- Service Details
- Case Study
What is Glycan Sulfation Modification?
Glycan sulfation modification refers to a crucial biochemical process involving the addition of sulfate groups to complex carbohydrates known as glycans. These glycans are typically attached to proteins or lipids and play pivotal roles in various biological functions within living organisms. Glycan sulfation is a post-translational modification, meaning it occurs after proteins are synthesized, contributing to the diversity of molecular structures and functions in cells.
The sulfation of glycans is a highly regulated and specific process that occurs in the Golgi apparatus of the cell. Sulfate groups are covalently attached to specific positions on the glycan structure, creating sulfated glycans. This modification significantly influences the physicochemical properties and biological activities of glycans, thereby impacting cellular processes.
Glycan sulfation analysis is pivotal for understanding its roles in cellular processes and diseases. Sulfated glycans influence cell signaling, immune response, and development. Accurate analysis is essential for identifying disease biomarkers and developing targeted therapies. Gain valuable insights for advancements in glycobiology and clinical applications through precise glycan sulfation analysis.
Technical Platform for Glycan Sulfation Analysis
Glycan sulfation analysis employs advanced analytical techniques to characterize and quantify sulfated glycans. Key methods include mass spectrometry, liquid chromatography, and capillary electrophoresis. These techniques allow for the separation and identification of individual sulfated glycans, providing a comprehensive profile of the sulfation patterns.
Cutting-edge instrumentation and sophisticated software enable high-resolution analysis, ensuring precise identification of sulfation sites and determination of sulfation heterogeneity. The integration of multiple analytical platforms enhances the accuracy and reliability of glycan sulfation analysis.
LC–MS/MS (Liquid Chromatography–Tandem Mass Spectrometry):
- High-Resolution LC–MS/MS System: Enables precise separation and identification of sulfated glycans.
- Tandem Mass Spectrometer: Facilitates sequential mass spectrometric analysis for enhanced characterization.
MALDI-TOF Mass Spectrometer (Matrix-Assisted Laser Desorption/Ionization Time-of-Flight):
- MALDI-TOF Mass Spectrometer: Utilizes laser desorption and ionization for accurate mass determination of sulfated glycans.
- Reflectron TOF Analyzer: Enhances resolution and accuracy in determining glycan structures.
HPLC (High-Performance Liquid Chromatography):
- High-Performance Liquid Chromatograph (HPLC) System: Employs high-pressure pumps for efficient separation of sulfated glycans.
- UV or Fluorescence Detector: Detects and quantifies separated glycans with high sensitivity.
CE-MS (Capillary Electrophoresis–Mass Spectrometry):
- Capillary Electrophoresis System: Provides high-resolution separation of sulfated glycans based on electrophoretic mobility.
- Mass Spectrometer: Detects and characterizes sulfated glycans with precision.
Advanced Software for Data Analysis:
- Data Analysis Software (e.g., Proteome Discoverer, Byonic): Processes and interprets complex data generated by LC–MS/MS, MALDI-TOF, and other platforms.
Applications of Glycan Sulfation Modification Analysis
Disease Biomarker Discovery: Glycan sulfation analysis identifies specific patterns associated with diseases, serving as potential biomarkers for early detection and personalized medicine.
Therapeutic Glycoprotein Monitoring: Ensures the quality of therapeutic glycoproteins through monitoring glycan sulfation, impacting the efficacy and safety of biopharmaceuticals.
Understanding Cellular Processes: Illuminates the role of glycan sulfation in cell signaling, immune response, and development, deepening our understanding of normal physiology and disease.
Glycan Engineering for Biotechnology: Guides the modification of glycans in biotechnology, ensuring targeted engineering for improved therapeutic and diagnostic applications.
Investigation of Glycan Heterogeneity: Analyzes the heterogeneity introduced by sulfation, providing insights into the variations in glycan structures in biological systems.
Drug Development and Targeted Therapies: Identifies specific glycan targets for drug development, enhancing the precision of therapeutic interventions and targeted therapies.
