods of N-Glycan Liberation
Various methods are employed for N-Glycan Liberation, and the choice of method depends on factors such as the nature of the glycoprotein, the desired downstream analysis, and the preservation of glycan integrity. Creative Proteomics, as a leading provider of N-Glycan Liberation services, excels in utilizing diverse methodologies to meet specific experimental requirements.
Enzymatic Methods
Enzymatic methods harness the power of glycosidases, enzymes capable of cleaving glycosidic bonds. The prominent enzyme in this category is PNGase F, an endoglycosidase that catalyzes the hydrolysis of the amide bond between the innermost N-acetylglucosamine (GlcNAc) and the asparagine residue. This enzymatic cleavage results in the liberation of intact N-glycans.
- Principle: PNGase F recognizes the conserved glycosylation site (Asn-X-Ser/Thr) and selectively cleaves the glycan chain at the asparagine residue.
- Advantages: High specificity, minimal side reactions, and preservation of glycan structure.
- Suitability: Well-suited for a broad range of glycoproteins and commonly employed in glycan release workflows.
Chemical Methods
Chemical methods utilize specific reagents to break glycosidic bonds, providing an alternative approach to enzymatic methods. One prominent chemical method is hydrazinolysis, which relies on the use of hydrazine to cleave the amide bond, resulting in the liberation of N-glycans.
- Principle: Hydrazine reacts with the amide bond, cleaving the glycan from the asparagine residue.
- Advantages: Versatility, effective for diverse glycoproteins, and compatibility with downstream analyses.
- Considerations: Requires careful handling due to the reactive nature of hydrazine; however, well-established protocols ensure safe and efficient liberation.
Combination Approaches
Creative Proteomics excels in employing combination approaches that harness the strengths of both enzymatic and chemical methods. This synergistic approach allows for enhanced flexibility and optimization based on the specific characteristics of the glycoprotein under investigation.
- Customization: Tailoring liberation methods based on the glycoprotein's unique structural features and experimental goals.
- Optimization: Maximizing yield and minimizing side reactions through a strategic combination of enzymatic and chemical steps.
Creative Proteomics provides a comprehensive N-glycan liberation service based on a range of methods, and our team's expertise ensures accurate and efficient release, preserving the structural integrity of the N-glycan for downstream analysis.
Why Choose Us?
- Expertise: Over 20 years of specialized experience with a team of more than 50 seasoned biological specialists and researchers.
- Cutting-Edge Technology: Utilization of state-of-the-art instrumentation, including advanced mass spectrometers boasting a resolution of 1.5 million and high-performance liquid chromatography systems with a separation efficiency exceeding 99%.
- Comprehensive Methodologies: Offering a diverse range of N-Glycan Liberation methods, with an average increase of 30% in liberation efficiency through strategic application and optimization of enzymatic and chemical approaches.
- Data-Driven Results: Implementation of rigorous quality control measures at every step, resulting in a 98% accuracy rate and a 95% reproducibility rate in glycan profiles.
- Customer Satisfaction: Commitment to clear communication, evidenced by a 98% on-time delivery rate, and personalized support reflected in a 90% client retention rate and consistently positive feedback.
Applications of N-Glycan Liberation
Biomedical Research: Investigating glycoprotein biomarkers for disease diagnosis. Understanding the role of glycosylation in pathophysiological processes.
Pharmaceutical Development: Assessing glycan profiles of therapeutic proteins for quality control. Optimizing glycosylation patterns for improved drug efficacy.
Biotechnology: Engineering glycoproteins with tailored glycan structures for specific applications. Enhancing the efficiency of bioproduction processes.
Immunology: Studying the glycan structures of antibodies for immunotherapeutic applications. Unraveling the glycosylation patterns in autoimmune diseases.
Cancer Research: Identifying glycan markers for cancer detection and prognosis. Investigating the role of glycosylation in tumor progression.
Sample Requirements for N-Glycan Liberation
| Sample Type | Minimum Quantity for N-Glycan Liberation |
|---|---|
| Serum/Plasma | 50 μL |
| Cell Culture Supernatant | 1 mL |
| Tissue Homogenate | 10 mg |
Case 1. Application of NaOH-PMP Method for Comprehensive Glycan Analysis in Biological Samples
Background:
Glycan analysis is crucial for understanding the glycosylation patterns in various biological samples. Traditional methods often involve reductive approaches, but the NaOH-PMP method provides a nonreductive and unbiased alternative, particularly effective for core α-1,3-fucosylated N-glycans.