Functional Studies in Glycobiology: Essential for functional studies, allowing researchers to explore the roles of sulfated glycans in cellular interactions and signal transduction.
Sample Requirements for Glycan Sulfation Modification Analysis
| Sample Type | Sample Size Recommendations |
|---|---|
| Serum/Plasma | 10-100 μL |
| Tissue Homogenates | 5-50 mg |
| Cell Culture Supernatant | 1-10 mL |
| Urine | 5-50 mL |
| Cerebrospinal Fluid (CSF) | 0.5-2 mL |
| Saliva | 1-5 mL |
| Synovial Fluid | 0.5-5 mL |
Case. Characterization of Sialylated Sulfoglycopeptides: Enrichment Challenges and MS2 Analysis
Background:
The study focuses on the characterization of sialylated sulfoglycopeptides, investigating challenges in their enrichment and employing mass spectrometry (MS2) analysis to overcome identification difficulties. Sialylated sulfoglycopeptides are of interest due to their functional implications in biological processes.
Samples:
Two glycoproteins, recombinant erythropoietin (rEPO) produced in BHK21 cells and bovine thyroglobulin (bTG), were selected as samples. rEPO, known for high sulfation and low sialylation, served as an ideal model to evaluate sulfate-based enrichment. bTG, a larger glycoprotein, provided a source of sialylated and sulfated glycopeptides for further analysis.
Technical Method:
Enrichment of Sulfated Glycopeptides: The study employed TiO2 for glycopeptide enrichment, optimizing sample loading conditions with 1% TFA. The authors explored the challenges of enriching sulfated glycopeptides and compared methods used for sialylated glycopeptide enrichment.
LC-MS2 Detection of Sulfated Glycopeptides: LC-MS2 analysis was conducted on rEPO glycopeptides, revealing monosulfated glycopeptides with characteristic MS2 patterns. The study employed HCD MS2 for extensive glycopeptide fragment ion analysis, identifying sulfated oxonium ions for specific sulfation confirmation.
Sialylated Sulfated N-Glycans of Bovine Thyroglobulin: bTG, a sialylated glycoprotein, was analyzed to explore the distribution of sulfated glycoforms. Total N-glycans were released, permethylated, and analyzed by MALDI-MS to determine sulfated and non-sulfated glycans.
LC-MS2 Characteristics of Sialylated Sulfoglycopeptides: LC-MS2 analysis of bTG glycopeptides demonstrated the challenges in detecting sialylated sulfoglycopeptides without prior enrichment. The study highlighted differences in MS2 patterns and sulfite loss between non-sialylated and sialylated sulfoglycopeptides.
Results:
Enrichment Efficiency: TiO2 enrichment efficiency for sulfated glycopeptides was evaluated. Neutralizing or removing sialic acids did not significantly improve enrichment specificity.
MS2 Analysis: HCD and EThcD on an Orbitrap Fusion instrument allowed for the detection of sulfo LacNAc oxonium ions, aiding in sulfoglycopeptide identification. The results emphasized the importance of reliable diagnostic MS2 ions for confident identification.
Sulfoglycopeptide Distribution: The study revealed site-specific distributions of sulfated, sialylated N-glycans on bTG. Different N-glycosylation sites exhibited preferences for specific sulfated glycoforms.