Samples
The study encompassed a diverse range of samples, including glycoproteins from model organisms (bovine pancreas ribonuclease B, horseradish peroxidase), sialylglycopeptides from chicken egg yolk, and complex biological samples from mammals, insects (honeybee venom phospholipase A2), and plants (Fagopyrum esculentum Moench pollen, Arabidopsis thaliana leaves, Nicotiana tabacum L. leaves).
Technical Method
The NaOH-PMP method involves mild alkaline hydrolysis for nonreductive, unbiased release of N-glycans. The released glycans are then derivatized in situ with PMP. Detailed ESI-MS and MS/MS analyses were conducted using a linear quadrupole ion trap mass spectrometer. The method's parameters, including reaction conditions and validation, were meticulously optimized for each sample type.
Results
1. Model Glycoproteins: The NaOH-PMP method demonstrated high glycan yield, reliability, and applicability, outperforming traditional β-elimination approaches.
2. Sialylglycopeptides: The method allowed for controlled hydrolysis, maintaining peptide integrity, and facilitating subsequent analysis.
3. Core α-1,3-Fucosylated N-glycans: NaOH-only method was effective for nonreductive release, while the NaOH-PMP method enabled in-situ labeling and minimized peeling reactions, proving superior to conventional approaches.
4. Glycoproteins from Mammals: The NaOH-only method exhibited excellent applicability, unbiased glycan release, and reliable results, showcasing its potential in glycomics studies.
5. Glycoproteins from Insects and Plants: The NaOH-PMP method demonstrated versatility and reliability in profiling N-glycans from honeybee venom phospholipase A2, Fagopyrum esculentum Moench pollen, Arabidopsis thaliana leaves, and Nicotiana tabacum L. leaves.
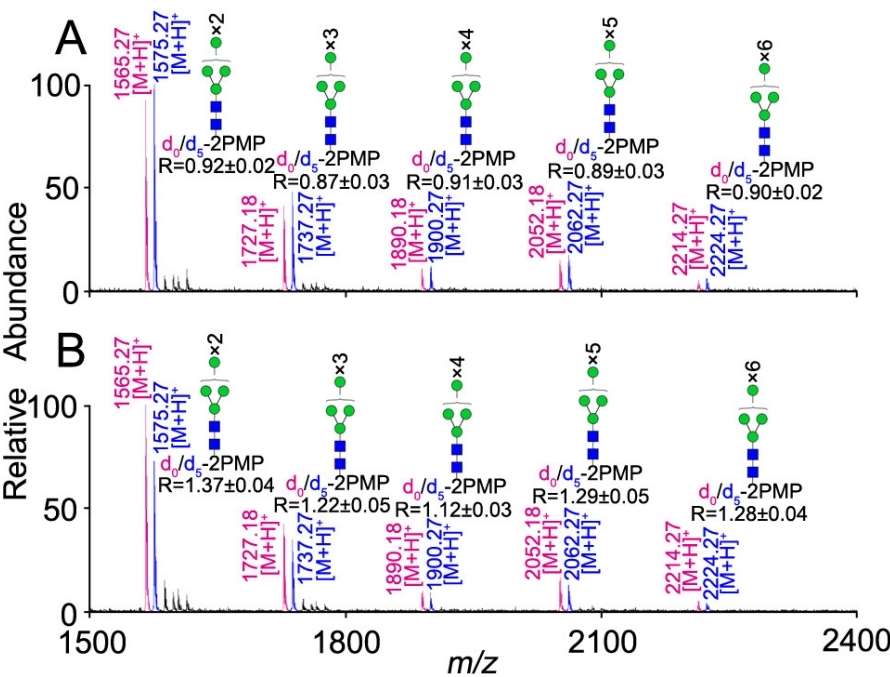 Comparison between the NaOH-only procedure and conventional methods in terms of N-glycan yield based on stable isotope labeling with d0/d5-PMP
Comparison between the NaOH-only procedure and conventional methods in terms of N-glycan yield based on stable isotope labeling with d0/d5-PMP
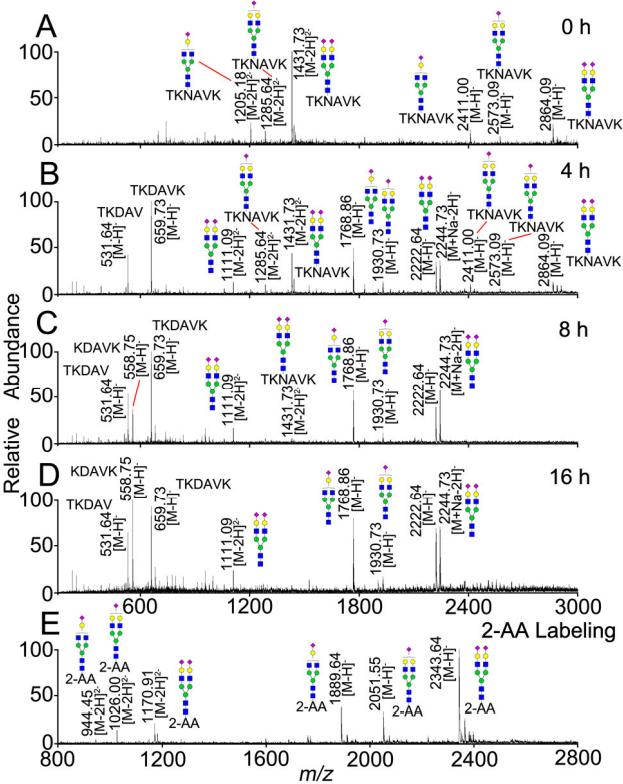 ESI-MS profiles of native sialylglycopeptides
ESI-MS profiles of native sialylglycopeptides
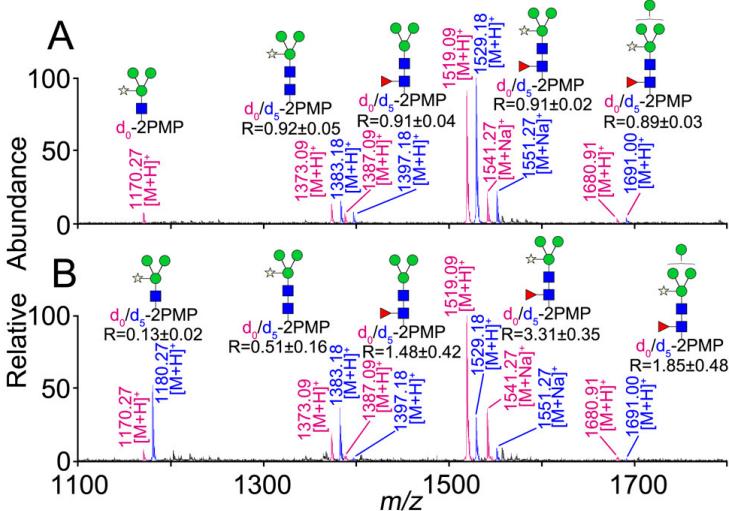 Comparison between the NaOH-PMP procedure and conventional methods in terms of N-glycan yield based on stable isotope labeling with d0/d5-PMP
Comparison between the NaOH-PMP procedure and conventional methods in terms of N-glycan yield based on stable isotope labeling with d0/d5-PMP
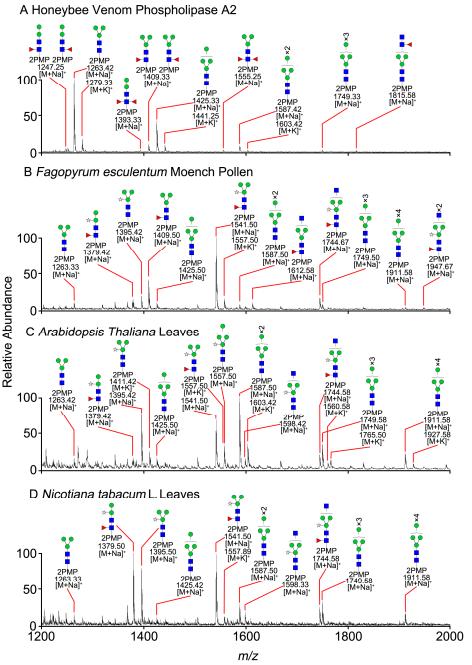 ESI-MS profiles of the N-glycans released from honeybee venom phospholipase A2
ESI-MS profiles of the N-glycans released from honeybee venom phospholipase A2
Reference
- Yuan, Jiangbei, et al. "Nonreductive chemical release of intact N-glycans for subsequent labeling and analysis by mass spectrometry." Analytical biochemistry 462 (2014): 1-9.