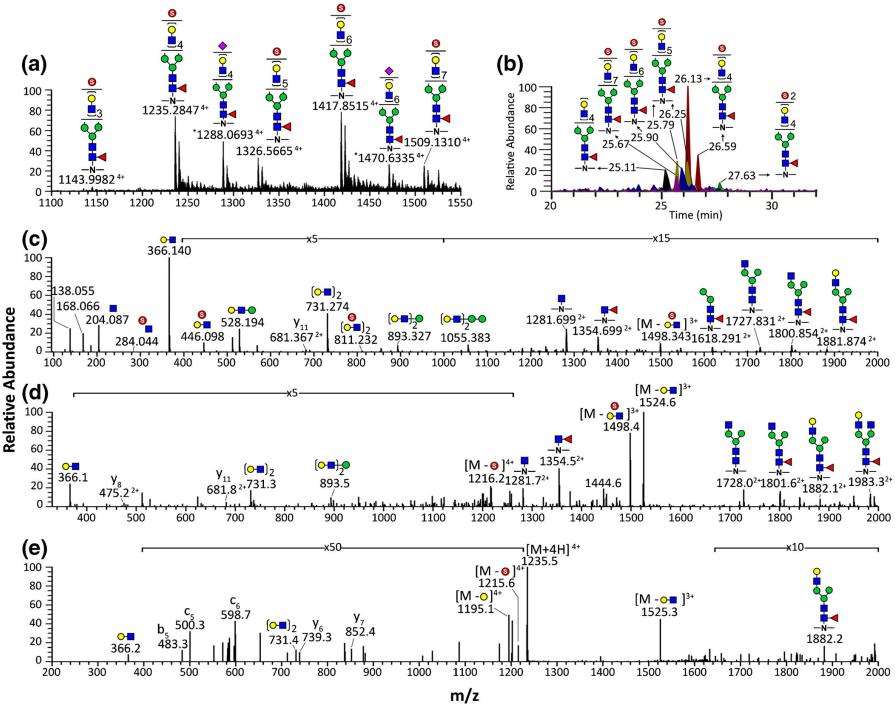 LC-MS and MS2 of TiO2-enriched N-glycopeptides from EPO expressed in BHK21 cells.
LC-MS and MS2 of TiO2-enriched N-glycopeptides from EPO expressed in BHK21 cells.
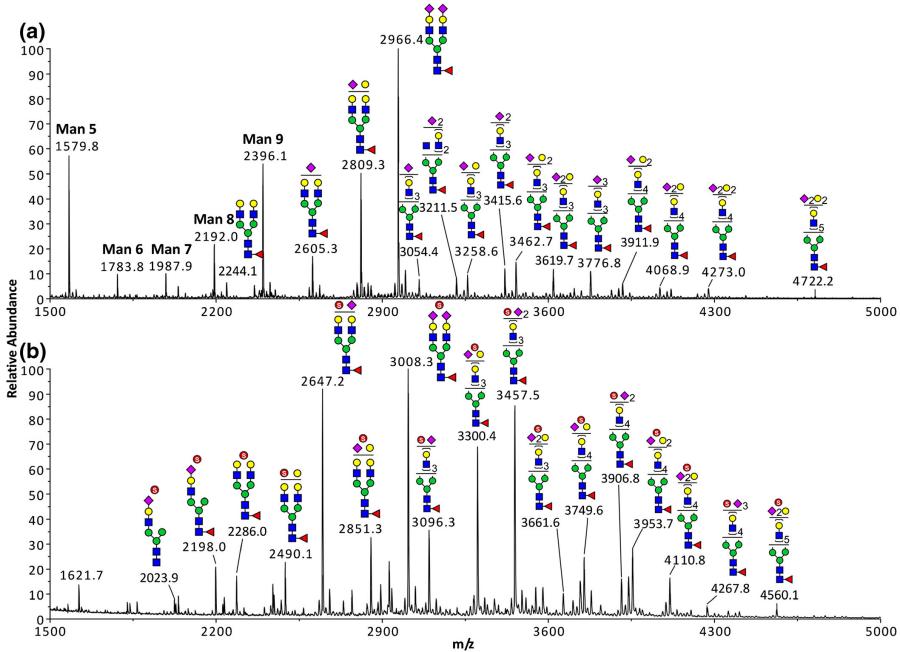 MALDI MS profile of permethylated N-glycans from bTG in positive (a) and negative ion mode (b).
MALDI MS profile of permethylated N-glycans from bTG in positive (a) and negative ion mode (b).
Reference
- Kuo, Chu-Wei, Shih-Yun Guu, and Kay-Hooi Khoo. "Distinctive and complementary MS2 fragmentation characteristics for identification of sulfated sialylated N-glycopeptides by nanoLC-MS/MS workflow." Journal of the American Society for Mass Spectrometry 29.6 (2018): 1166-1178.




